| |
Interferon-Free Treatment Regimens for Hepatitis C: Are We There Yet? Editorial - pdf attached
|
| |
| |
Download the PDf here
Gastroenterology Dec 2011
Anna Lok, Pratima Sharma
"IFN-free regimens are no longer a dream, but a reality that may be available in the clinic in the next 5 years" (from Jules: I think it will be less than 5 yrs)
Combination therapy with pegylated interferon (PEG-IFN) and ribavirin (RBV) was the standard of care for chronic hepatitis C (CHC) for over a decade. Sustained virologic response (SVR) rates vary from 40% to 45% among patients with genotype 1 to 75% to 80% in patients with genotype 2 or 3 infection.1 However, PEG-IFN and RBV treatment are associated with many side effects. In registration trials that enrolled highly selected patients, 13%-15% of patients discontinued treatment early and 25%-42% had dose reductions because of adverse events or laboratory abnormalities.1, 2, 3 Because of the poor tolerability, many patients with CHC have elected not to pursue treatment or were not offered treatment. IFN and RBV are also contraindicated in many conditions, such as autoimmune diseases and severe/uncontrolled psychiatric illnesses.1 Therefore, there is an urgent need for more efficacious and better tolerated therapy for CHC.
Advances in the understanding of the hepatitis C virus (HCV) life cycle have led to the development of many promising direct-acting antiviral agents (DAA) in the last decade (Figure 1).4, 5 Two DAAs-telaprevir and boceprevir-linear inhibitors of NS3/4A serine protease were approved for HCV treatment in the United States in May 2011. Although the approval of boceprevir and telaprevir represents a major breakthrough for the treatment of CHC with SVR rates of 67% and 73%, respectively, in treatment-naïve genotype 1 patients, both drugs require concomitant use of PEG-IFN and RBV to achieve SVR by preventing viral breakthroughs owing to drug resistance.6, 7 Furthermore, both drugs need to be administered every 8 hours and are associated with additional adverse events.
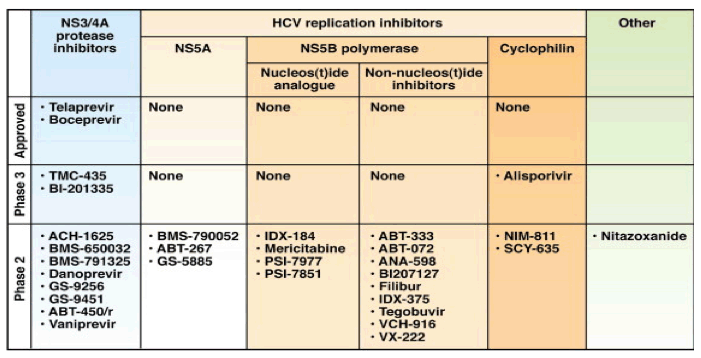
With the development of DAAs directed at multiple targets in the HCV life cycle (Figure 1), the obvious question is, "Are we ready for IFN-free treatment regimens?" Clinical trials involving telaprevir and boceprevir as monotherapy showed a rapid decline in plasma HCV RNA levels within the first day followed by virologic breakthrough as early as day 3.8, 9 HCV circulates as quasispecies, a mixture of viruses with heterogeneous virus sequences. It has been estimated that preexisting drug resistance variants with 1, 2, 3, and even 4 mutations may be present in most HCV-infected patients, and account for the rapid development of drug resistance on exposure to DAAs. The emergence of clinically relevant, drug-resistant variants depends on several factors, such as the potency of the drug, the genetic barrier to resistance, and the replication fitness of the resistance variants.10, 11 Based on modeling experiments, it has been suggested that an IFN-free regimen that can overcome the presence of variants with 4 drug-resistance mutations requires a combination of ≥3 DAAs with a low genetic barrier to resistance, namely, DAAs that select single amino acid resistant variants.12 Each of these drugs should have potent antiviral activity, possess nonoverlapping resistance profiles, and have limited or manageable drug interactions and minimal adverse events. Furthermore, these drugs should be at similar stages of clinical development so that they can be tested in combination.
The first study of combination DAAs, the INFORM-1 study, involved a combination of an NS5B polymerase inhibitor (RG7128) and an NS3/4A inhibitor (danoprevir). This study enrolled treatment-naïve as well as treatment-experienced patients.13 At day 14, 13%-63% of treatment-naïve and 25% of null responders had undetectable HCV RNA (Table 1). None of the patients in any treatment arm experienced virologic breakthrough during the 14-day course of IFN-free regimen suggesting that the addition of RG7128, which has a high barrier to resistance, may have prevented the emergence of resistance to danoprevir.
These promising results have encouraged other studies of combination DAAs. The design and preliminary results of these trials are summarized in Table 1. All the studies reported to date enrolled patients with genotype 1 infection only. Results of 1 phase Ib trial of a combination of an NS3/4A protease inhibitor BI201335, an NS5B polymerase inhibitor BI207127, and RBV are published in the current issue of Gastroenterology.14 In this study, the authors randomized 34 treatment-naïve CHC patients to either 400 or 600 mg TID BI207127, 120 mg once daily BI201335, and weight-based RBV for 4 weeks. All the patients were switched to triple therapy (BI201335 + PEG-IFN + RBV) from day 29 until week 24 or 48, depending on achievement of extended rapid virologic response. The primary endpoint was day 29 virologic response. All 5 genotype 1b but only 6 of 10 genotype 1a patients in the group that received low-dose protease inhibitor achieved day 29 virologic response (Table 1). One genotype 1a patient had virologic breakthrough on day 22, with variants resistant to both drugs (R155K in NS3 and P495L in NS5B) and another patient had an increase in HCV RNA of 0.7 log10 IU/mL from nadir, but sequencing could not be performed because of low HCV RNA level. Both patients had a decrease in HCV RNA to <100 IU/mL after 10 days of BI201335, PEG-IFN, and RBV. All patients (8 genotype 1a and 8 genotype 1b) in the high-dose protease inhibitor group achieved day 29 virologic response. There were no serious adverse events or adverse event-related premature treatment discontinuations, but decreases in hemoglobin, increases in platelet count, and increases in total bilirubin (predominantly indirect) were observed.
These results suggest that an IFN-free regimen comprising 2 DAAs (at the appropriate dose) plus RBV can achieve a very high rate of on-treatment virologic response for up to 4 weeks. However, this study does not address whether IFN-free combination DAAs will result in SVR. It also does not resolve the question of whether RBV contributed to the virologic response and whether RBV reduces relapse with IFN-free regimens. Furthermore, the safety of this combination treatment beyond 4 weeks remains to be determined. Finally, the presence of confirmed a dual drug-resistant variant in 1 patient and a persistent (<1 log) increase in HCV RNA after an initial decline in another patient is concerning, although not surprising, given that both drugs have a low barrier to resistance. Although it has been argued that HCV drug resistance variants are not archived and HCV drug resistance variants become undetectable at a median of 7 months after cessation of telaprevir,15 these data were based on population sequencing, which will not detect variants constituting <20% of the viral population and the real test, that is, response upon retreatment with DAA of the same class, has not been performed.
Another study of combination DAAs involved an NS3 protease inhibitor (GS-9256) and a non-nucleoside NS5B polymerase inhibitor (tegobuvir) with or without PEG-IFN or RBV. The groups that received triple or quadruple therapy achieved higher rates of virologic response at week 4 compared with the group that received dual therapy (Table 1).16 Thirteen of the 16 patients in the dual therapy group, 2 of 15 in the triple therapy group, and none of 15 in the quadruple therapy group experienced virologic breakthrough. Eleven of the patients in the dual therapy arm with breakthrough had variants resistant to both drugs.16 These findings suggest that addition of RBV may accelerate viral clearance, thereby reducing the risk of resistance to DAAs, at least in the short term. The 4th study evaluated a combination of 2 nucleotide RNA-dependent RNA polymerase inhibitors, a purine (PSI-938) and a pyrimidine (PSI-7977) analog. It showed robust and consistent reduction in HCV RNA in the groups that received combination therapy as well as absence of virologic breakthrough up to week 2 (Table 1).17
Collectively, these studies showed that a 14- to 28-day course of the right combination of 2 DAAs dosed appropriately can result in a high rate of virologic response with a low rate of drug resistance, but the likelihood of SVR and risk of drug resistance with longer courses of IFN-free DAA only regimens were not addressed.
To date, SVR data had been reported in only 1 study of combination DAAs (from Jules: at AASLD Nov 2011, Pharmasset reported 100% SVRs in GT2/3 with RBV+PSI-7977, their nucleotide). In this phase II study, genotype-1 null responders were randomized to receive a combination of an NS5A inhibitor (BMS790052) and an NS3 protease inhibitor (BMS650032) alone or together with PEG-IFN and RBV for 24 weeks.18 All 11 patients in the dual therapy arm had a rapid decline in HCV RNA, with 7 achieving undetectable HCV RNA; however, 6 experienced virologic breakthrough and had variants resistant to both DAAs selected (Table 1). Although most of these patients responded to rescue therapy with addition of PEG-IFN and RBV, it is unclear if they will achieve SVR. Four of the 11 patients achieved SVR.12 Responses were more encouraging in the quadruple therapy arm with all 10 patients achieving SVR.12 These data showed that addition of 2 DAAs to PEG-IFN and RBV may result in a greater rate of SVR compared with 1 DAA in nonresponders to PEG-IFN and RBV.19 More important, it provided proof of concept that SVR can be achieved with combination DAAs only.
Similar to Zeuzem et al's study,14 all patients with virologic breakthrough in Lok et al's study had genotype 1a infection.18 Genotypes 1a and 1b HCV may differ in their susceptibility to DAAs. In addition, a larger number of nucleotide changes are required to create a clinically significant protease inhibitor resistance variant for genotype 1b (higher barrier to resistance) than for genotype 1a HCV.10, 11 For example, 2 nucleotide changes are required to generate the resistance mutation R155K for 1b isolates (CGG->AAG), whereas only 1 nucleotide change is required for 1a isolates (AGG->AAG).19 These data indicate that different strategies may be needed for genotype 1a and 1b infection in the era of combination DAAs.
Eight years after the first clinical trial of DAA,20 development of direct-acting HCV treatments is now moving at a rapid pace with many products showing promising results. IFN-free regimens are no longer a dream, but a reality that may be available in the clinic in the next 5 years. It is possible that some of these regimens will also be RBV free. This will be good news for patients who wish to be treated but have to defer treatment because of contraindications to use of PEG-IFN or RBV, or out of concerns about their ability to tolerate these medications. However, caution must be taken in selecting which DAAs to combine and the appropriate dose and duration of therapy for each HCV genotype and subgenotype to prevent multidrug resistance
|
|
| |
| |
|
|
|