 |
 |
 |
| |
NOVEL, POTENT, PAN-GENOTYPIC HCV NS3/4A PROTEASE INHIBITORS WITH A HIGH BARRIER TO RESISTANCE from Roche/Intermune
|
| |
| |
Reported by Jules Levin EASL 2021 Berlin March 30-Apr 2
B.O. Buckman, K. Kossen, J.B. Nicholas, V. Serebryany, D. Ruhrmund, R. Rajagopalan, S. Misialek, L. Hooi, J.-X. Huang, N. Aleskovski, X. Qin, A. Arfsten, S.R. Lim, H. Ramesha, L. Pan, L. Huang, C.J. Schaefer, S. Pietranico-Cole2, S. Le Pogam2, I. Nájera2, K. Klumpp2, S. Seiwert
InterMune, Brisbane, CA, USA; 2Hoffmann La Roche, Nutley, NJ, USA
"We have developed a series of inhibitors that display both pan-genotypic NS3/4A activity and are equally potent against WT and R155K NS3. An advanced analog, ITMN-10082, is described."

ABSTRACT
Background: NS3 protease inhibitors (PIs) have emerged as useful components
in HCV treatment regimens. However, these compounds typically display a low
barrier to virologic escape and a restricted activity across HCV genotypes. Next
generation inhibitors with improvements in these two properties would be highly
desirable as components of direct acting antiviral agent (DAA) cocktail therapy.
Materials and Methods: Structure-guided rational drug design employing both
WT and mutant NS3 proteases was used to design compounds.
Results: Building on work we have previously presented at this meeting (J.
Hepatol., 50, 2009, S341), a lead series of NS3 protease inhibitors was identified.
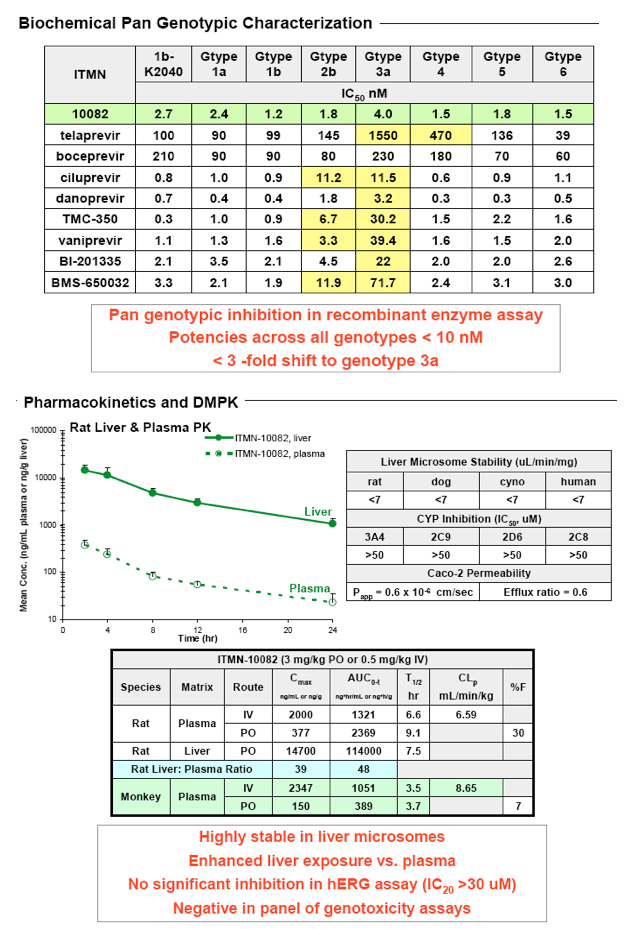
High potency was observed in biochemical assay against NS3/4A derived from
genotypes 1a, 1b, 2a, 3, 4, 5 and 6 (IC50 <4 nM). In biochemical assays against
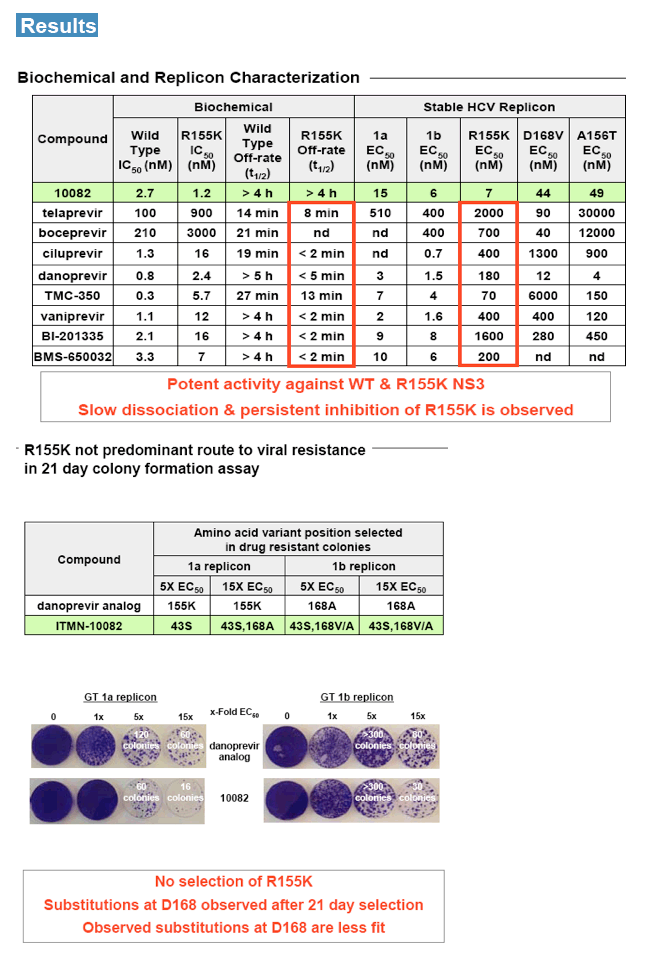
WT or R155K NS3/4A, similar potency (IC50 ~ 1 nM) and slow off-rate (t1/2 ≥4h) were observed. In HCV replicon assays, potent activity was observed against 1a, 1b and R155K-bearing NS3/4A (EC50 ~ 6 nM, 2 nM and 3 nM, respectively).
Resistance in vitro using GT 1a and 1b replicons was mapped to NS3 amino acid
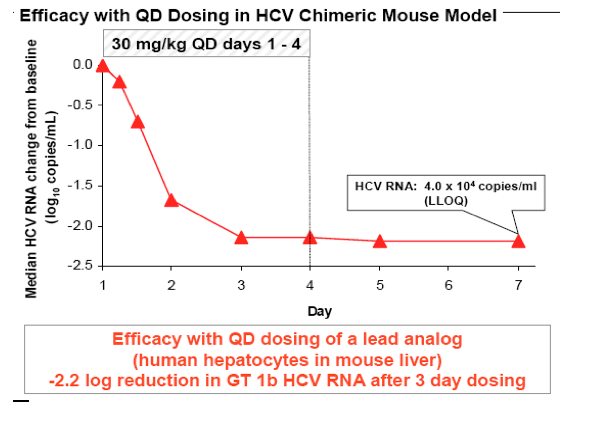
residue 168, with no selection of R155K mutant. Pharmacokinetic analysis of lead compounds in rat, dog and monkey suggested QD administration would provide efficacious exposure in humans. The antiviral activity of a lead compound was evaluated in chimeric mice chronically infected with HCV. Following daily 30
mg/kg doses, viral load was driven below the limit of quantification.
Conclusions: We report biochemically pan-genotypic NS3/4A PIs that are equally potent against WT and R155K NS3. These inhibitors have pharmacokinetic performance in preclinical species suggestive of QD administration. Evaluation of a lead compound in mice with a chimeric human liver showed rapid and sustained suppression of viral load. Further study of these compounds is warranted.
BACKGROUND
» In short duration human clinical studies, administration of NS3/4A protease inhibitors (PIs) can lead to multi-log10 reductions in HCV RNA
» PI monotherapy results in viral breakthrough due to the emergence of drug resistant viral variants
» PI in combination with SOC (pegylated interferon + RBV) suppresses the emergence of drug resistant viral variants except in:
- Treatment naive patients with suboptimal interferon responses
- Many patients who failed prior SOC therapy
» Current NS3/4A inhibitors are not equally active against all HCV genotypes which may restrict their use to certain genotypes
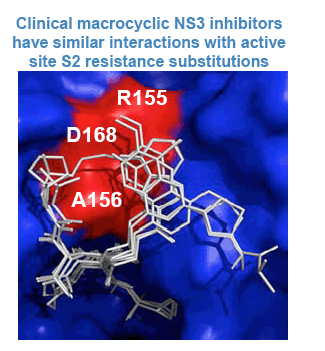
» R155K confers a resistant phenotype to both linear tetrapeptides & macrocycles
» Replicon and virus variants with substitutions at position 155 are generally fit and have a lower genetic barrier in genotype 1a compared to 1b (1 nucleotide change vs. 2)
- Other substitutions are less fit, including those at A156 & D168
» NS3/4A PIs that are active against R155K could have value either as a frontline therapeutic option or for treatment of HCV variants resistant to the current generation of PIs
» We have developed a series of inhibitors that display both pan-genotypic NS3/4A activity and are equally potent against WT and R155K NS3. An advanced analog, ITMN-10082, is described.
|
| |
|
 |
 |
|
|