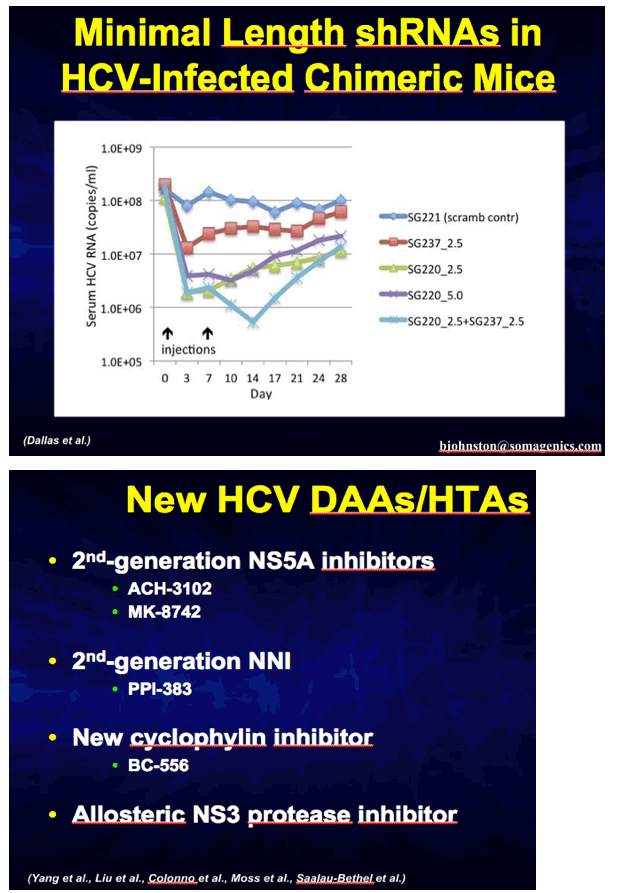
 |
 |
 |
| |
Closing Summary by Jean-Michel Pawlotsky: New HCV Drug Development, Goodbye Interferon?, Triple, QUAD, IFN-Free, Adherence, New HCV Drugs
|
| |
| |
EASL 2012 Sunday Morning Apr 22 Barcelona Spain
From Jules: I added the Program Abstract just below slides which include author & contact info.





PROGRAM ABSTRACT
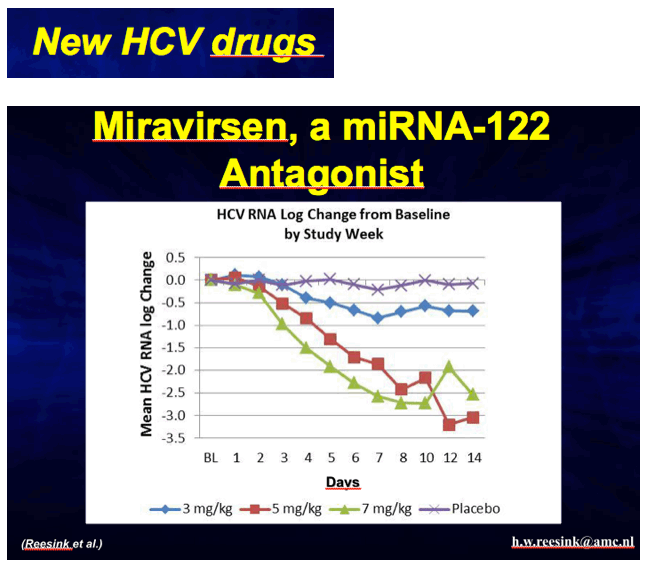
Final Results - Randomized, Double-blind, Placebo-controlled Safety, Anti-viral Proof-of-Concept Study of Miravirsen, an Oligonucleotide Targeting miR-122, in Treatment-naïve Patients with Genoty
Speaker: Henk Reesink Author: H.W. Reesink1*, H.L.A. Janssen2, S. Zeuzem3, E. Lawitz4, M. Rodriguez-Torres5, K. Patel6, A. Chen7, C. Davis7, B. King7, A. Levin7, M.R. Hodges7 Affiliation: 1Department of Hepatology, Academic Medical Center, University of Amsterdam, Amsterdam, 2Department of Gastroenterology and Hepatology, Erasmus MC University Medical Center Rotterdam, Rotterdam, The Netherlands, 3Department of Medicine, J.W. Goethe University Hospital, Frankfurt/Main, Germany, 4Alamo Medical Research, San Antonio, TX, USA, 5Fundacion de Investigacion, San Juan, Puerto Rico, 6Duke University, Durham, NC, 7Santaris Pharma, San Diego, CA, USA. *h.w.reesink@amc.nl
Background: Miravirsen (MIR), a ß-D-oxy-Locked Nucleic Acid (LNA)-modified phosphorothioate anti-sense oligonucleotide, targets liver-specific microRNA-122 (miR-122), an HCV-required host factor. Prior non-clinical studies have demonstrated MIR activity against all HCV genotypes (GT) and long-lasting suppression of HCV viremia, without evidence of viral resistance. MIR has a high genetic barrier to resistance and is not metabolized by p450 enzymes. In prior human studies, MIR was safe and well-tolerated in single (1-12 mg/kg) and multiple-dose (1-5 mg/kg x 5 doses) phase 1 trials; no dose-limiting toxicities were identified.
Methods: This multiple ascending dose phase 2a trial evaluated safety, pharmacokinetics (PK) and anti-viral activity of MIR. Patients with chronic HCV GT1 were enrolled into 3 sequential cohorts (9 active & 3 placebo/cohort). MIR (3, 5, or 7 mg/kg) or placebo (PLB) were administered subcutaneously (SC) weekly (x 5 doses) over 29 days. At the discretion of the investigator, patients could receive standard of care (SOC) anti-viral treatment from Week 7 (Cohort 1) or Week 10 (Cohorts 2 & 3); patients were followed until Week 18.
Results: All patients (27 MIR + 9 PLB) have completed the study. MIR was associated with dose-dependent reductions in HCV RNA (Abbott RealTime, LLOD 23 IU/mL) that were prolonged following dosing (graph). One of 9 MIR patients in the mid-dose group and 4 of 9 in the high-dose group became HCV RNA undetectable without the addition of SOC.
ALT, AST, GGT levels decreased during MIR dosing. No significant nucleotide changes were observed in the HCV miR-122 binding sites. AEs were generally infrequent, mild, and similar between treatment groups. Injection-site events (e.g., pain, hematoma, erythema) were mild, but more frequent with highest MIR dose. No adverse clinically-significant changes in laboratory safety tests, vital signs or ECGs occurred.
Conclusions: MIR is a novel host-targeting agent directed against liver-specific miR-122. In GT1 HCV-infected patients, MIR administration resulted in dose-dependent reductions in HCV RNA without evidence of resistance. MIR was safe and well-tolerated in this study. Further trials of MIR alone and in combination with direct acting anti-viral agents are planned.


|
| |
|
 |
 |
|
|