 |
 |
 |
| |
Lack of Pharmacokinetic Interaction Between HCV Protease Inhibitor MK-5172 and HCV NS5A Inhibitor Daclatasvir in Healthy Volunteers
|
| |
| |
Reported by Jules Levin
AASLD 2013 Nov 1-4 Wash DC
Wendy W. Yeh,1 Iain P. Fraser,1 Marc Bifano,2 Luzelena Caro,1 Jennifer Talaty,1 Zifang Guo,1 Jennifer McCarthy,1Stephen P. Youngberg,3 Joan R. Butterton1
1Merck & Co., Inc, Whitehouse Station, NJ; 2Bristol-Myers Squibb Research and Development, Hopewell, NJ, USA; 3Celerion, Lincoln, NE, USA

ABSTRACT
Background: MK-5172, a potent, once-daily competitive inhibitor of the hepatitis C virus (HCV) NS3/4A protease with improved potency compared with the approved first generation protease inhibitors, and daclatasvir (DCV), an HCV NS5A replication complex inhibitor with pan-genotypic activity in vitro, could be components of an all-oral combination regimen for the treatment of chronic HCV infection. The aim of the present study was to evaluate potential pharmacokinetic interactions as well as safety and tolerability of MK-5172 and daclatasvir co-administration in healthy subjects.
Methods: This was a single-center, open-label, fixed-sequence, multiple-dose study in 14 healthy adult male and female volunteers, ages 21-49 years. Since MK-5172 in HCV-infected patients demonstrates ~2-fold higher exposure compared with healthy subjects, a 200 mg dose of MK-5172 in healthy subjects was used in this study to match the exposure of a 100 mg dose (the intended clinical dose) in HCV-infected patients. In Period 1, subjects received oral doses of 60 mg daclatasvir once daily on Days 1 to 7. Following a 4-day washout, subjects received oral doses of 200 mg MK-5172 once daily on Days 1 to 7 in Period 2. In Period 3, which commenced immediately after Period 2, subjects were co-administered once daily oral doses of 200 mg MK-5172 and 60 mg daclatasvir on Days 1 to 8. Plasma pharmacokinetic samples were obtained for daclatasvir on Day 7 in Period 1 and Day 8 in Period 3, as well as for MK-5172 on Day 7 in Period 2 and Day 8 in Period 3. Safety assessments included electrocardiograms, vital signs, clinical laboratory tests, physical examination, and adverse event monitoring.
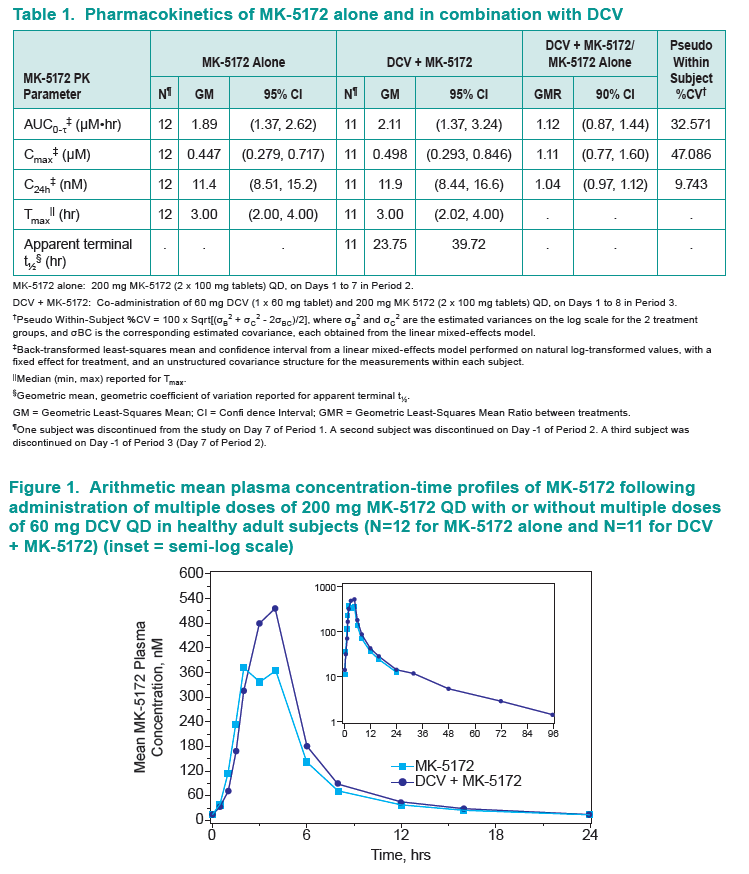
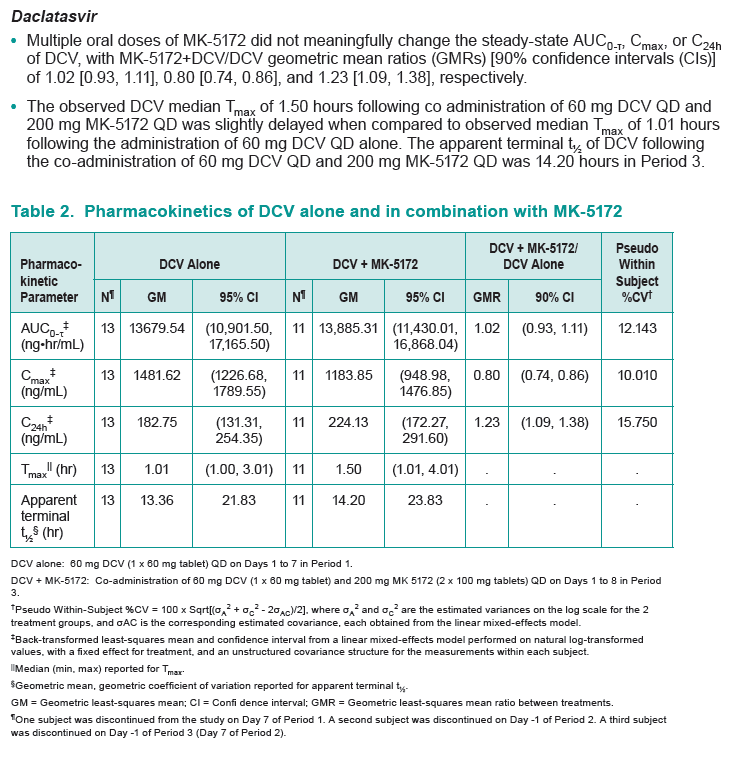
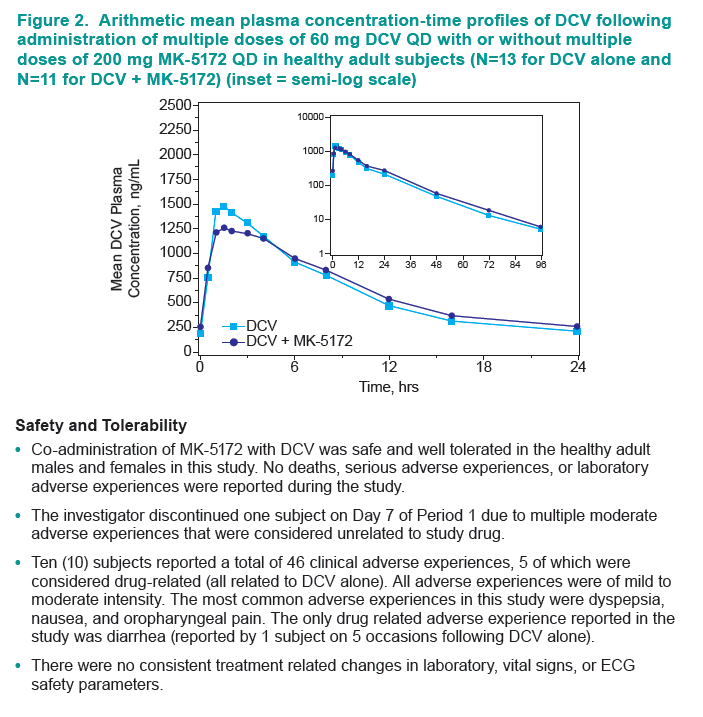
Results: Co-administration of MK-5172 with daclatasvir was safe and well tolerated. Multiple oral doses of MK-5172 did not meaningfully change the steady-state AUC0-τ, Cmax, or C24h of daclatasvir, with MK-5172+DCV/DCV geometric mean ratios (GMRs) [90% confidence intervals (CIs)] of 1.02 [0.93, 1.11], 0.80 [0.74, 0.86], and 1.23 [1.09, 1.38], respectively.
Multiple oral doses of daclatasvir did not meaningfully change the steady-state AUC0-τ, Cmax, or C24h of MK-5172, with MK-5172+DCV/MK-5172 GMRs [90% CIs] of 1.12 [0.87, 1.44], 1.11 [0.77, 1.60], and 1.04 [0.97, 1.12], respectively.
Conclusions: Co-administration of MK-5172 and daclatasvir in healthy volunteers did not result in clinically significant drug-drug interactions. MK-5172 and DCV were safe and well tolerated when coadministered. These results suggest that no dose adjustments of MK-5172 or daclatasvir are needed for co-administration of these drugs without other direct acting antiviral agents in interferon-free, combination regimens in HCV-infected patients.


|
| |
|
 |
 |
|
|