 |
 |
 |
| |
ABT-450/Ritonavir (ABT-450/r) Combined with Pegylated Interferon Alpha-2a and Ribavirin After 3-Day Monotherapy in Genotype 1 HCV-Infected Treatment-naïve Subjects: 12-Week Interim Efficacy and Safety Results
|
| |
| |
Reported by Jules Levin EASL 2011 March 30-April 2 Berlin Germany
Eric Lawitz1, Isabelle Gaultier2, Fred Poordad3, Daniel Cohen2, Rajeev Menon2, Lois M. Larsen2, Thomas Podsadecki2, Barry Bernstein2
1Alamo Medical Research, San Antonio, TX, United States; 2Global Pharmaceutical R & D, Abbott Laboratories, Abbott Park, IL, United States; 3Hepatology, Cedars-Sinai Medical Center, Los Angeles, CA, United States
46th Annual Meeting of the European Association for the Study of the Liver (EASL 2011) March 30 - April 3, 2011 Berlin, Germany
Background
ABT-450 is a potent protease inhibitor of the hepatitis C virus (HCV), identified as a lead compound by Abbott and Enanta, and is being developed for the treatment of HCV genotype 1 infection in combination with other anti-HCV agents
Ritonavir (RTV) co-administration boosts the ABT-450 plasma concentrations, with ABT-450 Cmax and AUC increased 28- to 48-fold respectively1; therefore, ABT-450 is being developed with low dose ritonavir (ABT-450/r) to enhance exposure and allow once-daily dosing
ABT-450/r was safe and well tolerated in single and multiple dose studies in healthy volunteers1, 2
This is the first study of ABT-450/r in HCV-infected subjects. Treatment consisted of monotherapy with ABT-450/r or placebo for 3 days at 3 different doses followed by 12 weeks of ABT-450/r or placebo at the same dose in combination with pegylated interferon + ribavirin (P/R). After week 12, subjects received up to an additional 36 weeks of P/R therapy.
Preliminary data from this study including monotherapy and week 4 results have been presented earlier3, 4
Interim results from a planned analysis through week 12 are presented here
Objective
To analyze the efficacy and safety of various doses of ABT-450/r administered alone for 3 days, followed by co-administration of ABT-450/r with P/R through 12 weeks of treatment in HCV genotype 1 infected subjects

Results
Subject Disposition and Baseline Characteristics
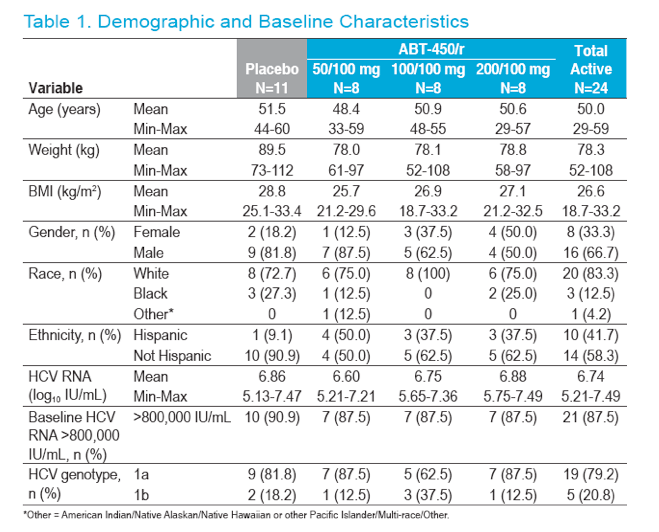
A total of 35 HCV genotype 1-infected treatment-naïve subjects were enrolled and randomized to ABT-450/r (n=24) or placebo (n=11)
One subject receiving ABT-450/r 100/100 mg + P/R withdrew consent after the study day 18 visit at which time the HCV-RNA was <25 IU/mL. Six subjects receiving placebo + P/R discontinued treatment before week 12: three due to virologic failure , two were lost to follow-up, and one discontinued due to pegylated interferon alfa-2a-related severe adverse events. No subjects
discontinued treatment due to DAA-related adverse events.
Demographic and baseline characteristics were similar between groups (Table 1)
- 80% of subjects overall were infected with genotype 1a HCV
- 89% of subjects overall had baseline HCV RNA >800,000 IU/mL
Protocol-defi ned virologic failure = HCV RNA decrease from baseline <1 log10 IU/mL at week 4; or virologic breakthrough
(confirmed detectable HCV RNA following a confirmed HCV RNA value below the LLOD); or virologic rebound (confi rmed increase in HCV RNA of ≥0.5 log10 IU/mL from nadir).
Efficacy Through Week 12
The primary endpoint of the study was the mean maximum change in HCV
RNA during the 3-day monotherapy with ABT-450/r or placebo. Through 3
days of monotherapy, response was similar in the 3 ABT-450/r dose groups and mean maximum decreases from baseline in HCV RNA of 3.91 to 4.11 log10 IU/mL were observed in all 3 dose groups compared to 0.36 log10 IU/mL for the placebo group (P<0.001 for each comparison to placebo)3.
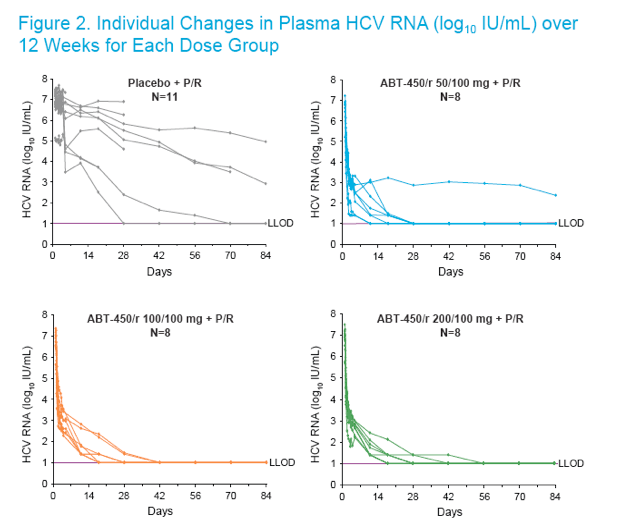
The HCV RNA levels for all subjects in the ABT-450/r + P/R and placebo +
P/R study groups are shown in Figure 2
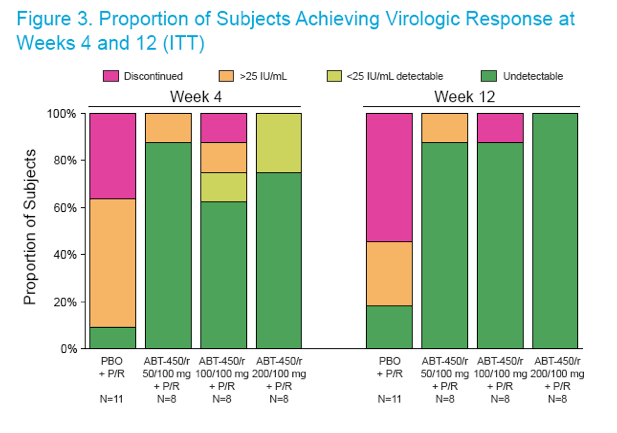
At week 4, when compared to subjects receiving placebo + P/R, a significantly higher proportion of subjects receiving ABT-450/r + P/R achieved RVR (HCV RNA <25 IU/mL) in the intent-to-treat analysis (ITT): 21/24 (87.5%) in the ABT-450/r + P/R and 1/11 (9.1%) in the placebo + P/R dose groups, respectively (P<0.001, Figure 3)4
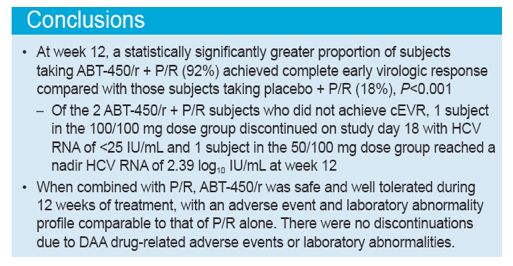
At week 12, 22/24 (91.7%) of subjects receiving ABT-450/r + P/R achieved cEVR (HCV RNA <25 IU/mL) compared with 2/11 (18.2%) of subjects receiving placebo + P/R (P<0.001)
No subject receiving ABT-450/r + P/R experienced a virologic rebound during 12 weeks of treatment
Safety Through Week 12
Overall, ABT-450/r was well tolerated when administered alone and in combination with P/R
No DAA-related serious or severe adverse events were reported
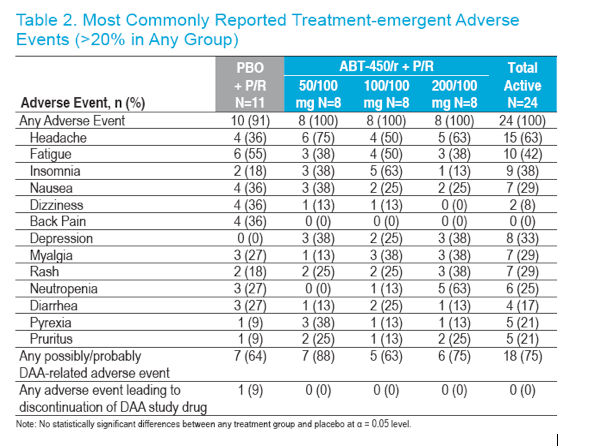
Similar proportions of subjects reported at least one adverse event in all treatment groups (Table 2)
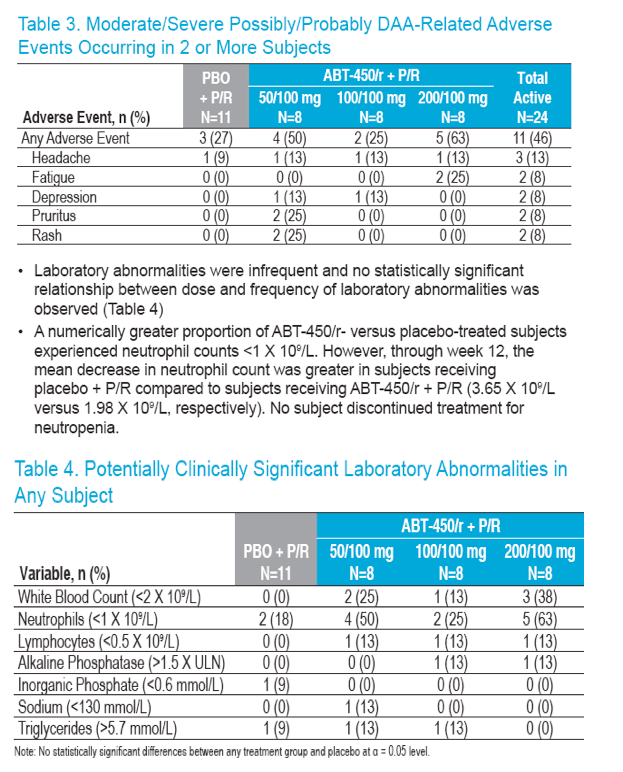
No apparent relationship between dose and frequency of moderate/severe
possibly/probably DAA-related adverse events was observed (Table 3)
references
1. Menon R, et al. Pharmacokinetics and Tolerability of the HCV Protease Inhibitor ABT-450 Following Single Ascending Doses in Healthy Adult
Volunteers with and Without Ritonavir. Annual Meeting of HepDART 2009; 06-10 December 2009; Kohala Coast, Hawaii. Poster #57.
2. Bernstein B, et al. Pharmacokinetics, Safety and Tolerability of the HCV Protease Inhibitor ABT-450 with Ritonavir Following Multiple Ascending
Doses in Healthy Adult Volunteers. Annual Meeting of HepDART 2009; 06-10 December 2009; Kohala Coast, Hawaii. Poster #58.
3. Lawitz E, et al. Initial antiviral activity of the HCV NS3 protease inhibitor ABT-450 when given with low dose ritonavir as 3-day monotherapy:
Preliminary results of study M11-602 in genotype 1 (GT1) HCV infected treatment-naïve subjects. AASLD 2010; 30 October-3 November 2010;
Boston, MA; Abstract #1855.
4. Lawitz E, et al. 4-Week Virologic Response and Safety of ABT-450 Given with Low-dose Ritonavir (ABT-450/r) First As 3-Day Monotherapy then
in Combination with Pegylated Interferon Alpha-2a and Ribavirin (SOC) in Genotype 1 (GT1) HCV-infected Treatment-naïve Subjects. AASLD
2010; 30 October-3 November 2010; Boston, MA. Abstract #LB10.
5. Gaultier I, et al. 12-Week Effi cacy and Safety of ABT-072 or ABT-333 with Pegylated Interferon + Ribavirin, Following 3-Day Monotherapy in
Genotype 1 HCV-Infected Treatment-Naïve Subjects. APASL 2011; 17-20 February 2011; Bangkok, Thailand. Abstract #PP13-95.
|
| |
|
 |
 |
|
|