| |
HCV Update Selected Highlights: key new HCV drugs, timelines & recent news developments
|
| |
| |
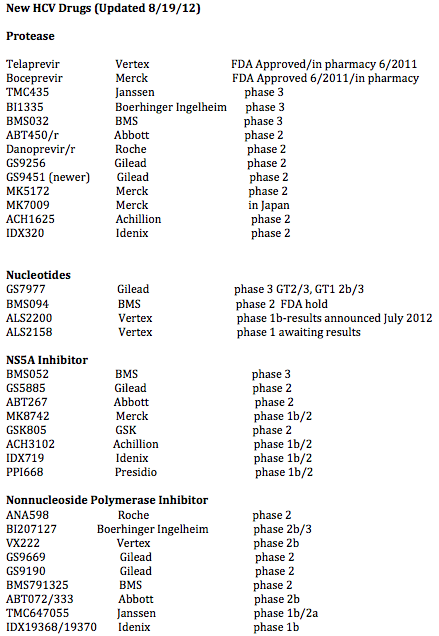
written for NATAP by Jules Levin: Currently approved are 2 protease inhibitors telaprevir & boceprevir. In phase 3 now are 4 drugs: 2 proteases TMC435 & BI1335; nucleotide GS7977; and NS5A BMS052. Also in phase 3 is Peg Lambda, a peginterferon that has showed in trials similar efficacy to current Peginterferons with little of the side effect. Phase 3 for these drugs should be finished in about one year with varying finish timelines between these drugs. Abbott is about to start phase 3 studies, they have 4 drugs in 3 classes: protease, NS5A, HCV polymerase nonnucleoside inhibitors (NNIs). Gilead has drugs in 4 classes: nucleotide, protease, HCV polymerase nonnucleoside inhibitors (NNIs), NS5A. BMS has drugs in 4 clases: NS5A, HCV polymerase nonnucleoside inhibitors (NNIs), nucleotide (trial just suspended, evaluating), protease; Roche/Genentech has drugs in 3 classes: protease, nuke, HCV polymerase nonnucleoside inhibitors (NNIs). Vertex has drugs in 3 classes: protease, nucleotide, HCV polymerase nonnucleoside inhibitors (NNIs). Merck has drugs in 2 classes: protease in development in addition to boceprevir, NS5A. Tibotec has protease TMC435 in phase 3 now with other drugs further back in development.
So in about 1 year we will have 2 brand new classes of drugs BMS052 the potent NS5A, GS7977 the potent nucleotide, plus the 2 new proteases currently in phase 3 BI1335 & TMC435. At this time we don't know if patients & clinicians will be able to combine the NS5A+GS7977 or a 3-drug combination of a protease TMC435 or BI1335 + the NS5A BMS052+GS7977. A small phase 2 study at EASl in April of about 40 patients showed SVR rates of 100% using GS7977+BMS5435, but there is not a followup study ongoing with these 2 drugs. There is an ongoing study now in null responders lookimg at TMC435+GS7977, we await these data, expecting good results. For null responders we have some data below in small studies finding that 2 BMS orals, the protease+NS5A + peg/RBV could cure 100% of patients.
Is there any doubt that we should be able to achieve 100% cure rates for all treatable patients?
Following this chart are links to the key studies recently reported that capture the data for these drugs.

Phase III Hallmark QUAD: ASV+DCV+Peg+Rib (Nulls/Partials) asunaprevir+BMS NS5A
Phase III Hallmark QUAD: ASV+DCV+Peg+Rib (Nulls/Partials)
This study is currently recruiting participants.
Verified July 2012 by Bristol-Myers Squibb
First Received on April 5, 2012. Last Updated on July 26, 2012
Phase III Hallmark DUAL: ASV+DCV (Nulls/Partials, Intolerants/Ineligibles. Naives)
Phase III Hallmark DUAL: ASV+DCV (Nulls/Partials, Intolerants/Ineligibles. Naives)
This study is currently recruiting participants.
Verified July 2012 by Bristol-Myers Squibb
First Received on April 18, 2012. Last Updated on July 23, 2012
EASL /AASLD Key Studies: interferon-free + studies for key new oral HCV drugs:
ASL: Potent Viral Suppression With the All-Oral Combination of Daclatasvir (NS5A Inhibitor) and GS-7977 (Nucleotide NS5B Inhibitor), +/- Ribavirin, in Treatment-Naive Patients With Chronic HCV GT1, 2, or 3 (100% SVR gt1, 91% gt2) - (04/19/12)
EASL: TMC435 with peginterferon and ribavirin in treatment-experienced HCV genotype 1 patients: the ASPIRE study, a randomised Phase IIb trial - (04/19/12)
EASL: TMC435 in patients infected with HCV genotype 1 who have failed previous pegylated interferon / ribavirin treatment: Virologic analyses of the ASPIRE trial - (04/19/12)
EASL: Once Daily GS-7977 Plus Ribavirin in HCV Genotypes 1-3: The ELECTRON Trial - (04/21/12)
EASL: GS-7977 + PEG/RBV in HCV Genotype 1: The ATOMIC Trial An End To Response-Guided Therapy - (04/20/12)
EASL: GS-7977 400 mg QD Safety and Tolerability in the Over 500 Patients Treated for at Least 12 Weeks - (04/25/12)
AASLD: Treatment with the 2nd generation HCV protease inhibitor BI 201335 results in high and consistent SVR rates - results from SILEN-C1 in treatment-naïve patients across different baseline factors - (11/08/11)
AASLD: SILEN-C3: treatment for 12 or 24 weeks with BI 201335 combined with peginterferon alfa-2a and ribavirin in treatment-naïve patients with chronic genotype-1 HCV infection - (11/07/11)
EASL: Peginterferon Lambda-1A (Lambda) Compared With Peginterferon Alfa-2A (Alfa) in Treatment- Naive Patients With HCV Genotypes 2 or 3: First SVR24 Results From EMERGE Phase IIb - (04/20/12)
Once-daily NS5A Inhibitor (BMS-790052) Plus Peginterferon-alpha-2a ... Apr 17, 2010 ... Once-daily NS5A Inhibitor (BMS-790052) Plus Peginterferon-alpha-2a And Ribavirin Produces High Rates Of Extended Rapid Virologic Response In ... www.natap.org/2010/EASL/EASL_32.htm
BMS-790052 is a First-in-class Potent Hepatitis C Virus (HCV) NS5A ...
BMS-790052 is a First-in-class Potent Hepatitis C Virus (HCV) NS5A Inhibitor for Patients with Chronic HCV Infection: Results from a Proof-of-concept Study -
EASL: A 12-Week Interferon-Free Regimen of ABT-450/r + ABT-333 + Ribavirin Achieved SVR12 in More Than 90% of Treatment-Naïve HCV Genotype-1-Infected Subjects and 47% of Previous Non-Responders - (04/23/12)
EASL: A 12-Week Interferon-Free Regimen of ABT-450/r, ABT-072, and Ribavirin was Well Tolerated and Achieved Sustained Virologic Response in 91% Treatment-Naïve HCV IL28B-CC Genotype-1-Infected Subjects - (04/19/12)
EASL: Dual Oral Therapy with NS5A Inhibitor Daclatasvir (BMS-790052) and NS3 Protease Inhibitor Asunaprevir (BMS-650032) in HCV Genotype 1b-Infected Null Responders or Patients Ineligible/Intolerant to Peginterferon/Ribavirin - (04/19/12)
EASL: SVR4 and SVR12 with an interferon-free regimen of BI 201335 AND BI 207127, +/- ribavirin, in treatment-naïve patients with chronic genotype-1 HCV infection: Interim results of SOUND-C2 - (04/22/12)
EASL: The efficacy and safety of the interferon-free combination of BI 201335 and BI 207127 in genotype 1 HCV patients with cirrhosis: Interim analysis from SOUND-C2 - (04/20/12)
Quadruple Therapy With BMS-790052, BMS-650032 and Peg-IFN/RBV for 24 Weeks Results in 100% SVR12 in HCV Genotype 1 Null Responders: original slide presentation at EASL April 2011 Proof of Concept that SVR is Achievable Without Peg/RBV - 4/11 null responder patients achieved SVR w/o Peg/Rbv, with only BMS-790052 (NS5A inhibitor) + BMS-650032 (protease inh) - (02/03/12)
EASL: Gilead: Interim Sustained Virologic Response Rates in Treatment-Naïve HCV Genotype 1a and 1b Patients Treated for 12 or 24 Weeks with an Interferon-Free All-Oral Quad Regimen - (04/21/12)
EASL: RAPID AND SUSTAINED ACHIEVEMENT OF UNDETECTABLE HCV RNA DURING TREATMENT WITH RITONAVIR-BOOSTED DANOPREVIR/PEG-IFNα-2A/RBV IN HCV GENOTYPE 1 OR 4 PATIENTS: DAUPHINE WEEK 36 INTERIM ANALYSIS - (04/22/12)
------------------------------------
Key Recent Developments and other EASL Studies of note:
EASL: In Vitro Resistance Analysis of Merck's HCV NS5a Inhibitor MK-8742 Demonstrates Increased Potency AgainstClinical Resistance Variants and Improved Resistance Profile - (04/23/12)
EASL: Safety and Antiviral Activity of ABT-267, a Novel NS5A Inhibitor, During 3-Day Monotherapy: First Study in HCV Genotype-1 (GT1)-Infected Treatment-Naïve Subjects - (04/25/12)
EASL: Antiviral Activity and Resistance Profile of the Novel HCV NS5A Inhibitor GS-5885 - (04/25/12)
EASL: A Phase 2a Study of BMS-791325, an NS5B Polymerase Inhibitor, With Peginterferon Alfa-2a and Ribavirin in Treatment-Naive Patients With Genotype 1 Chronic Hepatitis C Virus Infection - (04/25/12)
SVR24 AMONG G1/4 TREATMENT-NAIVE PATIENTS RECEIVING MERICITABINE IN COMBINATION WITH PEG-IFNα-2A/RBV: FINAL ANALYSIS FROM THE JUMP-C STUDY - (04/24/12)
INTERFERON-FREE TREATMENT WITH A COMBINATION OF MERICITABINE AND DANOPREVIR/R WITH OR WITHOUT RIBAVIRIN IN TREATMENT-NAIVE HCV GENOTYPE 1-INFECTED PATIENTS - (04/24/12)
GS-9669, A Novel NS5B Non-Nucleoside Thumb Site II Inhibitor, Demonstrates Potent Antiviral Activity, Favorable Safety Profile and Potential for Once-Daily - (04/24/12)
A Phase 2a Study of BMS-791325, an NS5B Polymerase Inhibitor ...
www.natap.org/2012/EASL/EASL_60.htm
A Phase 2a Study of BMS-791325, an NS5B Polymerase Inhibitor, With Peginterferon Alfa-2a and Ribavirin in Treatment-Naive Patients With Genotype 1
Patients of all IL28B Genotypes have High SVR Rates when Treated with VX-222 in Combination with Telaprevir/Peginterferon/Ribavirin in the ZENITH Study - (04/24/12)
VX-222 Vertex HCV polymerase nonnucleoside inhibitors (NNIs)Polymerase Inhibitor 3 Days Monotherapy
www.natap.org/2010/EASL/EASL_02.htm
VX-222 Vertex HCV polymerase nonnucleoside inhibitors (NNIs)Polymerase Inhibitor 3 Days Monotherapy. Reported by Jules Levin EASL Apr 14-18 2010. Vienna Austria.
Vertex QUAD Therapy Yielded 83-93% SVR with 12 weeks duration ...
www.natap.org/2012/APASL/APASL_11.htm
VX-222 is a selective, noncompetitive inhibitor of the hepatitis C virus (HCV) NS5B polymerase.1 Telaprevir (TVR)
Achillion Announces Positive SVR4 Results From Phase 2 Study of Sovaprevir (Formerly ACH-1625) and Advancement of ACH-3102 - (08/09/12)
BMS Suspends Study of Nucleotide BMS094 Formerly INX189 - (08/02/12)
Vertex Nucleotide Analysis - (08/01/12)
Vertex Announces Positive Results from Viral Kinetic Study of the Nucleotide Analogue ALS-2200 in People with Hepatitis C - (07/30/12)
Gilead Begins Single Pill Hepatitis C Study for 2014 Approval - (07/27/12)
Open-Labeled Study of PSI-7977 and RBV With and Without PEG-IFN in Treatment-Naïve Patients With HCV GT2 or GT3, and GT1 - (07/13/12)
A phase 1, randomized, placebo-controlled, 3-day, dose-ranging study of GS-5885, an NS5A inhibitor, in patients with genotype 1 hepatitis C - (06/20/12)
Medivir announces an interferon-free phase II combination trial with TMC435 and daclatasvir to commence shortly - (07/10/12)
Open-Label Study of GS-7977 + Ribavirin Pre-Transplant - (06/22/12)
Presidio Pharmaceuticals Successfully Completes Phase 1 Proof-of-Concept for PPI-668, its Potent HCV NS5A Inhibitor, in Hepatitis C Patients with Genotype-1 Infection - press release - (06/26/12)
Idenix Announces Positive Clinical Data for HCV Drug Candidates IDX184 and IDX719(NS5A) - (06/20/12)
Antiviral activity of TMC435 monotherapy in patients infected with HCV genotypes 2-6: TMC435-C202, a phase IIa, open-label study - (05/23/12)
EASL: Use of Telaprevir Plus Peg Interferon/Ribavirin for Null Responders Post OLT With Advanced Fibrosis/Cholestatic Hepatitis C - (05/04/12)
EASL: Ribavirin Dose Modification in Treatment-naïve and Previously Treated Patients Who Received Telaprevir Combination Treatment: No Impact on Sustained Virologic Response in Phase 3 Studies - (05/04/12)
EASL: NOVEL NS5A INHIBITOR ACH-2928 PHASE 1 RESULTS IN HEALTHY VOLUNTEERS AND HCV GT-1 PATIENTS - (05/02/12)
EASL: CUPIC: French Early Access Program - Compassionate Use of Protease Inhibitors in Viral C Cirrhosis - (04/26/12)
EASL/2011: Genotypic and Phenotypic Characterization of NS3 Variants Selected in HCV-Infected Patients Treated with ABT-450 - (04/05/11)
EASL: Alisporivir plus ribavirin is highly effective as interferon-free or interferon-add-on regimen in previously untreated HCV-G2 or G3 patients: SVR12 results from VITAL-1 Phase 2b study - (04/22/12)
EASL: Alisporivir (ALV) plus Peg-interferon/Ribavirin (PR) in HCV G1 Treatment-experienced Patients Achieves Primary Endpoint with Superior Efficacy at Treatment Week 12 Compared to Retreatment with PR - (04/22/12)
GSK2336805 HCV NS5A Inhibitor Demonstrates Potent Antiviral Activity in Chronic Hepatitis C (CHC) Genotype 1 Infection: Results from a First Time in Human (FTIH) Single and Repeat Dose Study - (11/09/11)
EASL: Presidio Pharmaceuticals Announces Phase 1a-1b Clinical Results with PPI-668, a Potent Pan-genotypic HCV NS5A Inhibitor - (04/25/12)
Safety and Efficacy of Vaniprevir (MK-7009) in Combination with Peg-interferon a-2a (Peg-IFN)/Ribavirin (RBV) in Genotype 1 Treatment-Experienced HCV-Infected Japanese Patients - (11/16/11)
MK-5172, a Second Generation HCV NS3/4A Protease Inhibitor is Active Against Common Resistance Associated Variants (RAVs) and Exhibits Cross-Genotype Activity - (11/07/11)
Safety and Antiviral Activity of MK-5172, a Next Generation HCV NS3/4a Protease Inhibitor with a Broad HCV Genotypic Activity Spectrum and Potent Activity Against Known Resistance Mutants, in Genotype 1 and 3 HCV-Infected Patients - (11/07/11)
ANA-598 HCV Non-Nuke Phase 2b Study Preliminary Data Results Background to Phase IIb Study - (10/14/11)
New Data Reported on INX-189, HCV Nucleotide
www.natap.org/2011/HCV/112911_01.htm
Nov 29, 2011 - top-line safety and antiviral data from its ongoing clinical trial designed to evaluate additional doses of INX-189, an oral nucleotide polymerase
BI 207127 is a potent HCV RNA polymerase inhibitor during 5 days monotherapy in patients with chronic hepatitis C - (11/04/09)
Safety, pharmacokinetics and antiviral effect of BI 207127, a novel HCV RNA polymerase inhibitor, after 5 days' oral treatment in patients with chronic hepatitis C - (04/27/09)
Pharmacokinetics of a Polymerase Inhibitor, ABT-333, in Treatment-naïve HCV Genotype 1-Infected Subjects Following Treatment with 2 days of ABT-333 Followed by 26 Days of ABT-333 Plus Pegylated Interferon and Ribavirin (12/08/09)
Safety, Tolerability, Pharmacokinetics and Antiviral Activity of the HCV Polymerase Inhibitor ABT-072 Following Single and Multiple Dosing in Healthy Adult Volunteers and Two Days of Dosing in Treatment-Naïve HCV Genotype 1-Infected Subjects (12/08/09)
|
|
| |
| |
|
|
|