 |
 |
 |
| |
Pharmacokinetics, Safety and Tolerability of the HCV NS5A Inhibitor ABT-267 Following Single and Multiple Doses in Healthy Adult Volunteers
|
| |
| |
Reported by Jules Levin
EASL 2011, Berlin, Germany, March 30-April 3, 2011
Emily Dumas, Adebayo Lawal, Rajeev Menon, Thomas Podsadecki, Walid Awni, Sandeep Dutta, Laura Williams
Global Pharmaceutical Research and Development, Abbott Laboratories, Abbott Park, IL, USA
AUTHOR CONCLUSIONS
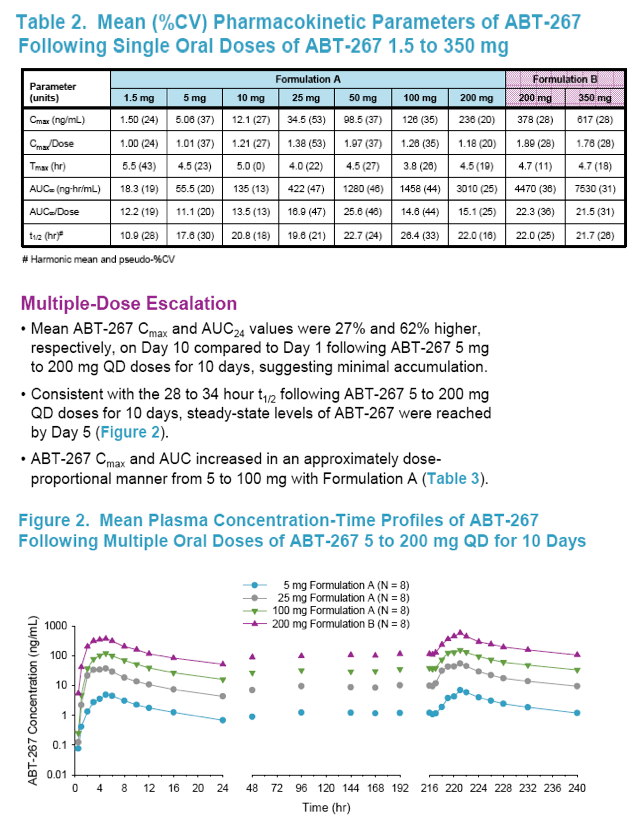
The pharmacokinetics of ABT-267 were approximately dose-proportional with minimal accumulation following multiple dosing.
The bioavailability of Formulation B was higher relative to Formulation A.
Consistent with the 28 to 34 hour t1/2 observed following multiple dosing, steady-state levels of ABT-267 were reached by Day 5.
ABT-267 mean Cmax and AUC values increased by 68% and 62%, respectively, when coadministered with ritonavir compared to ABT-267 administered alone.
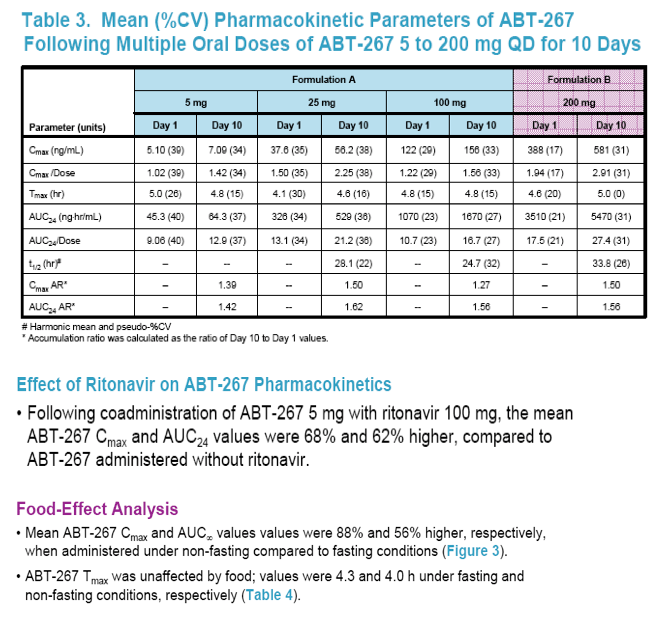
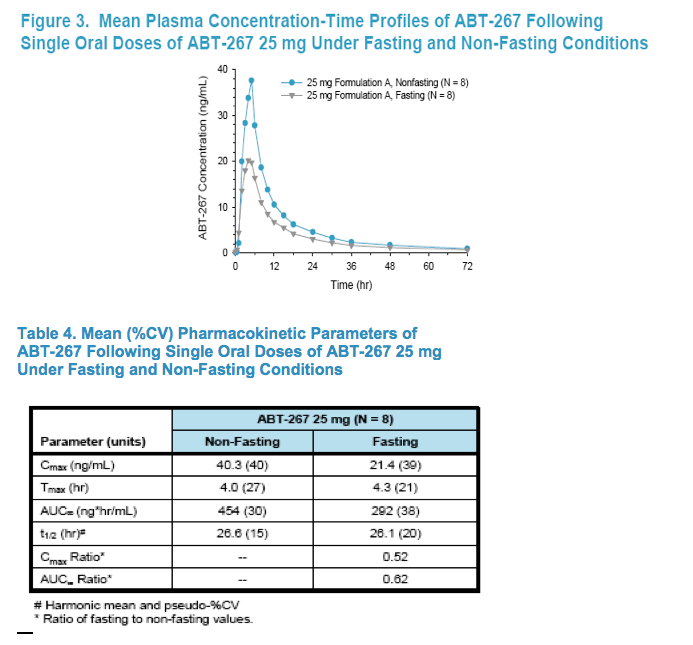
Food had a moderate positive effect on the pharmacokinetics of ABT-267.
ABT-267 was safe and well tolerated across all dose groups.
No association between the character or frequency of adverse events or laboratory abnormalities and ABT-267 dose was observed; the frequency was comparable among the subjects receiving ABT-267 and placebo
Adverse events were infrequent and generally mild. There were no discontinuations in the study.
These findings support continued development of ABT-267 as a once-daily NS5A inhibitor.
Background & Objectives
· ABT-267 is a novel NS5A inhibitor with inhibitory concentrations in the picomolar range against genotype 1a and 1b NS5A proteins in subgenomic replicon systems.
· Abbott is developing ABT-267 to be used in combination with other direct-acting antiviral agents for the treatment of HCV genotype 1 infection.
· A blinded, randomized, placebo-controlled, first-in-human Phase 1 study (registered with ClinicalTrials.gov as NCT01181427) was conducted using two oral formulations of ABT-267.
The objective of this study was to assess the single- and multiple-dose pharmacokinetics, safety and tolerability of escalating doses of ABT-267 in healthy human volunteers.

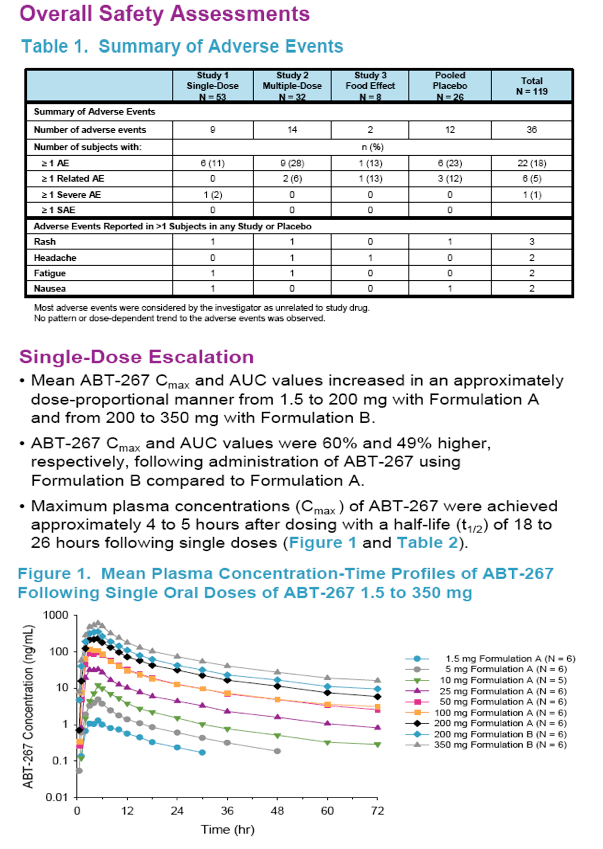
|
| |
|
 |
 |
|
|