 |
 |
 |
| |
Nonclinical and Cross-genotypic Profiles of GS-9669, a Novel HCV NS5B Non-nucleoside Thumb Site II Inhibitor
|
| |
| |
Reported by Jules Levin EASL 2011 March 31-Apr 2 Berlin Germany
Martijn Fenaux, Yang Tian, Mike Matles, Eric M. Mabery, Jingyu Zhang, Stacey Eng, Bernard P. Murray, Judy Mwangi, Scott E. Lazerwith, Willard Lew, Eda Canales, Qui Liu, Daniel Byun, Edward Doerffl er, Hong Ye, Michael O. Clarke, Michael Mertzman, Philip Morganelli, Jennifer Zhang, Stephanie A. Leavitt, Todd C. Appleby, Ahmad Hashash, Alison M. Bidgood, Steve H. Krawczyk, William J. Watkins
Introduction
· GS-9669 is a novel HCV replication inhibitor
· GS-9669 inhibits the NS5B RNA-dependent RNA polymerase (RdRp) activity by non-covalently binding to the Thumb Site II pocket
Author Summary
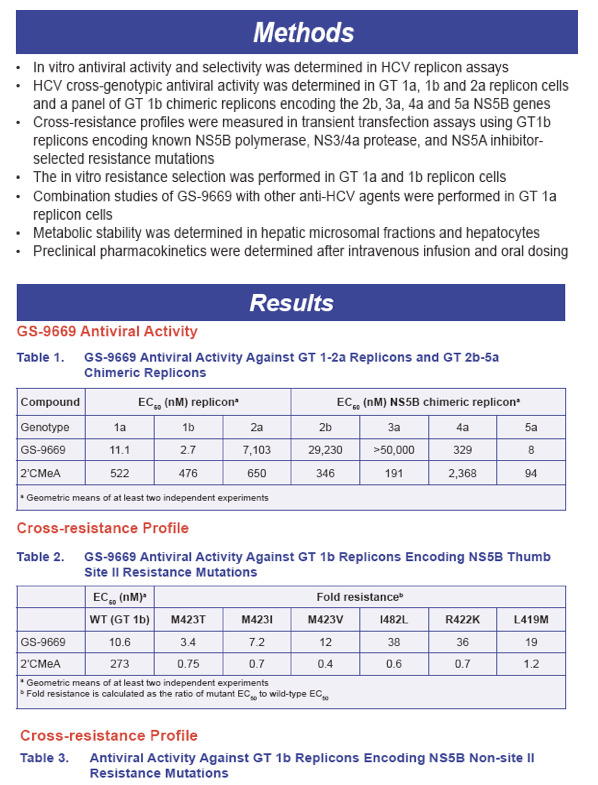
· GS-9669 is a potent inhibitor of HCV GTs 1a, 1b and 5a replication with EC50s of 11.1nM, 2.7nM and 8nM, respectively
· GS-9669 is significantly less potent against HCV GT 2a replicon, and GT 1b chimeric replicons encoding the GT 2b, GT 3a and GT 4a NS5B genes
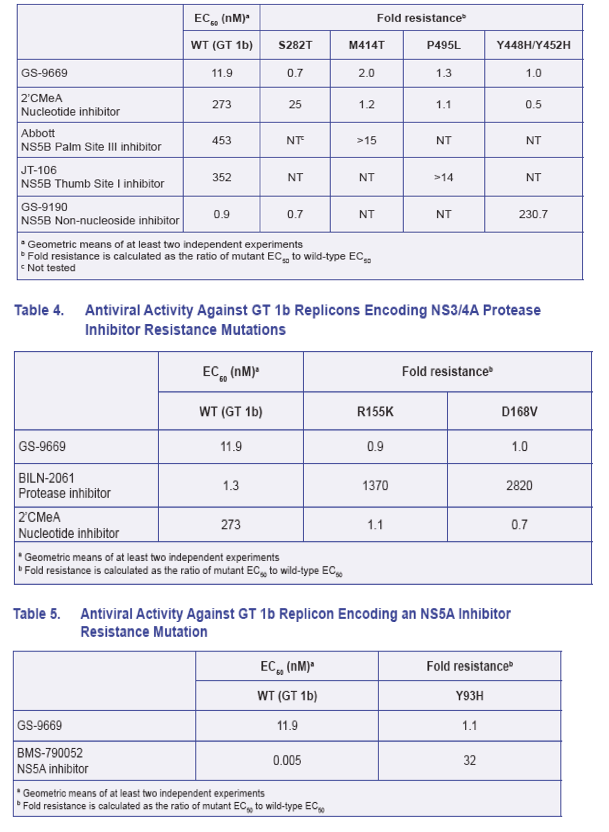
· GS-9669 is fully active against HCV replicons encoding signature resistance mutations to NS3/4A protease inhibitors, NS5A inhibitors, NS5B nucleotide inhibitors, and non-nucleotide non-thumb site II NS5B inhibitors
· GS-9669 exhibits low potency shifts for the common M423T/I/V site II resistance mutations
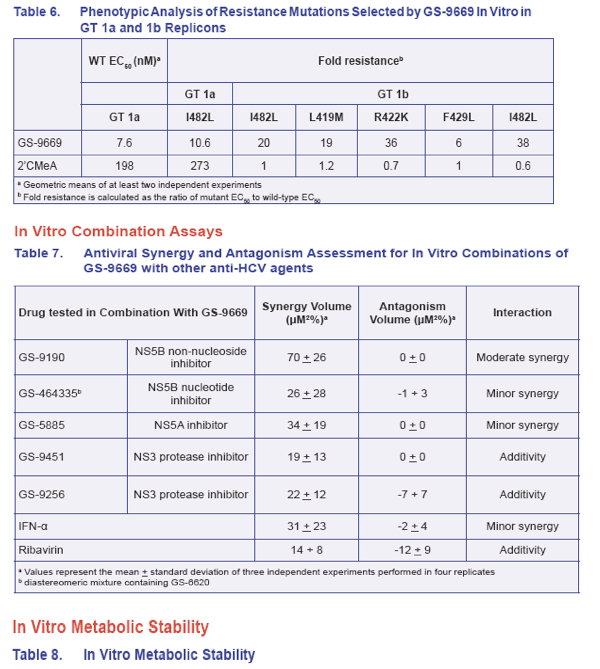
· GS-9669 selected for thumb site II resistance mutations I482L and L419M in GT 1a and I482L, L419M, R422K, and F429L in GT 1b replicon cells
· GS-9669 in combination with IFN-α, ribavirin, and other HCV DAAs discovered at Gilead Sciences resulted in additive to moderate synergistic interactions
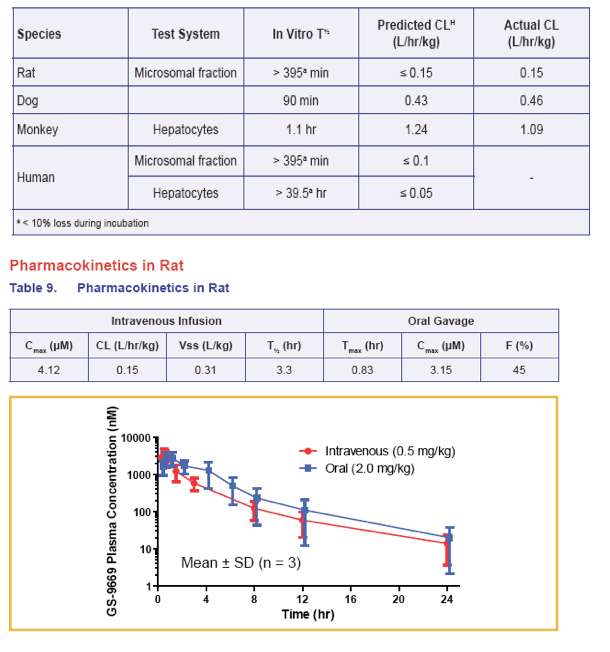
· Pharmacokinetic studies in rat revealed low clearance, moderate volume of distribution, and moderate bioavailability
· Clearance in rat, dog and monkey is well predicted by in vitro metabolism and GS-9669 is stable in human hepatic microsomal fractions and hepatocytes

Background
· Effective treatment of chronic hepatitis C will likely require a combination of HCV-specific inhibitors with non-overlapping resistance profiles to fully suppress the emergence of resistant mutants and eradicate the virus
· The NS5B polymerase of HCV, an RNA-dependent RNA polymerase, is required for HCV replication
· Filibuvir and VX-222, both non-nucleoside allosteric NS5B thumb site II inhibitors, produced multilog10 declines in HCV RNA loads in genotype 1 HCV patients1,2,3
Objective
· Assess the antiviral activity, cytotoxicity, genotypic coverage, cross-resistance and resistance selection profiles of GS-9669
· Assess GS-9669 antiviral activity in combination with IFN-α, ribavirin, and other HCV direct acting antivirals (DAA) discovered at Gilead Sciences
· Gather in vitro and in vivo data to assess the feasibility of oral dosing of GS-9669 in humans
|
| |
|
 |
 |
|
|