 |
 |
 |
| |
Pharmacokinetics of a Novel EVG/COBI/FTC/GS-7340 Single Tablet Regimen
|
| |
| |
Reported by Jules Levin
13th HIV Clinical Pharmacology Workshop Apr 16-18 2012 Barcelona Spain
S Ramanathan, X Wei, J Custodio, H Wang, A Dave, A Cheng, and B Kearney
Gilead Sciences, Inc., Foster City, CA, USA

Below Excerpted from:
Clinical Pharmacology at the 13th Workshop on Clinical Pharmacology of HIV Therapy - Courtney V. Fletcher, Pharm.D. - (05/14/12)
Cobicistat Increases TFV Concentrations when given in a single tablet formulation of GS-7340, cobicistat, elvitegravir and emtricitabine.
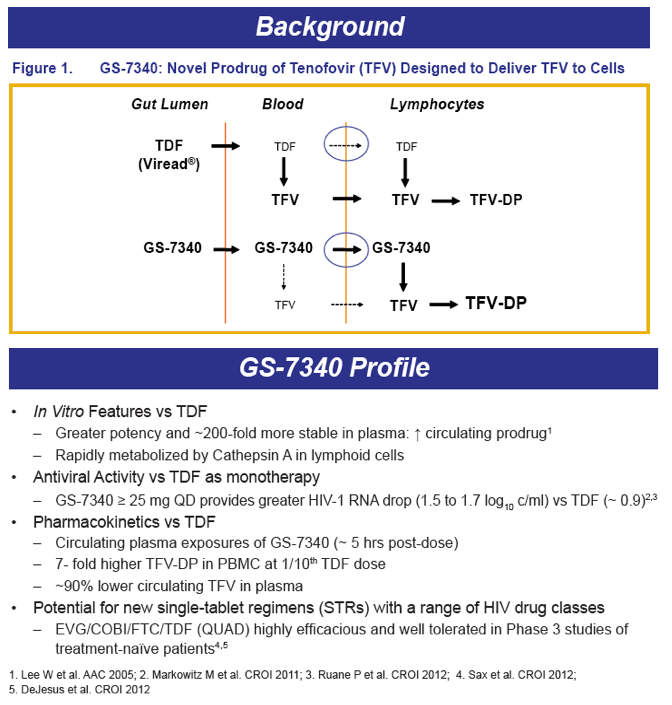
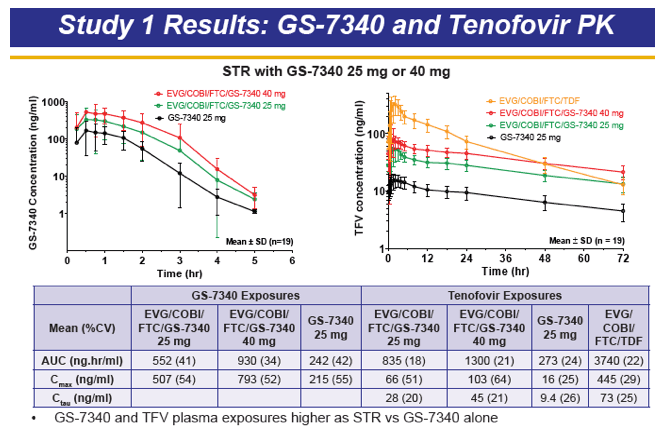
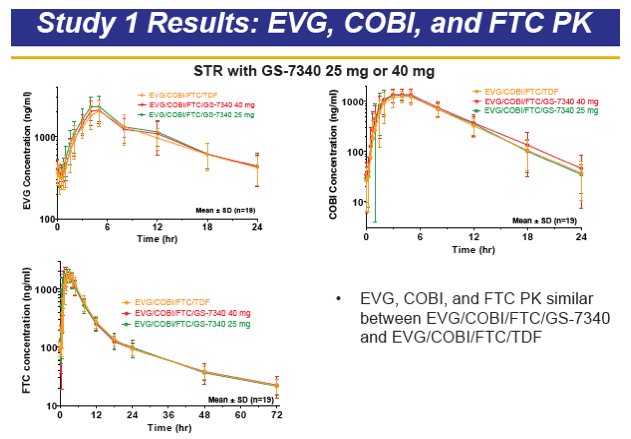
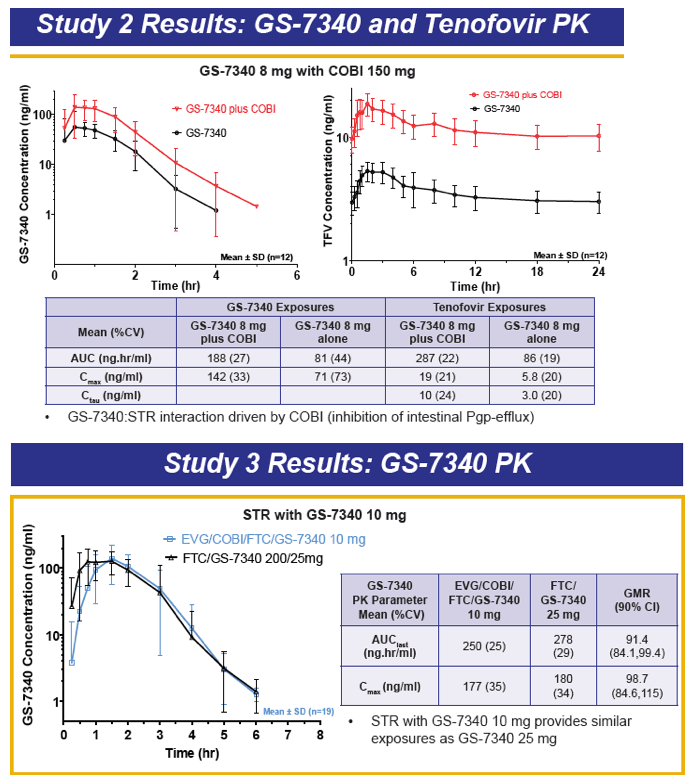
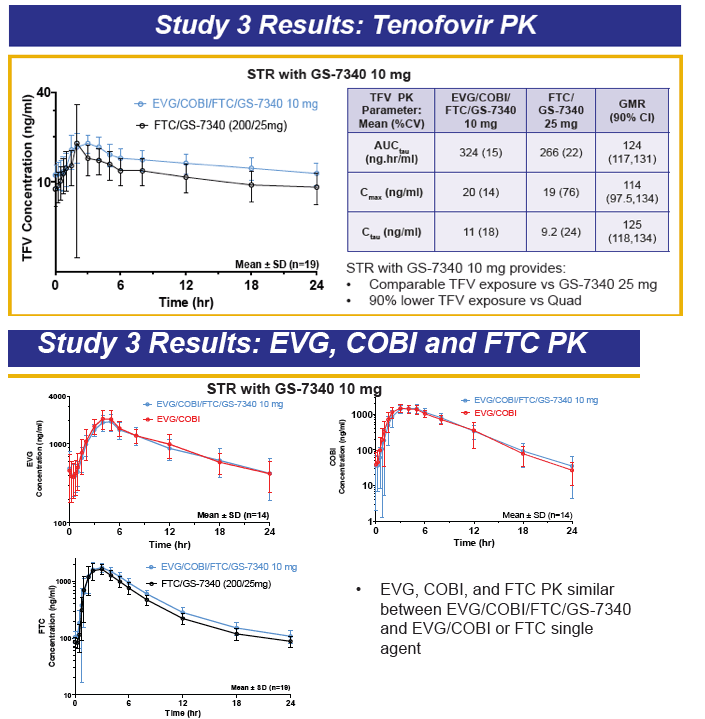
GS-7340 is a prodrug of TFV that achieves higher intracellular concentrations from lower plasma concentrations, and therefore is more potent than TDF. For example, the IC50 for TFV is 1.2 μM, 0.015 μM for TDF, and is 0.003 μM for GS-7340. The PK of TFV following administration of the TFV prodrug GS-7340 in a single tablet formulation of GS-7340, COBI, EVG and FTC, were evaluated in a series of healthy volunteers studies (abstract O_13 and see also, http://www.natap.org/2012/pharm/Pharm_13.htm ). When GS-7340 was given in combination with COBI only, compared with GS-7340 alone, the AUC, Cmax and Cmin of TFV were all increased 3-fold, providing data that indicate an inhibitory interaction between GS-7340 and COBI. This interaction appears to be the result of COBI inhibiting the P-gp mediated intestinal efflux of GS-7340, which results in increased bioavailability of GS-7340 and therefore increased plasma concentrations of TFV. The single tablet formulation with a reduced dose of GS-7340 to 10 mg, with COBI (150 mg), EVG (150 mg) and FTC (200 mg) achieved concentrations of GS-7340 equivalent to administration of GS-7340 at a dose of 25 mg given without COBI (AUC ratio of +COBI to -COBI = 0.91) and achieved TFV plasma concentrations that were slightly higher (AUC ratio of +COBI to -COBI of 1.24). Because administration of TFV as the GS-7340 prodrug achieved higher intracellular concentrations of TFV-diphosphate (the pharmacologically-active moiety) than an equivalent dose of TDF, plasma TFV concentrations are actually lower (approximately 90%) with GS-7340 than with TDF.
I think GS-7340 is a very intriguing compound. The enhanced lymphatic delivery and intracellular concentrations of active drug (TFV-diphosphate) may represent an important next step in HIV therapeutics: improving the delivery of drug to where you want it to enhance efficacy while reducing concentrations of drug where you don't want it to improve safety. The proof of this concept of course requires comparative data on safety and efficacy. A Phase II study comparing EVG/COBI/FTC/GS-7340 with EVG/COBI/FTC/TDF (QUAD) is now underway.
|
| |
|
 |
 |
|
|