 |
 |
 |
| |
Aging with HIV: Where do we stand after CROI 2013?
|
| |
| |
David Alain Wohl, MD -
Associate Professor, Division of Infectious Diseases,
The University of North Carolina at Chapel Hill
Introduction
Getting old may be tough, but it beats the alternative. More and more I am reminding my HIV+ patients, and myself, of this truism as together we reach the milestones of middle age: knee braces, colonoscopies, and misplaced car keys. However, these men and women, having dodged the early reaping of HIV worry that both time and the virus are catching up with them.
The Centers for Disease Control (CDC) estimate that over half of those living with HIV in the US will be older than 50 years of age by 2017.1 San Francisco, hit earliest by HIV, has already seen the median age of the HIV-positive population push past 50.2 With the graying of the domestic epidemic patients and their healthcare providers are now focused on how HIV influences the aging process. To believe the lay press, growing older with HIV will not be pretty - with handfuls of pills, heart attacks, and broken bones, awaiting the golden years.3
The scientific literature feeds a similar story of "premature" or "accelerated" aging for those living with HIV. For example, a recent Italian paper published in Clinical Infectious Diseases comparing HIV-positive and -negative cohorts suggests that people living with HIV can expect to experience some conditions associated with aging a decade earlier than would be expected.4 Not surprisingly, patients are petrified - suffering a double helping of the age-phobia pervasive among baby-boomers.
However, the clinical data are confusing and there are challenges to trying to ascertain the contribution of HIV to aging, including the difficulty of identifying an appropriate control group with whom to compare HIV-infected individuals. The Italian study, for example, included HIV-infected patients from a single clinic while the HIV-negatives came from a large national electronic cohort. While the controls were matched to HIV-infected cases based on age, sex, and race there was no adjustment for traditional risk factors for cardiovascular and other age-related conditions, except for the presence of hypertension. The word 'smoking' is nowhere to be found in the article, despite rates of tobacco use among HIV-positive folks that are known to be double to triple those seen in the general population.
A more compelling picture of a virus-immune activation-organ disease connection is found in some of the more basic science work, where elevated levels of immune responses and markers of inflammation are seen to decline with HIV treatment but, in some patients, persist despite evident control of viremia. Such chronic activation of the immune system and low-level inflammation have been linked to cardiovascular disease (CVD) outside of HIV.
So, does HIV lead to a greater risk of age-related conditions? At CROI 2013, you had to work hard to avoid talk of statins, coronary calcium, soluble markers of inflammation, and other topics related to the effects of HIV and its therapies on our most beloved organs. This report reviews the major conference presentations that focused on this question and discusses how what we learned shapes where we are headed.
HIV and Excess Risk of CVD
CVD is common throughout the world and is a major cause of mortality, especially in developed nations. Are HIV+ individuals at increased risk for CVD? The short answer is, yes. And, why shouldn't they be? HIV+ persons heft a disproportionate burden of traditional risk factors for CVD relative to the general population. They smoke more and exercise less. Other factors such as diet, stress, and cocaine abuse - call it hard living - are harder to measure but also play a role. In addition, most of those living with HIV are patients, meaning they are in medical care, and thus subjected to more diagnostic scrutiny than most folks who see their doctor infrequently.
Undeniably, there are other factors that can pile-on risk of end-organ disease among those living with HIV, including, as mentioned above, chronically elevated levels of inflammation and/or immune activation and much of the research being conducted in this field aims to isolate the effects of HIV on aging organ systems, usually by adjusting for the known and measurable factors that ante-up risk.
A good example of this type of research was presented by investigators from the Veteran's Aging Cohort Study (VACS).5 These researchers have taken pains to make the distinction between accentuated risk of diseases associated with aging and premature aging. The former entails an increased risk of disease given the presence of HIV and the latter, the diagnosis of a disease at a younger than expected age. In an analysis of data collected from over 100,000 VACS participants, 31% of whom are HIV+, the team examined age of diagnosis of MI, end stage renal disease, and non-AIDS defining cancers to determine whether HIV was associated with earlier development of these conditions. HIV+ cases were matched to two HIV- controls based on age, race, and clinical site.
Among these veterans there was an increased risk for MI associated with HIV infection - about an 81% increased relative risk after adjustment for a number of relevant and major confounders. However, the age at which HIV-infected and -uninfected patients first were diagnosed with an MI was not different. The absence of a difference in age at the time of MI but a higher rate among HIV+ patients is explained by looking at the MI rates across the range of participant ages. For those younger than 50 years of age, HIV+ patients had similar rates of MI as the uninfected. Yet, at older ages, the rates of MI were slightly higher for those with HIV infection.
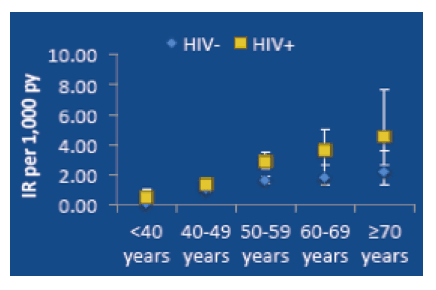
Similarly, in examining end stage renal disease the study found no evidence of premature incidence with HIV, but rates were higher in general among infected versus uninfected patients. Non-AIDS malignancies did appear to occur earlier among HIV+ patients (by only about 7 months) and they also had higher rates across all ages.
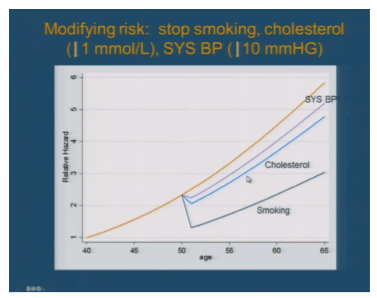
Another report looking at excess risk comes from the D:A:D study, a large European cohort of HIV+ patients (the study does not enroll HIV-uninfected controls).6 In this analysis, the effect of age on the development of major CVD events was examined by comparing actual data from the cohort with several equations designed to predict CVD risk (e.g., Framingham Risk Score). Basically, what this modeling showed was that as HIV+ men (they just looked at men) age beyond 40, their relative risk of CVD events tends to increase at a rate that is higher than expected based on the population derived risk equations. The increase, however, was modest and became most evident at around 60 years. Importantly, the effects of modification of risk was also modeled, revealing that smoking cessation and declines in blood pressure and total cholesterol erase much of this excess age-related risk. This is an interesting finding as it suggests that bread and butter risk modification is likely the most effective intervention for any 'excess' CVD risk accompanying HIV infection.
A group from Harvard conducted a different type of modeling study.7 These investigators attempted to estimate CVD risk for people living with HIV in the US compared to two other populations: a) the general US population and b) a group that is HIV negative but with risks for CVD that are similar to the HIV positive population (i.e., high rates of smoking). A variety of data sources were used to generate assumptions regarding CVD rates. Importantly, for the HIV+ population, the paper from Guaraldi et al (the Italian study published in Clinical Infectious Diseases in 2011) 4 was used to estimate CVD risk. As mentioned above, that case-control analysis found that the prevalence rates of poly-morbidity, including CVD, were higher among those with HIV and that infected persons experienced such diseases earlier than those who are uninfected. Therefore, the Harvard study assumes the Italian data (i.e., premature CVD with HIV) are reality and apply them to the US. What they found, of course, was that there was more CVD among HIV+ people especially as they age - a finding that is as consistent as it is obvious.
A bit more compelling evidence for disproportionate risk of CVD accompanying HIV comes from studies that quantify and compare cardiovascular pathology between HIV+ patients with HIV- controls.
Wendy Post from Hopkins presented data from the Multicenter Men's Cohort Study (MACS) Cardiovascular Study - a MACS substudy in which men underwent cardiac CT scans to detect calcified plaque as well as CT angiography to identify non-calcified and mixed plaques.8 HIV+ (n=571) and HIV- (n=302) MSM were enrolled. HIV+ men were younger, but were more often non-white, and had higher smoking rates (32% versus 21%), and lower cholesterol levels (including HDL cholesterol) than the HIV- men. It is notable that even in this study where only urban MSM are enrolled there are major differences in the characteristics of those who are and are not HIV-infected- demonstrating the challenge in finding a control group with whom to compare with HIV+ populations.
Most of the MACS HIV+ men (81%) had undetectable HIV RNA levels and high CD4 cell counts. Coronary calcium scores (which are a good predictor of CAD in the general population) were higher in HIV+ versus HIV- men but this effect was attenuated dramatically when adjustment was made for race, age, and a number of traditional CVD risk factors. This again highlights the contribution of non-HIV risks the CVD in infected individuals. However, by CT angiography the presence of any plaque and non-calcified plaque was significantly more likely to be seen in the HIV+ men, even with the adjustments, and the extent of these plaques was also greater. Interestingly, the prevalence non-calcified plaque (considered less stable and thus potentially more dangerous than those that are calcified) was not directly associated with increasing age in the HIV- men but was in the HIV+ men.
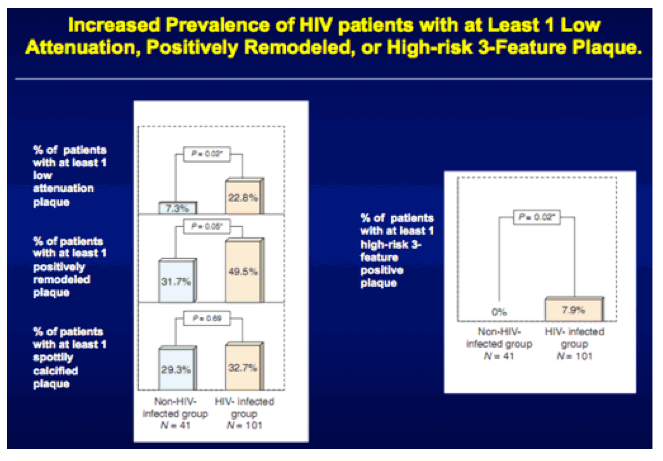
Steve Grinspoon from the Massachusetts General Hospital also looked at coronary plaques in HIV-infected and -uninfected patients. Specifically, the study focused on, so called, vulnerable plaques - those that are most at risk for rupture and triggering of an acute coronary event.9 He and his team recruited 102 HIV+ men and 41 HIV- men without known cardiac disease who were matched by age, blood pressure, LDL cholesterol and smoking status (although there still seemed to be many more smokers in the HIV+ group 41% vs. 21%). Coronary CT angiography was done here to look for plaque characteristics associated with vulnerability to rupture and here again the HIV+ men were significantly more likely to have the major features associated with plaque vulnerability in adjusted analyses (for example, 7.9% of the HIV+ men had all three features compared to none of the controls).
Correlation between low attenuation (vulnerable) plaques and immune markers was also examined and sCD163, a marker of monocyte activation, was independently associated with these lesions. This is interesting as Dr. Grinspoon had previously showed us elegant images of activated monocytes lighting up the walls of the aortas of HIV+ patients he has studied.10
These imaging studies further support the observational data demonstrating that HIV+ patients have more CVD disease. The MACS study, however, shows us that a big part of this risk is due to the usual suspects. Dr. Grinspoon has had to work hard to find comparable controls to hold HIV+ patients against. "In adjusting for these traditional factors these investigators are attempting to isolate and describe the contribution of simply being HIV+ to these adverse clinical outcomes, and in practically each case they find there is a greater risk for those who are HIV+."
Whether this because of an 'X factor' - perhaps associated with monocyte activation - or some unmeasured confounding factors still remains unclear. Further, while these and the other studies adjust for traditional risks and in doing so detect an HIV-effect that ante-ups CVD risk, when we see a patient in clinic we do not do any such adjustments. For the majority of those living and aging with HIV one need not look too deeply for CVD risk as it stares us in the face in the blood pressures, waist lines, lipid profiles, and bulging shirt pockets stuffed with a pack of Newport's. In these cases, the 'prematurity' of aging we see largely reflects a relative abundance of these traditional risk factors.
But, what about the patient without much in the way of CVD black marks? Does that non-smoking, eulipidemic, normotensive, non-diabetic patient face a greater than expected risk of CVD? Here again these data do suggest something amiss. Understanding how big a contribution this otherwise unexplained risk makes to the development of CVD during HIV infection is where this work need to go.
Preventing CVD
Regardless of what the mosaic of factors leading to CVD in HIV looks like, a considerable amount of energy is being exerted in our clinics to prevent the development of heart disease. Foremost, we are throwing statins and, to a lesser extent aspirin, at the problem. A few CROI presentations examined statin use in in HIV+ patients.
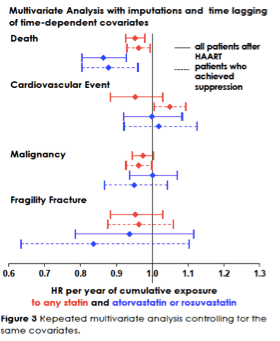
Another VA study that looked at the effects of statin therapy on mortality and select non-AIDS clinical outcomes (CVD, non-HIV associated malignancies, fractures) among 25,884 HIV+ patients treated with antiretrovirals between 1995-2009.11 Over a median follow-up of about 6 years, 35% of the patients had received statin therapy. In analyses adjusting for major risk factors and imputing missing values and time-lagging for time-dependent variables, cumulative exposure to statins was associated with a reduction in mortality with a stronger effect seen among the ~16,000 with virologic suppression and with use of more potent statins (atorvastatin and rosuvastatin). However, receiving a statin was not observed to reduced incidence of CVD (or non-AIDS malignancies or fractures). The authors, speculate that it could well be that statins do not reduce the occurrence of life-threatening events such as CVD but may make them more survivable.
An actual placebo controlled, randomized trial of statin therapy was conducted by Grace McComsey and colleagues at Case Western Reserve, who looked at the effect of rosuvastatin (10 mg) on an array of markers of inflammation, coagulation, and immune activation in patients with controlled viral replication and normal LDL cholesterol levels.12 Following 24 weeks of rosuvastatin, significant changes were seen in markers of vascular inflammation and monocyte activation in the patients treated with the statin (n=72) compared to the placebo group (n=75). Specifically, significant declines were measured in levels of Lp-PLA2 (a marker of vascular inflammation), sCD14 (a marker of monocyte activation), and CD14dim/CD16+TF+ (patrolling monocytes). There was a reduction of hsCRP with statin therapy by more than 10% and IL-6 dropped over 20% compared to placebo but these were reported to be not statistically significant. These data will certainly be a boost for more liberal use of statin therapy among the worried aging - although the next paragraph may temper that enthusiasm a bit.
The other side of the statin coin could be diabetes mellitus. Development of impaired glucose tolerance has been linked to statins in the general population (particularly post-menopausal women)13 and two studies presented at CROI examined this association in people living with HIV infection. One study found such an association, while the other not only did not it actually detected a protective effect of statins against diabetes. The first was an analysis of data from the US-based HIV Outpatient Study (HOPS) that found among ~5,000 patients (12.6% of whom started statins) 355 were diagnosed with diabetes mellitus.14 In multivariable analyses, each year of statin exposure was associated with a 10% hazard of development of diabetes (p=0.038) [insert sad face here]. But, glass half full types can take solace from the other study, done in Italy that found in a clinical cohort of ~5,300, statins significantly reduced the risk of incident diabetes.15
Xenophobic tendencies aside, it is hard to dismiss the HOPS study given it aligns with statin experience in HIV-uninfected cohorts (albeit the magnitude of the risk seen in HOPS was lower than that reported in the study of post-menopausal women). The bottom line is that monitoring for glucose intolerance (blood glucose testing, urinalysis, hemoglobin A1C) should be steeped up for those on statins.
Renal Function
Shifting to renal function, investigators from the CDC's Medical Monitoring Projects examined the prevalence of chronic kidney disease (CKD), defined as <60 ml/min/1.73m2 using the Modification of Diet in Renal Disease (MDRD) equation in this representative sample of patients in care in the US.16 Among those in the database in 2009, 90% (3,814) of patients had at least one serum creatinine measure. While, overall renal health of the population was good with an estimated glomerular filtration rate (eGFR) of 93.3 ml/min/1.73m2, 7.6% had grade 3 or greater CKD (eGFR <60 ml/min/1.73m2) - most of these with levels between 30 and 60. A number of factors were associated with CKD including female sex, longer duration of HIV, AIDS diagnosis, CD4 cell count <350/uL, and age. Older age had the greatest impact on renal function with those 40-59 years having an adjusted prevalence ratio almost three times that of younger patients and those over 60 years of age having a prevalence ratio of almost 8 compared to the youngest group.
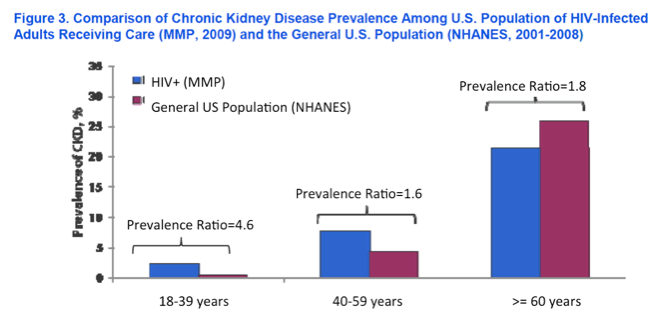
Interestingly, while the burden of CKD among those with HIV was greatest for the older patients, in comparing these data with those from the NHANES data from the general US population, rates of CKD were substantially higher for HIV+ patients who were younger than 60 years compared to the general population. This makes sense as CKD is uncommon in young persons in general and even among those with HIV, but among those with the virus there is a greater risk for CKD at an early age, possibly due to HIV-related and -unrelated risks, the latter more common among those with HIV. The study did not look at medication or co-morbidity as factors.
Another noteworthy study did look at ART effects on renal function. AIDS Clinical Trials Group (ACTG) trial compared lopinavir/ritonavir with nevirpaine in 741 African women also treated with tenofovir+FTC.17 There was a significantly shorter time to renal events among the women receiving lopinavir/ritonavir compared to those on nevirapine. In the lopinavir/ritonavir arm 4.9% experienced a renal event (2.4% had an increase in serum creatinine two times or greater than upper limit of normal), while 1.6% receiving nevirapine had a renal event (0.8% had an increase in serum creatinine two times or greater than upper limit of normal). Those on lopinavir had double the risk of a need to modify medication due to creatinine increase. Moreover, five of the women with renal toxicity died. These data, albeit from a resource-limited setting, provides further support for a role of ritonavir-boosted protease inhibitor therapy on nephrotoxicity of tenofovir.
Inflammation and Immune Activation
Perpetual immune activation and aberrant levels of inflammation are the glue that hold together a conceptual framework of aging and HIV. Supporting, if not introducing, this concept was the SMART Trial's finding of an increased risk of adverse outcomes among patients with relatively high d-dimer and inflammation markers, as well as demonstration in other studies of improvement in such markers once ART is initiated.18
However, an ACTG analysis of data collected from patients starting ART uncouples inflammation and immune activation - challenging our understanding of the basis of inflammation in treated HIV+ patients.19 This case-control study included patients who were initially ART-na�ve, started ART and maintained an HIV RNA level <400 copies/mL for a year. Cases were those who developed non-AIDS related events including non-accidental non-AIDS death, MI, stroke, non-AIDS cancer, or serious non-AIDS bacterial infection. Controls (2-3/case) had an endpoint-free follow-up time that was greater than the follow-up time of the case to whom they were matched by age, sex, baseline CD4+ cell count ART regimen and parent ACTG study. Stored blood was run for a host of inflammatory and coagulation markers, as well as markers of immune activation and senescence.
There were 143 cases and 315 controls. Events among the cases were: 25 MI, 16 stroke, 52 non-AIDS-related cancer, 37 serious bacterial infection and 13 non-accidental deaths. Controls had a greater CD4+ cell gain (+186/uL versus +129/uL) after ART and increase in CD4+ cell count was associated with reduced risk of non-AIDS events. However, inflammatory and coagulation markers including IL-6, sTNFR-I and -II, sCD14, K/T ratio and d-dimer, but not markers of T-cell activation, were strongly associated with the non-AIDS events. Therefore, inflammation happens and may not always be a consequence of immune activation.
Lastly, an intriguing randomized study presented at the conference demonstrated that intensification of ART with raltegavir in 31 patients on ART with HIV RNA levels <40 copies/mL for at least a year, increased 2-long terminal repeat (2-LTR) circles, indicating the interruption of HIV DNA integration within the cell nucleus.20 This effect was more profound in those on PI-based therapy. Raltegravir did not have an effect on immune activation or low-level viremia using an ultrasensitive assay. But, there was a significant decrease in d-dimer levels in the group receiving the active drug. This finding suggests that in some patients there may be persistent low-level replication of virus in some reservoirs that may have an inflammatory effect and can be suppressed.
Conclusions
While the exciting news of a baby fortunate to be spared HIV infection commanded attention in Atlanta, there were a number of other important presentations at CROI, described above, that help move us incrementally toward a more nuanced understanding of what HIV does to those growing old with this virus. A steady beat of studies make clear that people living with HIV face a heightened risk for diseases that are hallmarks of our built-in obsolescence. That much of this is at the hands of mundane contributors is important to appreciate - although it does not absolve HIV (or its treatment) of responsibility. It will be the next phase of research that will hopefully place all of this in perspective. Meanwhile, we carry on - vigilant in screening and diligent in following standard disease prevention guidelines. Our patients on the other hand also need to be reassured and to be calm, but not to carry on with behaviors that enhance the odds of diagnosis of serious non-AIDS events. That alone may not make all the difference but a difference it will make.
References
1. Justice AC. HIV and aging: time for a new paradigm. Curr HIV/AIDS Rep. 2010 May;7(2):69-76 (Estimated 50% of HIV+ population in US will be >50 years of age by 2015, subsequently updated to 2017).
2. Scheer S, et al. 6th IAS; Rome, Italy; July 17-20, 2011. Abst. TUPE131.
3. France, D. Another Kind of AIDS Crisis. New York Magazine. Nov 1, 2009. http://nymag.com/health/features/61740/
4. Guaraldi G, et al. Premature age-related comorbidities among HIV-infected persons compared with the general population. Clin Infect Dis. 2011 Dec;53(11):1120-6
5. Althoff K, et al. CROI 2013. Abstract 59.
6. Petoumenos K , et al. CROI 2013. Abstract 61
7. Losina E, et al. CROI 2013. Abstract 747
8. Post W, et al CROI 2013. Abstract 62
9. Zanni M, et al. CROI 2013. Abstract 63
10. Subramanian S, et al. Arterial inflammation in patients with HIV. JAMA. 2012 Jul 25;308(4):379-86
11. Drechsler H, et al. CROI 2013. Abstract 765
12. McComsey G, et al. CROI 2013. Abstract 186LB
13. Culver AL, et al. Statin use and risk of diabetes mellitus in postmenopausal women in the Women's Health Initiative. Arch Intern Med. 2012 Jan 23; 172(2):144-52
14. Lichtenstein K, et al. CROI 2013. Abstract 767
15. Spagnuolo V, et al. CROI 2013. Abstract 766
16. Garg S, et al. CROI 2013. Abstract 809
17. Mwafongo A, et al. CROI 2013. Abstract 152
18. Neuhaus J, et al. Markers of inflammation, coagulation, and renal function are elevated in adults with HIV infection. J Infect Dis 2010;201(12):1788-95.
19. Tenorio A, et al. CROI 2013. Abstract 790
20. Hatano H, et al. CROI 2013. Abstract 42
|
| |
|
 |
 |
|
|