 |
 |
 |
| |
Relationship Between Transaminase Levels and Plasma Pharmacokinetics Following Administration of MK-5172 With Pegylated Interferon Alfa-2b and Ribavirin to HCV Genotype 1 Treatment-Naïve Patients
|
| |
| |
Reported by Jules Levin
EASL 2013 Amsterdam April 24-28
Luzelena Caro, Lihong Du, Shelby Huang, Larissa Wenning, Jing Su, Peggy Hwang, Robert Valesky, Christopher Gilbert, Jacqueline Gress,
Frank Klaassen, Isaias Noel Gendrano, Joann Brunhofer, Michael Cooreman, Jan Huisman, Niloufar Mobashery
Merck, Whitehouse Station, New Jersey, USA
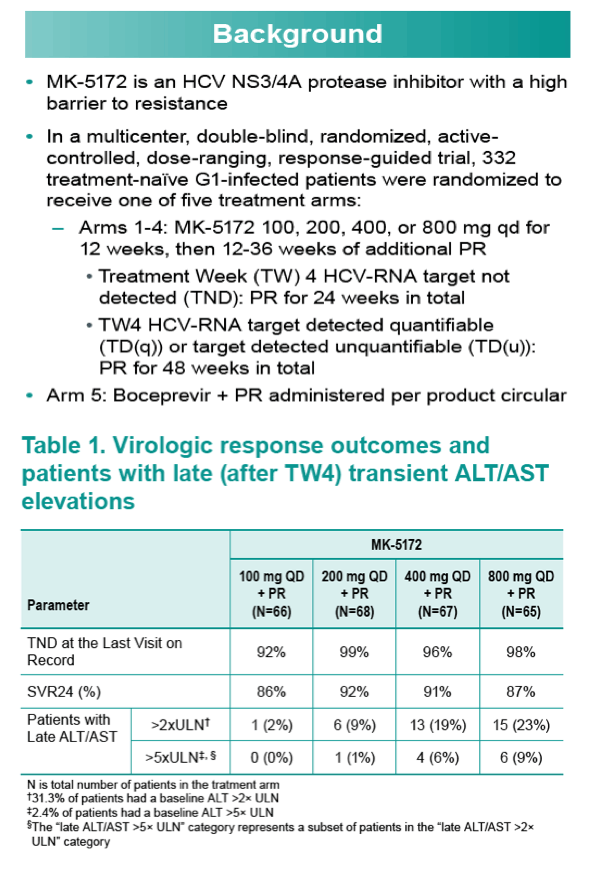
Background and Aims: MK-5172 is an HCV NS3/4A protease inhibitor with a high barrier to resistance. 332 treatment-naïve Genotype (G) 1-infected patients were randomized to receive boceprevir + Pegylated Interferon Alfa-2b and Ribavirin (PR) or MK-5172 100-, 200-, 400-, or 800-mg QD + PR for 12 weeks followed by 12 (RVR achieved) or 36 weeks (no RVR) of PR. Therapy was highly effective with >85% achieving SVR12. While ALT/AST levels normalized by week 4, they increased to >2X and >5X upper limit of normal (ULN) in a minority of patients in a dose-dependent fashion (Table). Analyses were conducted to explore the relationship between plasma pharmacokinetics (PK) and late ALT/AST elevations.
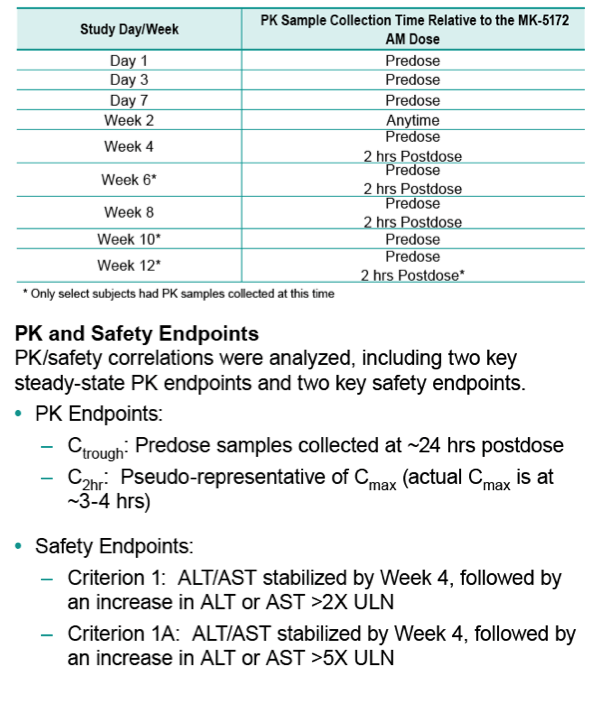
Methods: Patients in a PK sub-study provided MK-5172 plasma samples at select visits through Week 12. PK/safety correlations were analyzed, including PK endpoints for steady-state Ctrough and C2hr and two safety endpoints (late ALT/AST >2xULN or >5xULN). Logistic PK/safety regression analyses were conducted to assess the probability of the safety endpoint occurring at a given PK parameter value; the PK predictability for each endpoint was determined by performing Receiver Operating Characteristic and Negative Predictive Value analyses.
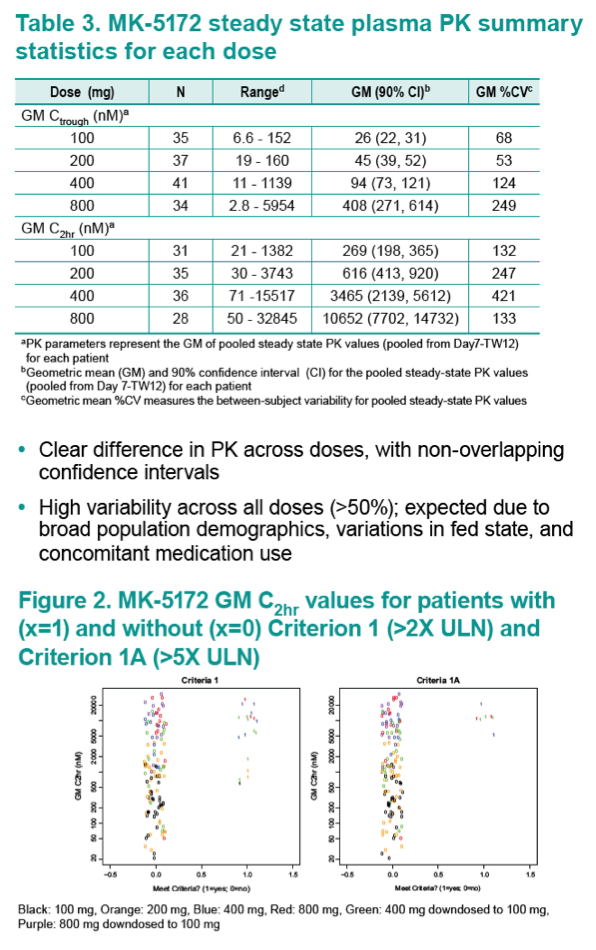
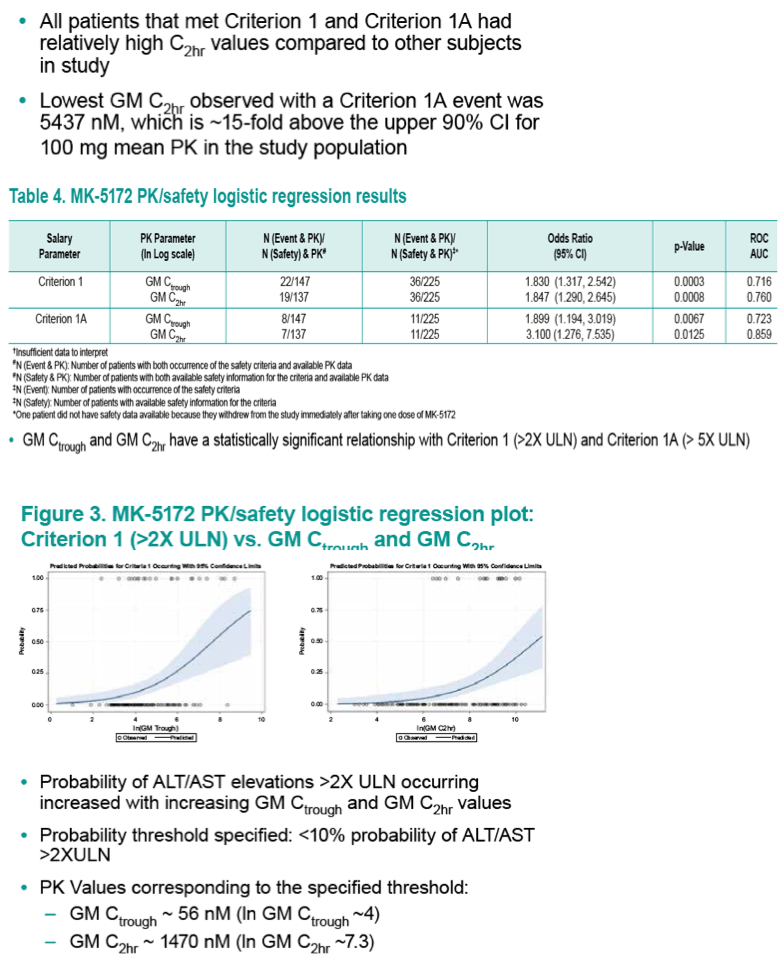
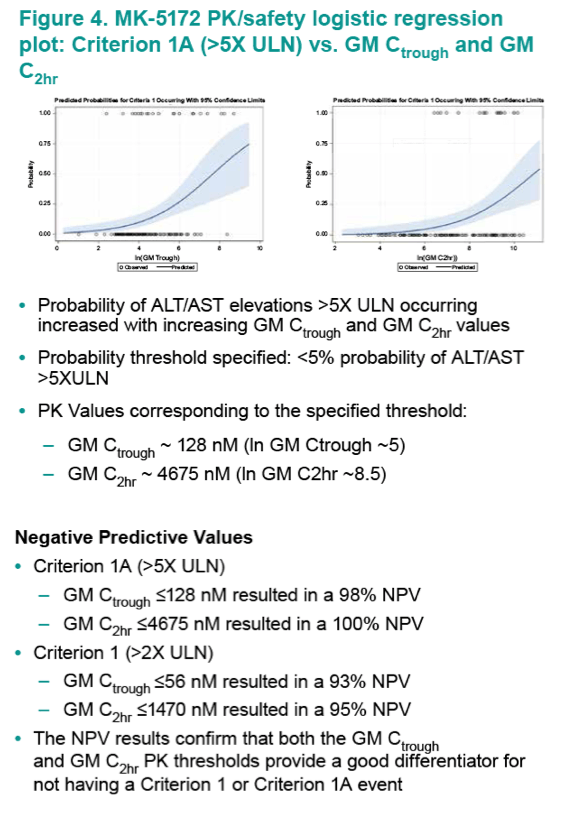
Results: Geometric mean (GM) Ctrough and C2hr values were well-correlated with both safety endpoints. A GM Ctrough and GM C2hr of 56 nM and 1470 nM, respectively, were predicted to have a ≤10% mean probability of ALT/AST >2xULN occurring, and are ~2- and ~4-fold above the upper 90% Confidence Interval (CI) for the 100-mg dose. A GM Ctrough and GM C2hr of 128 nM and 4675 nM, respectively, are predicted to have a mean probability of ≤5% of >5xULN occurring, and are ~4- and ~13-fold above the upper 90% CI for the 100-mg dose.
Conclusions: Plasma MK-5172 PK is associated with key ALT/AST safety endpoints. Combined with high SVR rates for the 100-mg dose, the PK/Safety analysis supports the low risk for a hepatic safety signal and continued development of MK- 5172 at a dose of 100-mg QD.



|
| |
|
 |
 |
|
|