 |
 |
 |
| |
Follow-up of HIV-Associated Neurocognitive Disorders in the cART Era: the Neuradapt Study
|
| |
| |
Reported by Jules Levin
CROI 2013
Matteo Vassallo*1, Brigitte Dunais2, Jacques Durant2, Virginie Biscay2, Muriel Laffon3, Christine Lebrun-Frenay3, Jacqueline Cottalorda2, Michel Ticchioni2, Christian Pradier2, Pierre Dellamonica2 and Neuradapt Study Group
1- Pierre Nouveau Hospital, Cannes France 2-L'Archet Hospital, Ctr Hosp Univ Nice, France; 3- Pasteur Hospital, Ctr Hosp Univ Nice, France

1- During a 2 year follow-up period, the proportion of patients with a diagnosis of HAND dramatically increased, most of them displaying mild cognitive disorders, whether symptomatic or not. The main novel domains of impairment concerned executive functions, learning and working memory.
2- We showed that patients with clinical worsening had lower CPEs at inclusion and at the end of follow-up. Even in the subgroup of patients who did not change treatment during the follow-up, differences were significant.
3- The majority of novel HAND diagnosis concerned patients with ND, while in those with completely normal tests upon inclusion, NP performance remained generally stable.
4- If confirmed by other studies on larger numbers of patients, our results suggest that the majority of neurocognitive disorders remains stable over time, while
novel cognitive deterioration could be prevented by high CNS penetrating regimens.
5- Further insights into the protecting role of high CNS penetrating drugs could be provided by the antiretroviral monocyte efficacy score, which was recently found by Shikuma et al. [12] to be more accurately linked to NP testing results than the CPE score
ABSTRACT
Background: The course of HIV-associated neurocognitive disorders (HAND) is poorly understood.
We assessed changes over time in neuropsychological test results (NPr) and their risk factors among a regularly followed HIV-infected patient population.
Methods: Patients followed in Nice were randomly selected to undergo NP testing. Test score was adjusted for age, gender and education. Patients were divided into 5 groups: Normal tests (NT), neuropsychological deficit (ND, 1 impaired cognitive domain), asymptomatic neurocognitive disorders (ANI), mild neurocognitive disorders (MND) and HIV- associated dementia (HAD). Patients were followed over time for changes in NPr and associated risk factors.
Initial parameters included CD4 and CD8 count, HIV-RNA viral load (VL), DNA-proviral load, hepatitis C co-infection (HCV), immune activation markers and Drug Concentration-Penetration Effectiveness score (CPEs) 2010.
During follow-up (FU), CD4, CD8, VL, change of cART and CPEs were recorded. Changes in impairment category over time according to NPr were investigated.
Means were compared using Student's t-test and ANOVA, and independent risk factors identified by multivariate analysis.
Results: From December 2007 to July 2009, 259 patients underwent NP tests and 96 accepted to continue FU from 2009 to 2011 (75% male, 80% on cART, mean age 46 yrs, 59% with VL <50 cp/ml, median CD4 487/ml, 24% HCV). The two groups were comparable.
Mean FU period was 22.4 months (95%CI: 21.5-23.3).
Upon NP retesting, 6 patients improved, 31 worsened and 59 were stable. The proportion of patients with HAND rose from 26% (13% ANI, 10% MND, 3% HAD) to 45% (26% ANI, 16% MND, 3% HAD) of the study population. Most patients initially diagnosed with HAND remained stable, 5/25 showed clinical improvement and 3/25 deteriorated. Of 33 patients with NT, 4 deteriorated, while 24/38 with initial ND had poorer test results, and contributed most of the new HAND cases.
Patients with clinical deterioration had a lower CPEs both at inclusion (6.9 vs 8.1, p=0.005) and at the end of FU (7.2 vs 7.8, p=0.07) than those with improved or stable performance. This was confirmed by multivariate analysis.
Conclusions: The proportion of patients with HAND increased during follow-up and those with ND were at higher risk than patients with NT.
Patients with higher CPEs upon inclusion and at the end of FU were at lower risk, suggesting that cART with better CSF penetration could protect against cognitive deterioration.
Background
The prevalence of HIV-associated neurocognitive disorders (HAND) remains (15-50%) in the era of combination antiretroviral treatment (cART). [1-4]
New insights in the treatment of HAND have been provided by the concentration penetration effectiveness (CPE) score, which ranked antiretrovirals according to their capability to penetrate into the CNS. [5]
Indeed, brain penetration of antiretrovirals seems crucial to achieve the goal of maximal suppression of HIV, but it is difficult to say how well CSF drug concentrations reflect the parenchymal diffusion of molecules. [1]
Moreover, dynamics of cognitive changes over time have been essentially studied on patients initiating or modifying treatment and results are contradictory in terms of clinical benefits of choosing drug combinations with high central nervous system (CNS) penetration. [6-9]
Indeed, Cysique et al. showed that high CNS penetrating drugs are linked to greater improvement in neuropsychological (NP) performance [6], while Marra et al. showed clinical worsening when choosing high penetrating regimens. [7]
An ongoing, unanswered question is whether antiretrovirals with high CNS penetration protect against cognitive deterioration and at present there is no evidence to support the initiation of cART with better CNS-penetrating molecules for preventing HAND. [1-10]
Our aim was to follow changes over time in NP performances in a population of randomly selected HIV-infected patients and to identify factors linked to clinical deterioration. In particular, we focused on the CPE score and its potential impact on clinical outcome.
Methods
Study design and participants
We first randomly selected HIV-1 infected patients above18 years of age among subjects followed in the Department of Infectious Diseases at Nice University Hospital. No limits were set concerning CD4 cell count or HIV viral load.
Exclusion criteria included active opportunistic infection, a change in psychotropic therapy within the last 3 weeks or any neurological history.
Patients were asked to provide informed consent. The study was approved by the Montpellier Ethics Committee.
Each patient performed a wide range of neuropsychological (NP) tests administered by a single trained neuropsychologist.
Tests explored a wide spectrum of cognitive domains: learning and recall episodic memory, attention/concentration, working memory, executive functions, language, visual agnosia and motor/psychomotor speed.
The NP scores from each test were transformed into z-scores as described elsewhere [11] and were adjusted for age, gender and years of education, using standardised norms. The mean duration of tests was 90 minutes per patient.
According to the AAN revised criteria [11], the definition of HAND includes the 3 following categories:
1) ANI, involving at least two cognitive domains and documented by a performance of at least 1 SD below the mean on NP tests, without interference in everyday functioning. The asymptomatic characteristics of impairment were defined by the InstrumentaI Activity of Daily Living short version battery and by interviewing the patient and his/her family.
2) MND, involving at least two cognitive domains and documented by a performance of at least 1 SD below the mean on NP tests, with mild interference in daily functioning.
3) HAD, involving at least two cognitive domains and documented by performing below 2 SD from the normative mean on NP tests, with marked interference in daily functioning.
According to their NP test results, patients were classified into two main groups:
1) Those with HAND (i.e. ANI, MCD or HAD)
2) Those without HAND. Patients included in this category could have either normal tests (NT), or one cognitive domain impaired, with 1 or 2 SD below the mean (which we defined as Neuropsychological Deficit, ND).
Demographic parameters and background measurements at inclusion
In order to correlate results to NP test group, the following parameters were recorded at inclusion for each patient: age, gender, education, co-morbid conditions (hypertension, smoking, dyslipidemia, illicit drug use, diabetes), use of psychotropic molecules (benzodiazepines, antidepressants, carbamates and anti-epileptic drugs), inclusion and nadir CD4 and CD8 T-cell count, HIV-RNA viral load (Abbott Real-time PCR), HIV-DNA viral load (Abbott Real-time
PCR), viral hepatitis markers and type, duration and 2010 CPE score [5] of current antiretroviral therapy upon inclusion.
Patient follow-up
Patients included in the Neuradapt study continued regular follow-up with clinical evaluation and blood tests. Those who also accepted to repeat NP evaluation were submitted to the same panel of tests as upon inclusion, approximately 2 years later, in order to identify any changes and continued to participate in the study.
The following parameters were considered during the follow-up: CD4 and CD8 T-cell count, HIV-RNA viral load, viral hepatitis markers, changes of cART and type of antiretroviral therapy and CPE score at the end of follow-up.
NP testing follow-up
For each patient, comparison between NP testing results at inclusion and at the end of follow-up allowed to identify three situations according to changes in AAN subgroups of impairment:
- Clinical improvement
- Clinical worsening
- Clinical stability
In the HAND group, changes over time were defined by changes in the sub-groups of impairment (i.e. ANI, MND and HAD).
Among the non-HAND group, changes from NT to one cognitive domain 2 SD below the mean were considered as worsening (or inversely as improvement), while in case of changes from NT to one cognitive domain 1 SD below the mean patients were considered as stable.
Statistical analysis
Patients with either improvement or stable NP tests were compared to those with clinical worsening.
Means were compared using Student's t-test and ANOVA, and independent risk factors were indentified by multivariate analysis. A p-value < 0.05 was considered significant.

From December 2007 to July 2009, 259 patients underwent NP tests, and 96 of them accepted to continue NP follow-up, from 2009 to 2011.
Among the 163 subjects who did not repeat NP evaluation, the main reason for refusal was lack of interest in the study. One patient died during the follow-up.
Participants and non-participants to follow-up were comparable with regard to age, gender, mean and nadir CD4 count, detectable viral load and HCV co-infection (data not shown).
Mean duration of follow-up was 22.4 months (95%CI: 21.5-23.3).
Among the 96 patients who participated, 25 (26%) were defined as suffering from HAND (13% ANI, 10% MND and 3% HAD) upon initial testing. Table 1 resumes characteristics of patients who participated to follow-up.
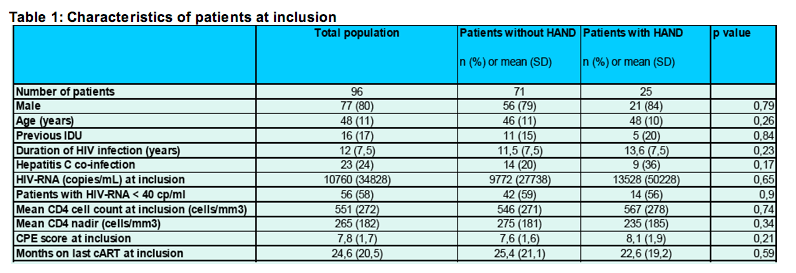
NP testing follow-up
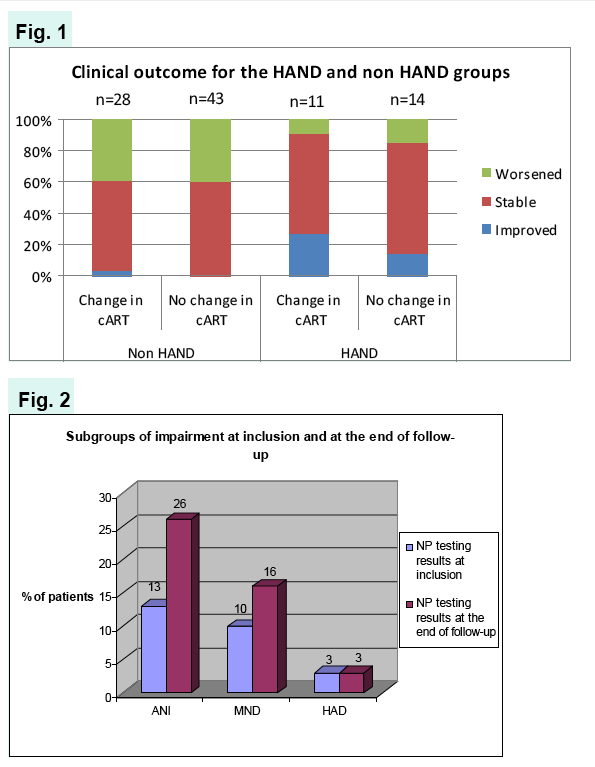
Upon NP retesting, 6 patients showed clinical improvement, 31 had worsened and 59 were stable, resulting in 43 patients presenting with HAND (45%).
The majority of HAND cases consisted in ANI (26%) and MND (16%), while the proportion of HAD remained stable at 3%.
The majority of patients newly diagnosed with HAND came from the subgroup of subjects with ND, who worsened in 24 of 38 cases, while only 3/25 patients with initial NT contributed new cases of HAND.
Main cognitive deterioration was found in executive functions, working and learning memory.
Figure 1 resumes clinical outcome for the HAND and the non HAND groups, while figure 2 describes subgroups of impairment according to the AAN definition
Factors linked to changes in NP performances
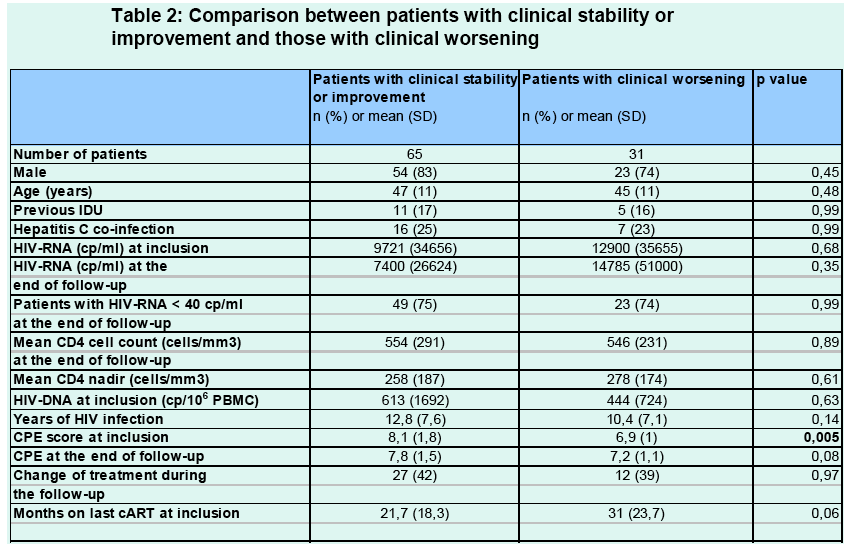
Comparison between patients with either improvement or stable NP tests and those with clinical worsening showed that lower CPEs at inclusion (6.9 vs 8.1, p = 0.005) and at the end of follow-up (7.2 vs 7.8, p =0.07) and higher duration of last cART (31 vs 22 months, p =0.06) were associated with clinical deterioration. (Table 2)
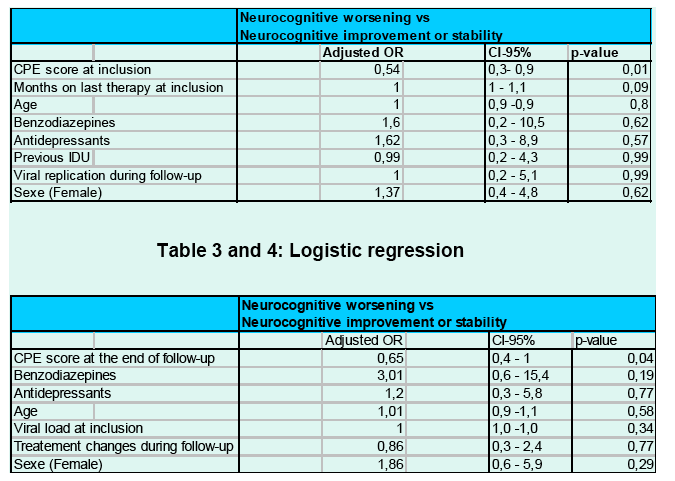
After adjustment for potential confounding factors, CPEs at inclusion and at the end of follow-up were still independent factors linked to NP performances. (Table 3 and 4)
Patients with stable treatment during follow-up
In order to exclude potential bias linked to changes on cART during the follow-up, we selectively looked at the 57 patients who did not modify treatment: even in this group, subjects with improvement or stability differed from those with worsening in terms of CPE score at inclusion (8.1 vs 7.1, p = 0.03), while they did not differ for the duration of the last antiretroviral treatment (25.8 vs 35.7 months, p = 0.15)
References
1. Mc Arthur JC, Steiner J et al. Human immunodeficiency virus associated neurocognitive disorders: mind the gap. Ann Neurol 2010; 67:699-714
2. Robertson KR, Smurzynski M et al. The prevalence and incidence of neurocognitive impairment in the HAART era. AIDS 2007;21:1915-21.
3. Woods SP, Moore DJ et al. Cognitive neuropsychology of HIV-associated neurocognitive disorders. Neuropsychol Rev 2009;19:152-168
4. Heaton RK, Clifford DB et al. HIV-associated neurocognitive disorders persist in the era of pot ent antiretroviral therapy: CHARTER Study. Neurology 2010;75:2087Đ2096.
5. Letendre S, Marquie-Beck J, Capparelli E et al. Validation of the CNS Penetration-Effectiveness rank for quantifying antiretroviral penetration into the central nervous system. Arch Neurol 2008; 65: 65-70
6. Marra CM, Zhao Y, Cliff ord DB et al. Impact of combination ant iretroviral therapy on cerebrospinal fluid HIV RNA and neurocognitive performance. AIDS 2009; 23:1359-66.
7. Cysique LA, Vaida F, Letendre S et al. Dynamics of cognitive change in impaired HIV-positive patients initiating antiretroviral therapies. Neurology 2009; 73:342-8.
8. Evers S, Rahmann A., Schwaag S et al. Prevention of AIDS dementia by HAART does not depend on cerebrospinal fluid penetrance. AIDS Res Hum Retroviruses 2004;20:483-91
|
| |
|
 |
 |
|
|