 |
 |
 |
| |
Antiviral Characteristics Of S/GSK1265744, An HIV Integrase Inhibitor (INI) Dosed By Oral Or Long-acting Parenteral Injection....once monthly or 3 months dosing/PrEP/HAART
|
| |
| |
Reported by Jules Levin
52nd ICAAC Sept 9-12 2012 San Francisco
T. Yoshinaga, PhD - Section Head1, M. Kobayashi, MS - Section Head 1, T. Seki, PhD - Team Leader 1, T. Kawasuji, MS - Section Head 1, T. Taishi, MS - Team Leader 1, A. Sato, PhD - Department Head 1, T. Fujiwara, PhD - Project Leader 1, B. Johns, PhD - Medicinal Chemist 2, R. Hazen, PhD - Virology 2, R. Ferris, PhD - Virology 2, M. Underwood, PhD - Virology 2;
1Shionogi, Toyonaka, Japan, 2GlaxoSmithKline, RTP, NC.
ABSTRACT
Background: S/GSK1265744 (S/GSK744) is a potent and selective HIV INI with improved in vitro resistance profile over 1st generation INIs. S/GSK744 has exceptional pharmacokinetic (PK) properties in early human oral delivery trials and robust antiviral activity with a -2.5 log10 copies/mL decrease at day11 during 30mg QD monotherapy. Additional S/GSK744 attributes (low solubility, low clearance) support parenteral dosing at ≤ once monthly. Infrequent dosing may enable improved adherence and efficacy for both HIV therapy and pre-exposure prophylaxis (PrEP). We report here the virological characteristics of 744 including results of in vitro passage and cross-resistance panel studies.
Methods: Antiviral potency of S/GSK744 in cell culture and the effect of human serum proteins were measured by MTT assay. HIV-1 IIIB was passaged in MT-2 cells under increasing S/GSK744 concentrations and the IN gene was sequenced. Fold changes (FC) of mutants during passages and site directed molecular clones (SDMs) were calculated as a ratio of IC50 against WT with a HeLa-CD4 assay.
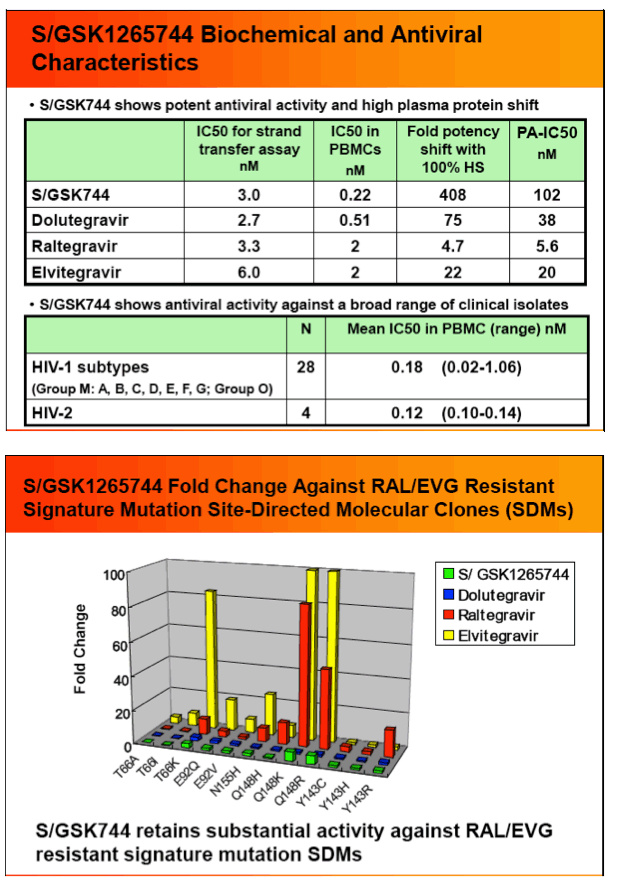
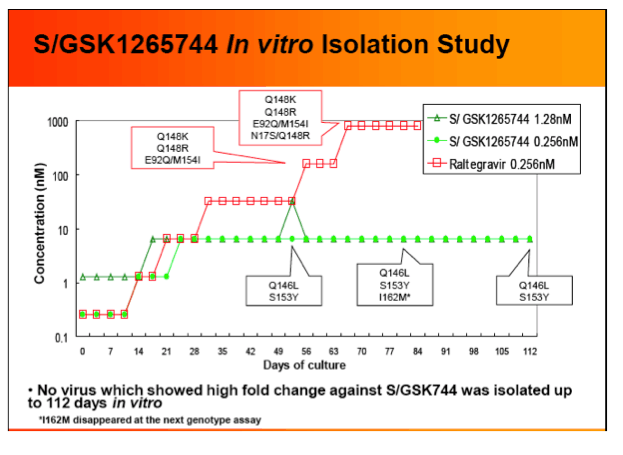
Results: S/GSK744 IC50's in human peripheral blood mononuclear cells (PBMC) ranged from 0.22 to 0.34 nM. In vitro studies provided a protein adjusted IC50 estimate of 102 nM. Exposure of MT-2 cells infected with HIV-1 IIIB to S/GSK744 for up to 112 days did not produce any highly resistant mutants, with a maximum 8.4 fold resistance. Eight SDM's containing single RAL mutaionts were < 2 FC, except for Q148K/R (5.6/5.1 FC, respectively). S/GSK744 FCs of 14 double mutants among 15 SDMs were < 12.
Conclusions: S/GSK1265744 showed limited cross-resistance to both RAL and EVG, and potential for a high barrier to resistance. Given these in vitro data and demonstrated therapeutic levels with LAP dosing, S/GSK744 may have a favorable profile for both HIV treatment and PrEP. Additional studies are underway to further characterize its potential.

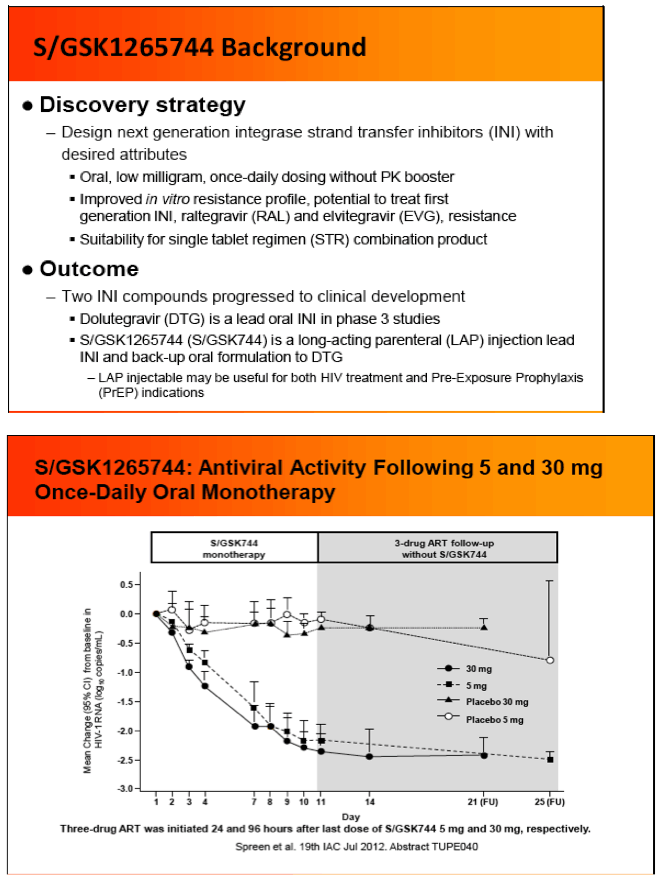
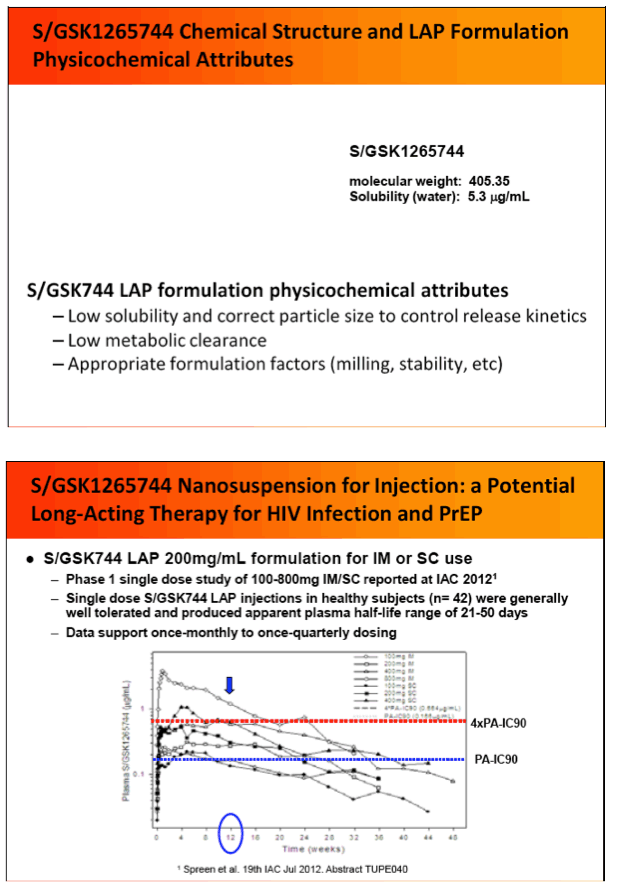
IAC: Pharmacokinetics, Safety and Tolerability of the HIV Integrase Inhibitor S/GSK1265744 Long Acting Parenteral Nanosuspension Following Single Dose Administration to Healthy Adults - (07/25/12)
|
| |
|
 |
 |
|
|