 |
 |
 |
| |
Coadministration of the HCV Protease Inhibitor Boceprevir Has No Clinically Meaningful Effect on the Pharmacokinetics
of the Selective Serotonin Reuptake Inhibitor Escitalopram in Healthy Volunteers
|
| |
| |
Reported by Jules Levin 16th Annual Meeting of HEP DART, December 4-8, 2011, Koloa, Hawaii
EGJ Hulskotte,1 S Gupta,2* F Xuan,2 MGJA van Zutven,1 E O'Mara,2* L Galitz,3 JA Wagner,2 JR Butterton2
1MSD, The Netherlands; 2Merck Sharp & Dohme Corp., Whitehouse Station, NJ, USA; 3Cetero Research, Miami, FL, USA
*Former employee of Merck Sharp & Dohme Corp., Whitehouse Station, NJ, USA

ABSTRACT
Background: Boceprevir is a potent, orally administered inhibitor of the hepatitis C virus (HCV) NS3 protease. It has recently been approved for the treatment of chronic hepatitis C (CHC) genotype 1 infection in combination with peginterferon alfa and ribavirin in both the United States and the European Union. Depression is one of the more common comorbidities associated with CHC infection.
In addition, depression has been reported as a common and dose-limiting side effect of peginterferon therapy in patients with CHC. Selective serotonin reuptake inhibitors (SSRIs) are a class of compounds typically used as antidepressants in the treatment of depression, anxiety disorders, and some personality disorders. Escitalopram is one of the most widely used SSRIs.
Biotransformation of escitalopram is mediated primarily by CYP2C19, with minor contribution by CYP3A4 and CYP2D6. Boceprevir is not an inducer of CYP enzymes but is an inhibitor of CYP3A4 and may therefore potentially affect plasma concentrations of escitalopram.
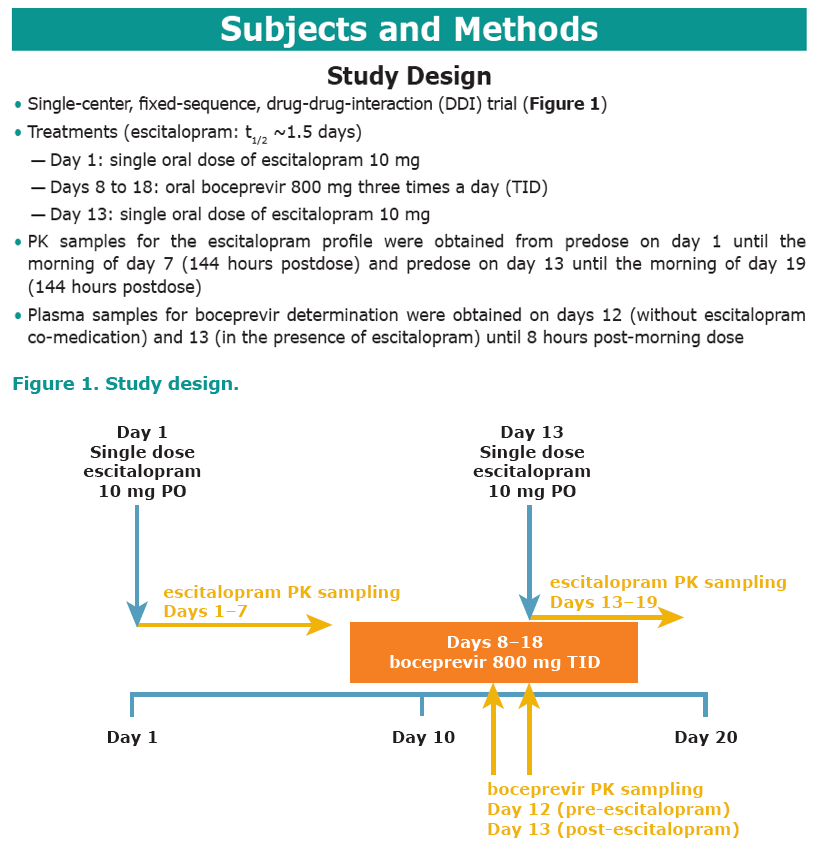
Methods: In an open-label, fixed-sequence design, 10 healthy volunteers received a single 10-mg dose of escitalopram. After a 7-day washout, subjects
received 800 mg boceprevir TID for 11 days. On day 6, subjects concomitantly received a single 10-mg dose of escitalopram. Blood samples were collected for determination of pharmacokinetics of escitalopram and boceprevir. Safety assessments included electrocardiograms, vital signs, clinical laboratory tests, physical examination, Columbia Suicide Severity Rating Scale rating, and adverse event monitoring.
Results: Co-administration of boceprevir with escitalopram was well tolerated in
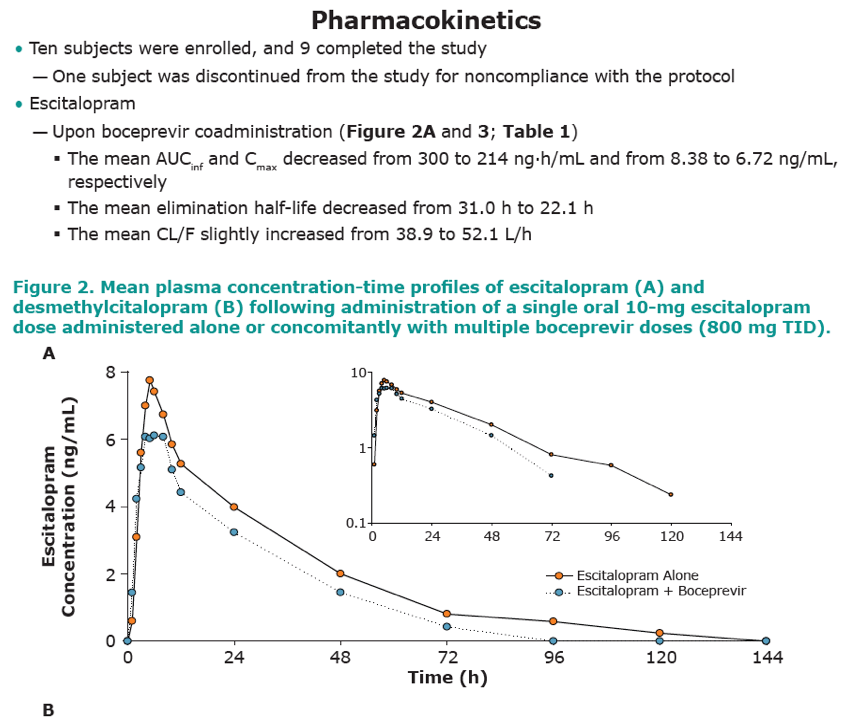
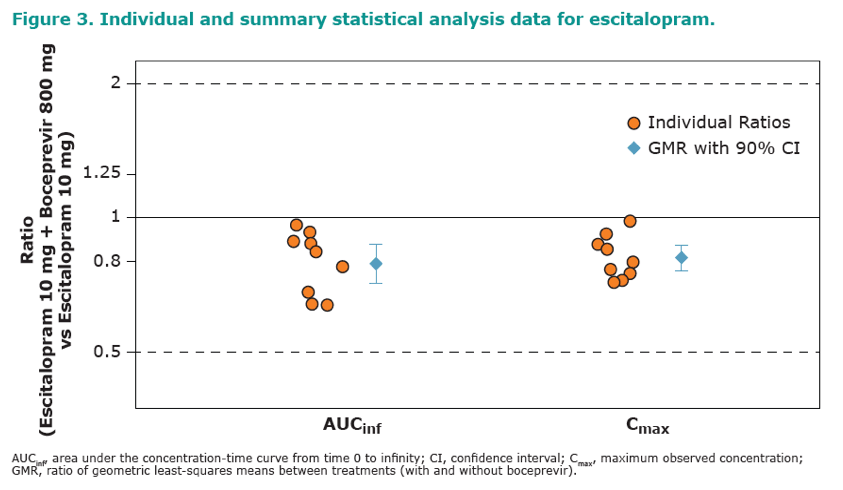
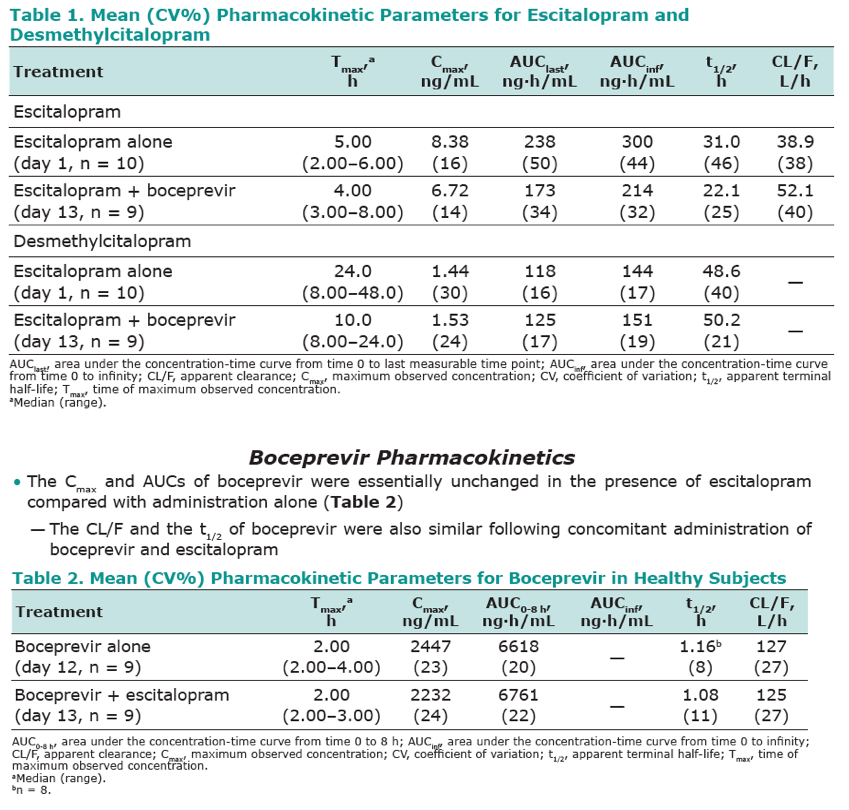
this group of healthy volunteers. Nine of the 10 subjects enrolled completed the trial. One subject was discontinued for noncompliance with the protocol. In the presence of boceprevir, the geometric mean AUCinf of escitalopram decreased from 276 to 218 ng·hr/mL (geometric mean ratio (GMR) [90% CI] 0.79 [0.72, 0.87]) and Cmax decreased from 8.28 to 6.73 ng/mL (GMR [90% CI] 0.81 [0.76,
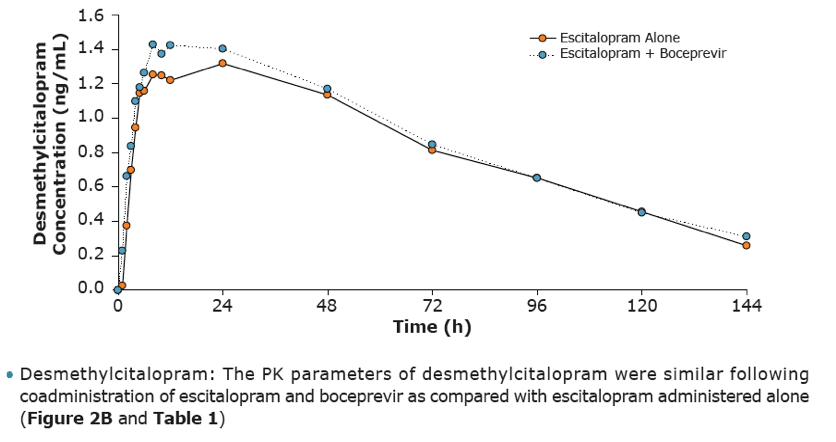
0.87]). The elimination half-life decreased from 31.0 h to 22.1 h. The pharmacokinetic parameters of the major metabolite, desmethylcitalopram were similar following co-administration of escitalopram and boceprevir as compared with escitalopram administered alone. Escitalopram co-administration had no meaningful effect on the pharmacokinetics of boceprevir (AUC0-8 h and Cmax GMR [90%CI] 1.02 [0.96, 1.08] and 0.91 [0.81, 1.02], respectively).
Conclusion: Co-administration of boceprevir had no clinically meaningful impact on the pharmacokinetics of escitalopram. Consequently, no dose adjustment of escitalopram is anticipated to be required with boceprevir co-administration.


|
| |
|
 |
 |
|
|