 |
 |
 |
| |
Single tablet regimens do not necessarily translate into more durable HIV treatments: "initial use of RAL-based regimens was associated with a better outcome........patients starting HAART with RAL were less likely to change regimen...... the main reason for 1st line discontinuation/switch remain the adverse effects, very few virological failure were observed"
|
| |
| |
IAS 2013 Kuala Lumpur June 30-July 3
Presented by Benoit Trottier (Canada).
B. Trottier, N. Machouf, R. Thomas, S. Lavoie, S. Vezina, M. Boissonnault, E. Huchet, D. Longpre, L. Charest, D. Legault, M. Munoz
Clinique Medicale L'Actuel, Montreal, Canada
Background: Recently, many studies depicted the superiority of single tablet regimen (STR) in terms of efficacy, adherence and treatment durability because they offer a very low pill burden and a QD regimen. However, most previous clinical studies had concluded that absence of drug toxicity was more important for a durable regimen than simplicity of administration. We decided to compare those 2 strategies.
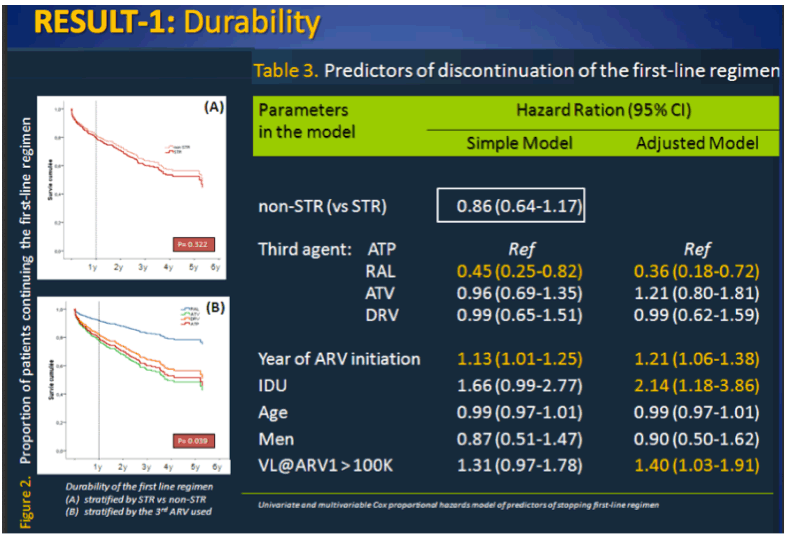
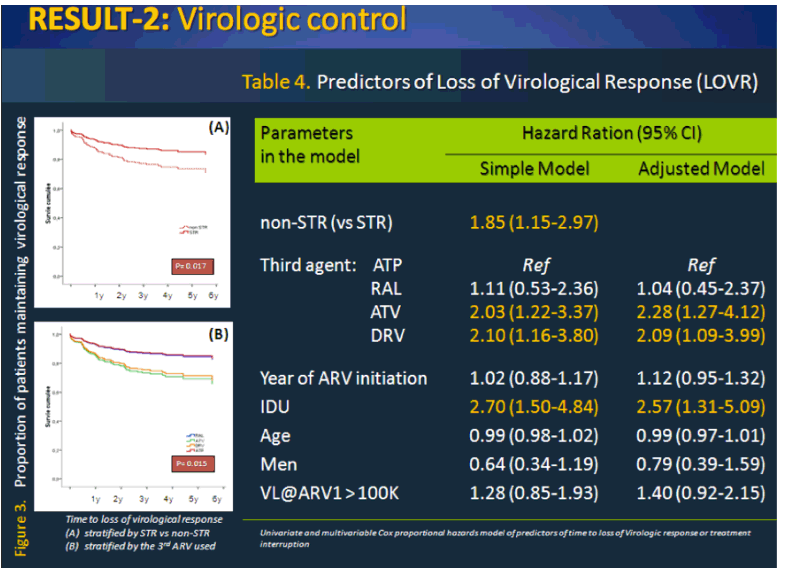
Methods: From our clinical cohort database, we selected patients having initiated, since 2007, any of the 4 recommended first-line regimens. One STR: efavirenz/emtricitabine/tenofovir (Atripla-ATP). Or 3 non-STR: 2 NRTI + either raltegravir (RAL), atazanavir/r (ATV/r) or darunavir/r (DRV/r). The main outcome was time until discontinuation of the first regimen. Cox proportional hazards models were used to estimate the hazard ratios (HR) of reaching the endpoint. Final model was adjusted for age, gender, baseline CD4, baseline HIV-RNA, risk factor and year of treatment initiation.
Results: A total of 586 patients were included. 94% were male; median age 42. The median length of follow-up was 2.7y (IQR:1.4-3.8). At baseline, the median HIV-RNA was 4.7 log (36%>100,000 copies/ml) and the median CD4 was 310 cells/mm3 (with 16%< 200 cells/mm3). No statistically significant difference between the groups was detected. Overall, 209 (36%) patients discontinued their regimen (33% in the STR; 37% in the group of non-STR, p=0.168). The treatment duration was equivalent in both categories. However, after adjustment for covariates, stratified analyses show that patients starting HAART with RAL were less likely to change regimen than those who started with either ATP or DRV/r, and the HR for ATV/r was the highest (cf.Table).
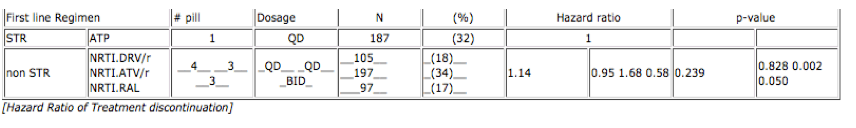
Conclusion: Our findings suggest that STR regimens do not necessarily result in a more durable treatment. Even with a higher pill burden and a BID schedule, the initial use of RAL-based regimens was associated with a better outcome.
------------------------------------------------------
Single Tablet Regimens do not Necessarily Translate into More Durable HIV Treatments




|
| |
|
 |
 |
|
|