 |
 |
 |
| |
Antiviral Activity of GS-9451, an HCV Specific NS3 Protease Inhibitor, in a Three-Day, Dose-Ranging, Monotherapy Study
in Patients with Genotype 1 Hepatitis C Infection
|
| |
| |
Reported by Jules Levin
22nd Conference of the Asian Pacific
Association for the Study of the Liver (APASL)
February 16-19, 2012
Taipei, Taiwan
J. Yang1, H. Mo1, H. Dvory-Sobol1, E. Lawitz2, L. Moorehead1, A. Mathias1, W. Delaney IV1, J. McHutchison1
1Gilead Sciences, Inc, Foster City, CA, USA; 2Alamo Medical Research, San Antonio, TX, USA

Background
· GS-9451 is a novel, reversible, non-covalent inhibitor of HCV NS3 serine protease with in vitro equipotent antiviral activity against genotype 1a and 1b
- In vitro EC50 ranging from 7-10 nM in HCV 1a or 1b replicon assays
- In vitro, GS-9451 selected resistance mutations at positions 168 and 156 in NS3 protease
Objectives
· Determine safety, tolerability, and pharmacokinetics of multiple ascending doses of GS-9451 in treatment-naïve, HCV-infected patients
· Characterize the antiviral activity of multiple ascending doses of GS-9451 monotherapy
- Identify mutations in the HCV NS3 gene from HCV patient isolates that are potentially associated with virologic resistance to GS-9451 monotherapy
- Investigate the emergence of resistant quasispecies not detected by population sequencing through deep sequencing
· Determine the persistence of resistance mutations following termination of treatment
Methods
· Randomized, double-blind, placebo-controlled, dose-escalation study conducted in treatment-naïve patients with genotype 1 chronic HCV infection
- Three days of once-daily GS-9451 monotherapy, dosed under fed conditions (active to placebo ratio of 4:1):
· 60 mg GS-9451 or placebo (N=10, GT1a)
· 200 mg GS-9451 or placebo (N=10, GT1a)
· 200 mg GS-9451 or placebo (N=10, GT1b)
· 400 mg GS-9451 or placebo (N=11, GT1a)
· Serial PK (Days 1 & 3)
- Resistance testing
- Population sequencing of the entire NS3/4A coding region at Baseline, Day 4, Day 14, Week 12 and Week 24
- The NS3 protease region (1-181) was analyzed using 454 deep pyrosequencing in 33 longitudinal samples from 7 patients
- Plasmid control was processed in parallel with patient samples to establish the background derived from PCR pre-amplifi cation and 454 sequencing
- Software was developed to accurately analyze deep sequencing results with an assay cut-off of 0.25%
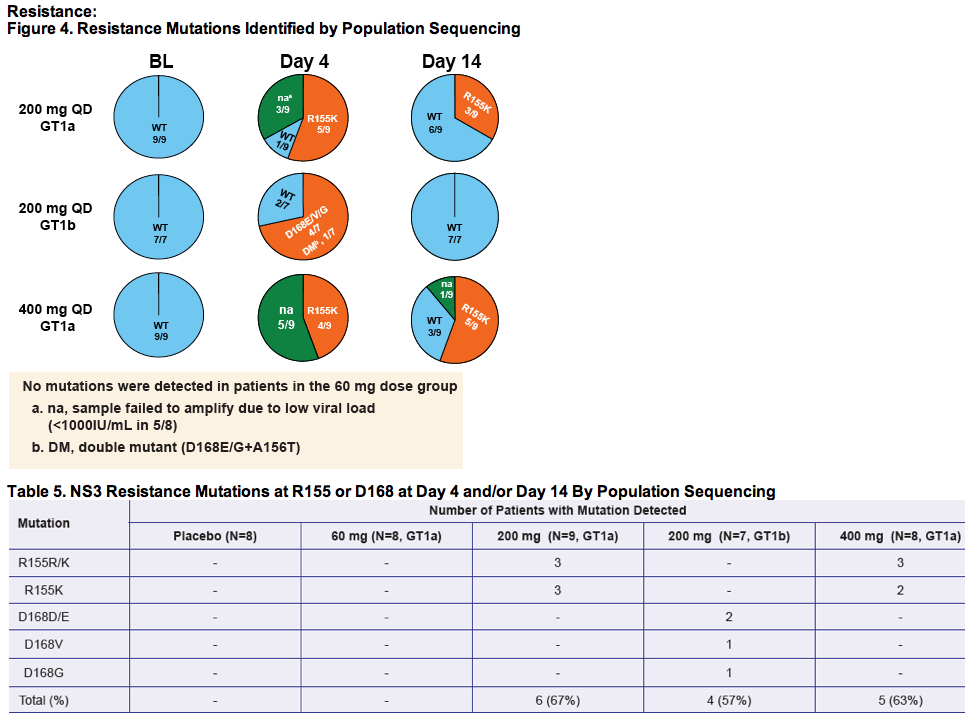
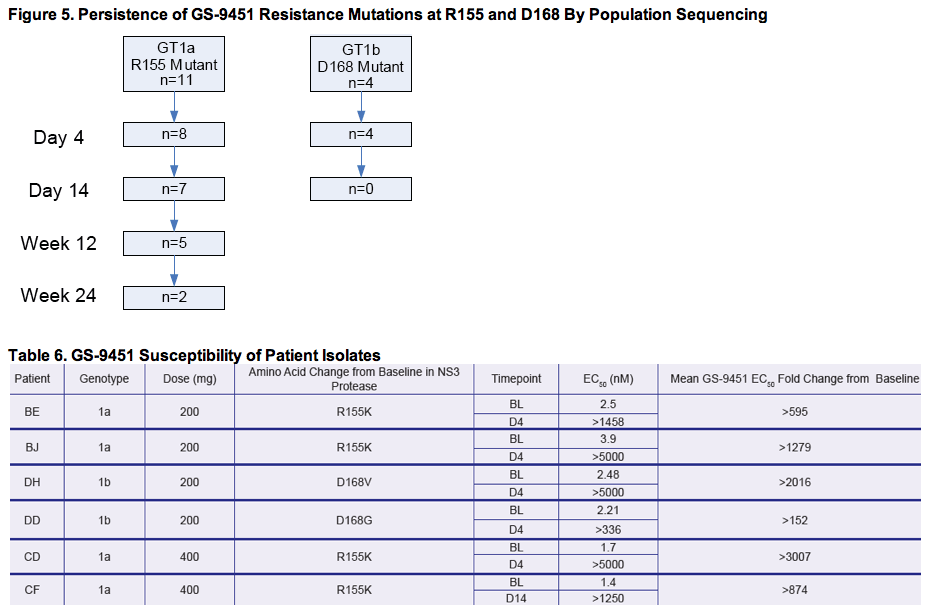
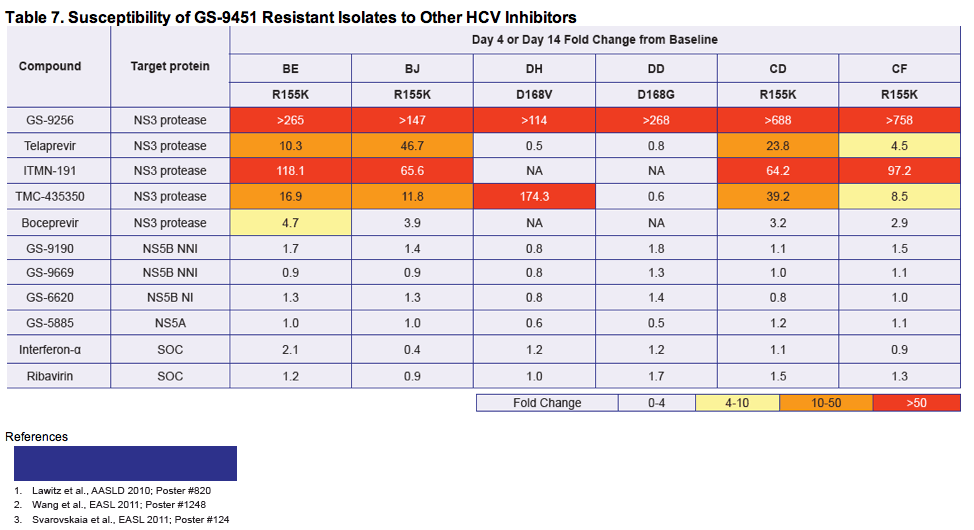
|
| |
|
 |
 |
|
|