 |
 |
 |
| |
REALIZE Trial Final Results: Telaprevir-based Regimen for Genotype 1 Hepatitis C Virus Infection in Patients with Prior Null Response, Partial Response or Relapse to Peginterferon/Ribavirin
|
| |
| |
Reported by Jules Levin EASL 2011 Berlin March 31
S Zeuzem,1 P Andreone,2 S Pol,3 EJ Lawitz,4 M Diago,5 S Roberts,6 R Focaccia,7
ZM Younossi,8 GR Foster,9 A Horban,10 PJ Pockros,11 R Van Heeswijk,12 S De Meyer,12
D Luo,13 G Picchio,13 M Beumont12
1Johann Wolfgang Goethe University Medical Center, Frankfurt am Main, Germany; 2Universita di Bologna, Bologna, Italy; 3Universite Paris Descartes, INSERM Unite 567, and AP-HP Cochin Hospital Paris, France; 4Alamo Medical Research, San Antonio, TX, USA; 5Hospital General de Valencia, Valencia, Spain; 6Department of Gastroenterology, Alfred Hospital, Melbourne, Australia; 7Emilio Ribas Infectious Diseases Institute, Sao Paulo, Brazil; 8Center for Liver Disease, Inova Fairfax Hospital, Falls Church, VA, USA; 9Queen Marys University of London, Institute of Cell and Molecular Science, London, UK; 10Medical University of Warsaw, Wolska, Warsaw, Poland; 11Scripps Clinic and The Scripps Research Institute, La Jolla, CA, USA; 12Tibotec BVBA, Beerse, Belgium; 13Tibotec Inc., Titusville, NJ, USA

from Jules:
The use of telaprevir in combination with peg/rbv increased SVR rates 3-7 times compared to patients receiving only peg/rbv for all patient types of nonresponders.
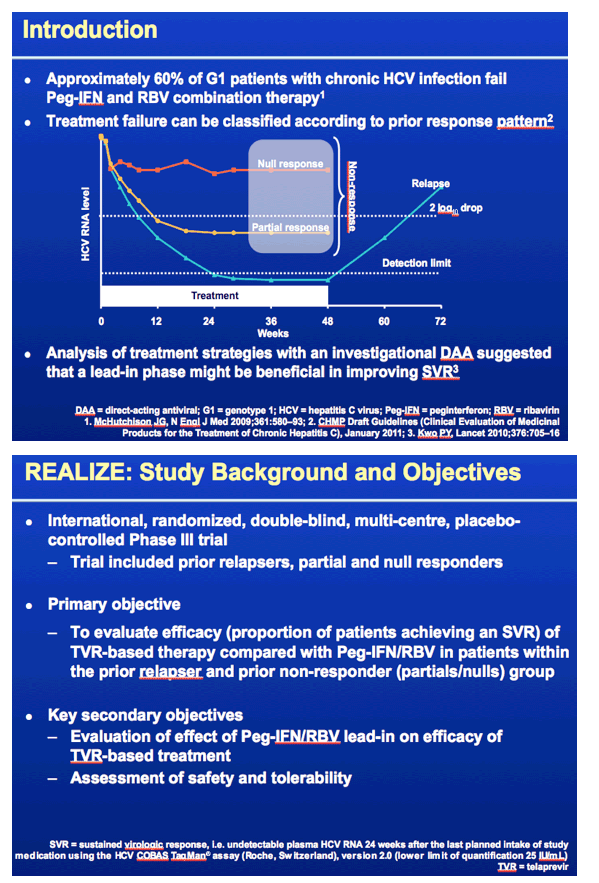
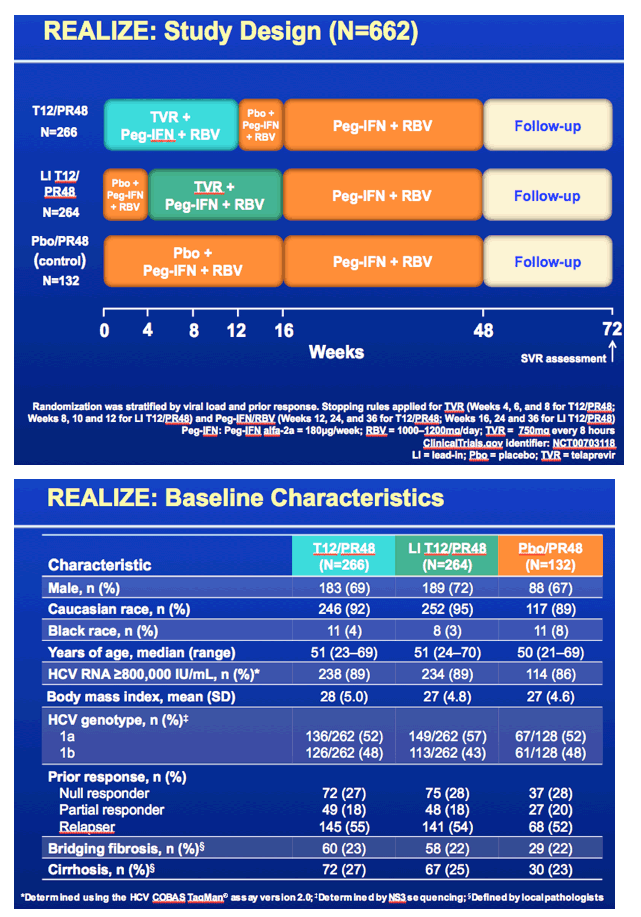
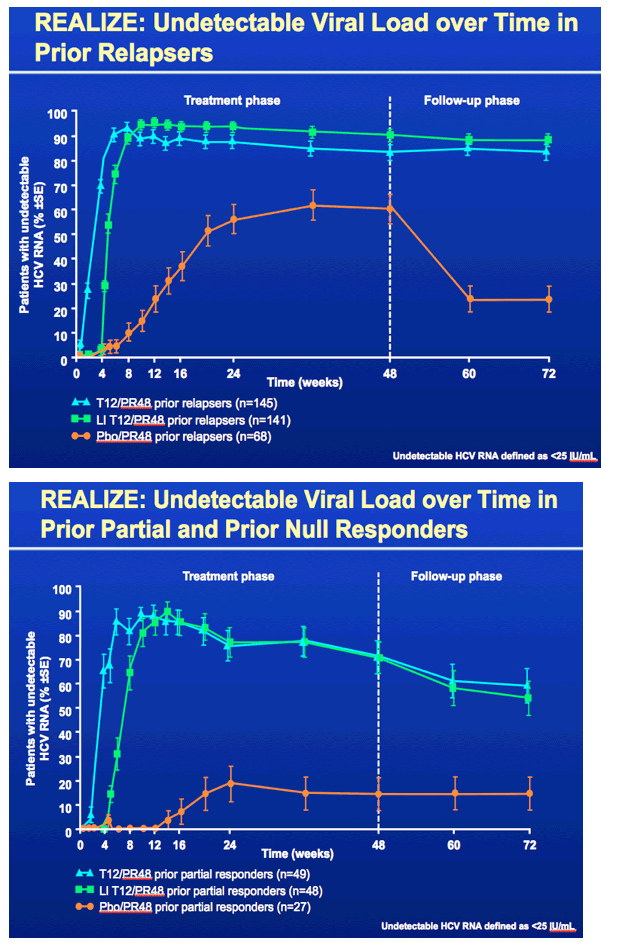
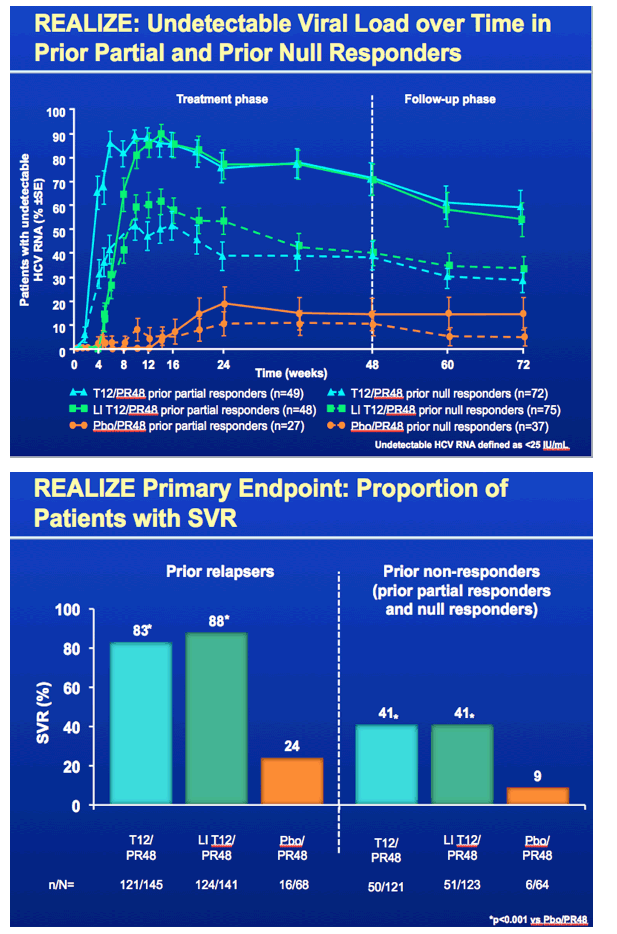
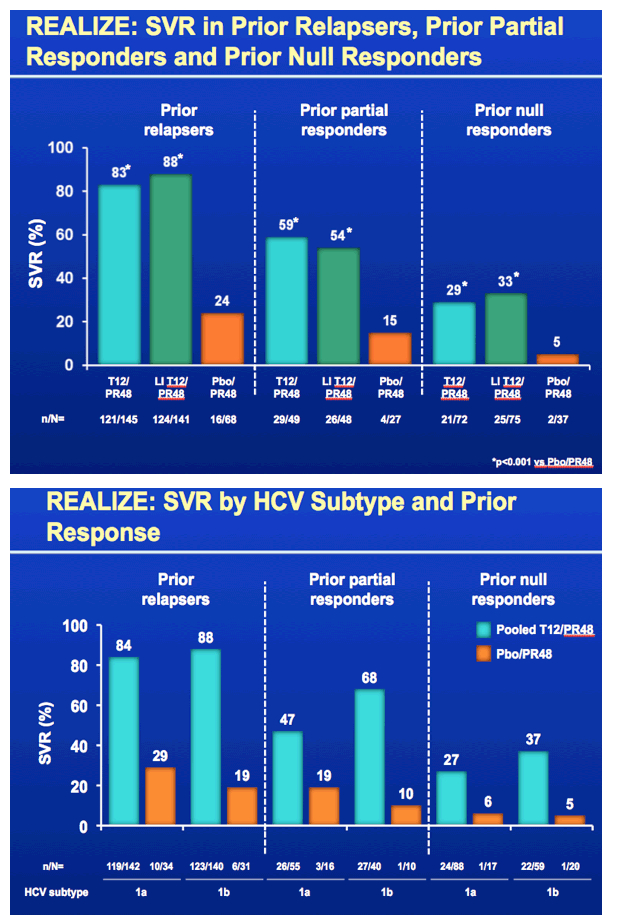
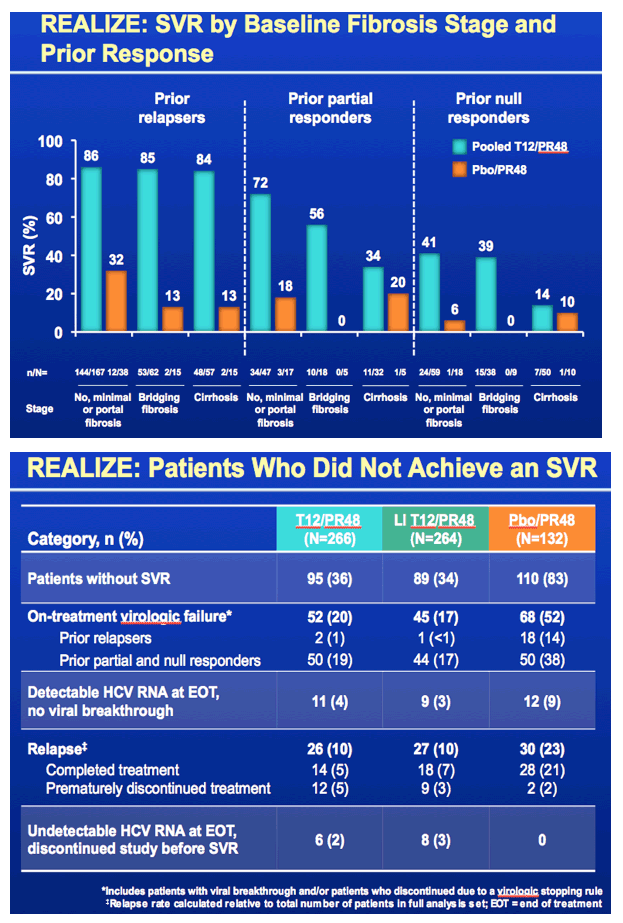
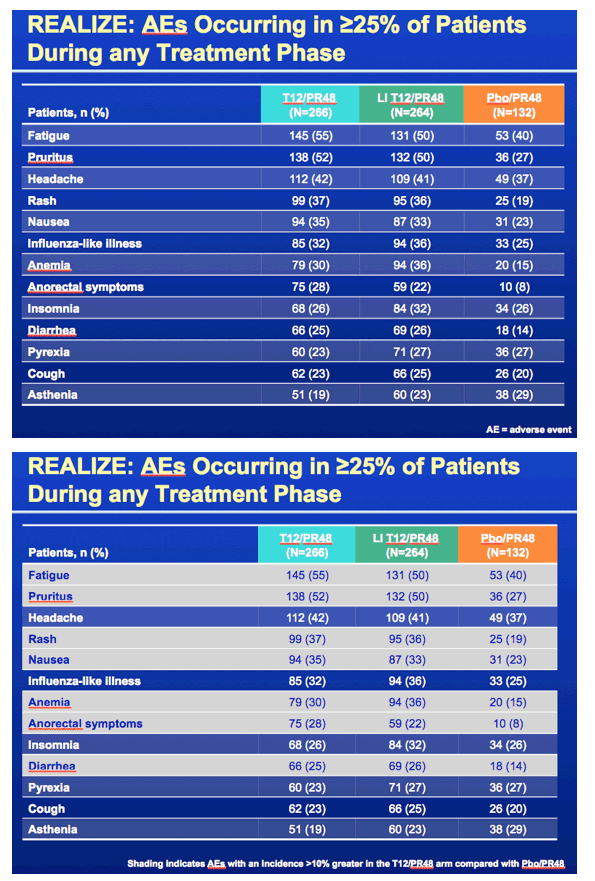
This study REALIZE was globally conducted to evaluate response to telaprevir plus peg/rbv in patients with prior treatment experience who did not achieve SVR, in 3 patient population - prior relapsers, nonresponders and null responders defined in the slide below. Patients were randomized to telaprevir for 12 weeks plus peg/rbv for 48 weeks, a 4-week lead-in of peg/rbv after which telaprevir was added & taken for 12 weeks and also peg/rbv taken for 48 weeks, or they received a placebo plus peg/rbv. 90% of the patients were Caucasians, average age was about 50 (23-70), 88% of patients had high viral load (>800,000 IU/mL), the average BMI was high at 28, in the subanalyses of this study they looked at differences in response by lead-in (this was a major component of the study - did a 4 week lead-in with peg/rbv alone improve SVR rates), they looked at rsponse by stage of cirrhosis, and whether a patient was 1a or 1b genotype, so there are slides below reporting the important results for each of these. 25% of patients in the study had cirrhosis, 23% bridging cirrhosis. There were 72-75 patients who were prior null responders who received telaprevir. RESULTS: There was no statistically significant benefit to taking a 4-week peg/rbv lead-in for relapsers as 83% of prior relapsers who did not use lead-in achieved SVR, 88% who did use a 4-week lead-in achieved SVR but the difference does not appear to be statistically significant, vs 24% receiving only peg/rbv achieved SVR; and for prior partial responders 59% not using the lead-in achieved SVR while 54% using the lead-in achieved SVR, and only 15% using peg/rbv achieved SVR; for prior null responders in this slideset you will see 29% not using lead-in achieved SVR while 33% using lead-in achieved SVR vs 5% using peg/rbv achieved SVR. So there is no apparent benefit for lead-in demonstrated for null responders in this analysis but in a further analysis in the following NATAP report on lead-in they conducted for this study for null responder patients whether the patient had <1 log or >1 log viral load reduction in lead-in did appear to providesome predictive ability in whether the patient achieved SVR as you will see in my next report. Regarding 1a vs 1b genotype there was little difference in SVR rate between 1a & 1b for prior relapsers (84% vs 88%, respectively), but for prior partial responders there was a difference with 47% of 1a patients achieving SVR & 68% of 1b patients achieving SVR, while for prior null responders 27% with 1a achieved SVR and 37% with 1b achieved SVR. The results based on stage of fibrosis is interesting as there are some differences in response. There were no differences for prior relapsers as whether one had minimal fibrosis, bridging fibrosis or cirrhosis SVR rates were 84-86%. For prior partial responders there was a difference with 72% of patients with minimal or portal fibrosis achieving SVR vs 18% who received only peg/rbv, 56% with bridging fibrosis achieving SVR vs 0 for those who received only peg/rbv, and 34% with cirrhosis who achieved SVR vs 20% who received only peg/rbv. For prior null responders 41% with minimal or portal fibrosis (compared to 6% who received only peg/rbv) & 39% with bridging cirrhosis (vs 0% who received only peg/rbv) achieved SVR while for those with cirrhosis 14% achieved SVR (compared to 10% who received peg/rbv alone). The adverse events & side effects are displayed in the slides below with additional fatigue, anemia, pruritis (itching), rash, anorectal symptoms, and diaarhea associated with telaprevir use compared only to peg/rbv use. The rash is described in the slides below as mainly grad 1-2 in severity (3% grade 3), there is a rash management algorithm, it was described below as primarily eczematous & resolved upon cessation of therapy. The anemia was mainly grade 1-2 in severity (6% grade 3), hemoglobin <10 g/dL was observed in 39% taking telaprevir+peg/rbv. Ribavirin dose reduction was the primary management strategy for anemia (dose reduced in 25% of TVR-treated patients). EPO was not permitted to be used in this study. Anorectal signs & symptoms were reported under various terms such as anal pruritis, anorectal discomfort, as well as hemorroids and led to discontinuation in 3 patients and of all study drugs in 2 receiving telaprevir. The discontinuation rates of TVR are low with 5% discontinuing TVR due to rash, 2% due to anemia, 1% or less due to pruritis, and 1% or less due to anorectal signs or symptoms. Remember use of telaprevir is only for 12 weeks.


|
| |
|
 |
 |
|
|