 |
 |
 |
| |
Utility of Historical Data Compared to Lead-In Response in Predicting Sustained Virologic Response in Nonresponders
and Relapsers to Peginterferon/Ribavirin When Retreated With Boceprevir+Peginterferon Alfa-2b/Ribavirin
|
| |
| |
Reported by Jules Levin EASL 2011 Berlin Germany March 30-Apr 2
R. Esteban1*, S. Gordon2, E. Lawitz3, P. Marcellin4, J. Vierling5, S. Zeuzem6, F. Poordad7, N. Boparai8, M. Burroughs8, J. Albrecht8, C. Brass8, B. Bacon9
1Hospital General Universitario Vall d'Hebron, Barcelona, Spain, 2Henry Ford Hospital, Detroit, MI, USA, 3Alamo Medical Research, San Antonio, TX, USA, 4University Paris 7-Hopital Beaujon, Clichy, France, 5Baylor College of Medicine, Houston, TX, USA, 6Universitätsklinikum des Saarlandes, Homburg/Saar, Germany, 7Cedars-Sinai Medical Center, Los Angeles, CA, USA,
8Merck Sharp & Dohme Corp., Whitehouse Station, NJ, USA, 9Saint Louis University School of Medicine, St. Louis, MO, USA; *resteban@vhebron.net
AUTHOR CONCLUSIONS
These findings support a 4-week lead-in with P/R to provide real-time interferon responsiveness.
The high percentage of patients with poor interferon responsiveness (26%) enrolled in a trial that rigorously excluded such patients highlights the unreliability of historical responses, even in a research setting.
-- Important recognition of the changes that can evolve over time in patients who had previously been treated and are awaiting retreatment
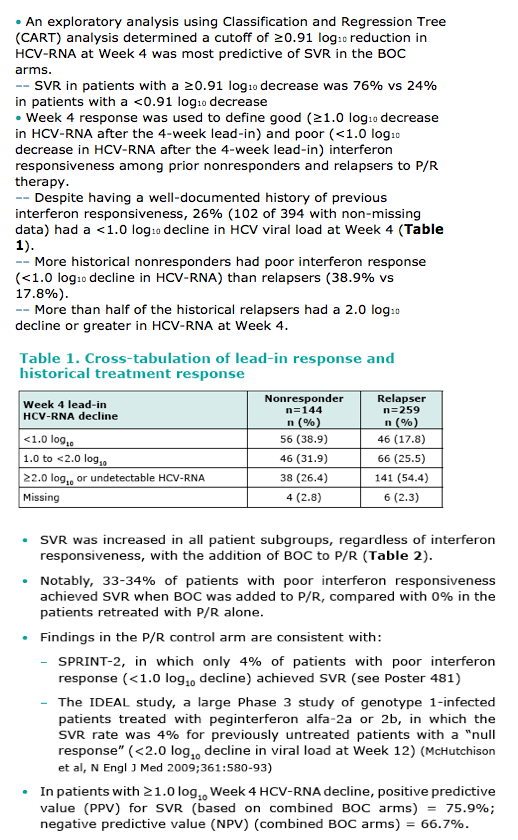
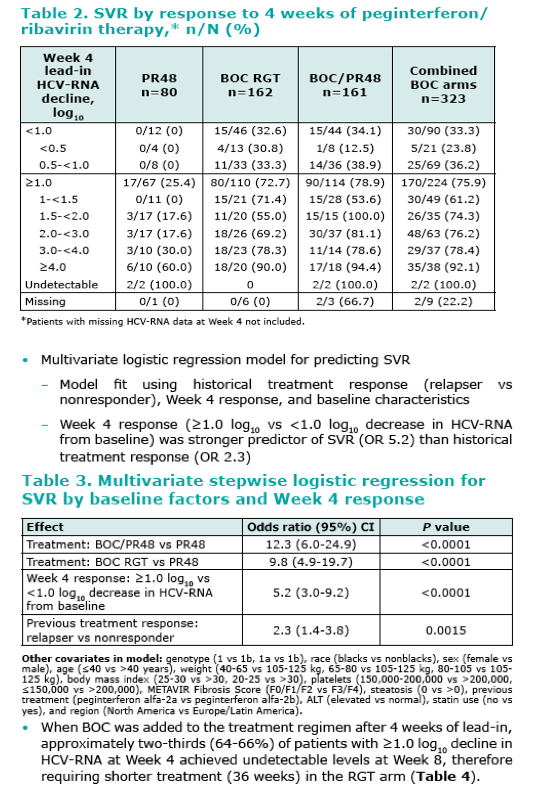
While HCV-RNA response to lead-in with P/R predicts SVR, it should not be used to define futility because one-third of patients with a <1.0 log10 decline HCV-RNA after the 4-week lead-in achieved SVR when BOC was added to P/R.
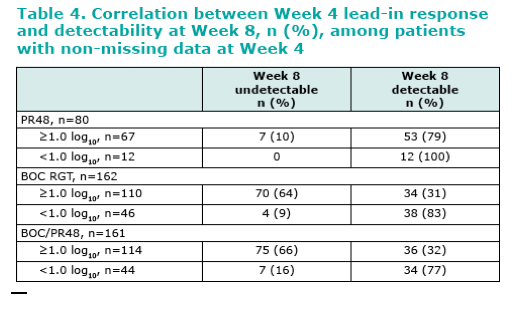
When BOC was added to the treatment regimen after 4 weeks of P/R lead-in, approximately two-thirds (64-66%) of patients with ≥1.0 log10 decline in HCV-RNA at Week 4 achieved undetectable levels at Week 8, therefore requiring shorter treatment (36 weeks) in the RGT arm.
Assessment of response after 4 weeks of P/R lead-in may be a more reliable approach to categorizing previous P/R treatment failures.
BACKGROUND
· In Phase 3 studies, addition of boceprevir (BOC), an HCV NS3 protease inhibitor, to peginterferon (P) and ribavirin (R) in previously untreated and previous-treatment-failure patients with hepatitis C virus genotype 1 (HCV-G1) produced superior sustained virologic response (SVR) rates compared
to standard of care (SOC) treatment with P/R.
· In the clinical trials, a 4-week P/R lead-in period preceded the addition of BOC to the P/R treatment regimen.
· Potential clinical benefits of 4-week P/R lead-in HCV-G1 patients (see Poster 481):
-- Reduction of viral load prior to introduction of direct-acting antiviral (DAA) agent
-- Prevention of exposure to DAA agents in patients who cannot tolerate P/R therapy
-- Accurate assessment of interferon responsiveness - strongest predictor of SVR
-- Determination of tolerance to and compliance with P/R therapy
-- Assessment of risk vs benefit when decision to treat unclear
PURPOSE
Outcomes are suboptimal after retreatment of patients with HCV-G1 infection who were nonresponders or relapsers after prior P/R
therapy. In the RESPOND-2 trial, the lead-in period allowed the comparison of historical categories of nonresponse with lead-in
responses in predicting SVR.

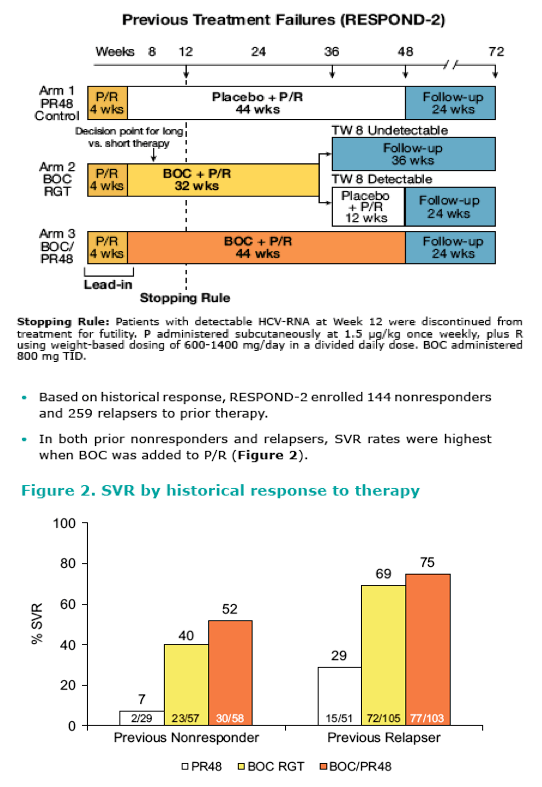
· An exploratory analysis using Classification and Regression Tree (CART) analysis determined a cutoff of ≥0.91 log10 reduction in HCV-RNA at Week 4 was most predictive of SVR in the BOC arms.
-- SVR in patients with a ≥0.91 log10 decrease was 76% vs 24% in patients with a <0.91 log10 decrease
· Week 4 response was used to define good (≥1.0 log10 decrease in HCV-RNA after the 4-week lead-in) and poor (<1.0 log10 decrease in HCV-RNA after the 4-week lead-in) interferon responsiveness among prior nonresponders and relapsers to P/R therapy.
-- Despite having a well-documented history of previous interferon responsiveness, 26% (102 of 394 with non-missing data) had a <1.0 log10 decline in HCV viral load at Week 4 (Table 1).
-- More historical nonresponders had poor interferon response (<1.0 log10 decline in HCV-RNA) than relapsers (38.9% vs 17.8%).
-- More than half of the historical relapsers had a 2.0 log10 decline or greater in HCV-RNA at Week 4.
|
| |
|
 |
 |
|
|