 |
 |
 |
| |
Improvement of Central Fatigue is Associated with Sustained Virologic Response (SVR)
Following Sofosbuvir-Containing Regimens
|
| |
| |
Reported by Jules Levin
EASL 2014 April 9-13 London, UK
Zobair M. Younossi 1,2, Maria Stepanova 2, Lynn Gerber 1.2, Fatema Nader 1,2, Spencer Frost 1, Sharon Hunt 1,2
1. Center for Liver Diseases, Department of Medicine, Inova Fairfax Medical Campus, Falls Church, VA, United States.
2. Betty and Guy Beatty Center for Integrated Research, Inova Health System, Falls Church, VA, United States.
Disclosure Information: Nothing to Disclose.

Program abstract
Background and aims: Fatigue, an important extrahepatic manifestation of HCV, is a complex condition that can be categorized into central fatigue (CF) and peripheral or physical fatigue (PF). The aim of this study is to evaluate the changes in fatigue in patients receiving SOF in FUSION and NEUTRINO clinical trials.
Methods: Fatigue was assessed using three validated questionnaires [FACIT-F (Functional Assessment of Chronic Illness Therapy-Fatigue), Chronic Liver Disease Questionnaire-HCV (CLDQ-HCV) and Short Form-36 (SF-36)]. Items related to fatigue were selected, normalized to 0-1 scale, and used for exploratory factor analysis. HCV-related fatigue items were selected: Peripheral Fatigue (9 items) and Central Fatigue (4 items).
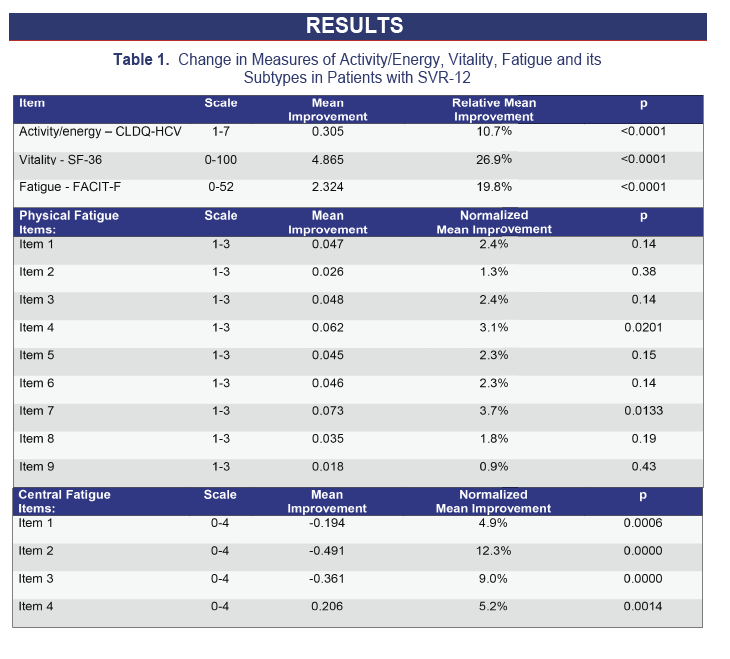
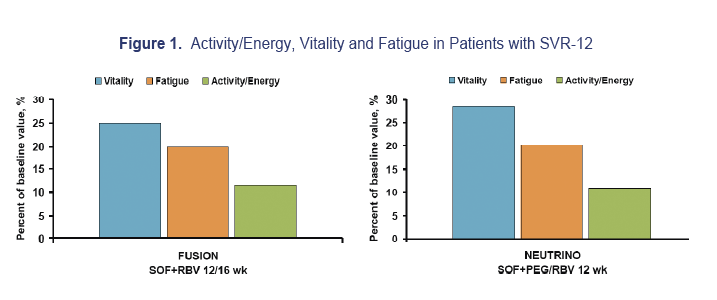
Results: A total of 413 patients achieved SVR-12 after treatment with SOF regimens. After achieving SVR-12, significant improvement of fatigue (compared to patients' own baseline score) was noted using all instruments: vitality scale of SF-36 (VT): 26.9%; activity-energy domain of CLDQ-HCV (AE): 10.7%; fatigue scale of FACIT-F (FA): 19.8% (all p< 0.0001). Additionally, after achieving SVR-12, the proportion of patients who scored below the general population norm by at least Minimally Clinically Important Difference (MCID) decreased from 32.7% to 27.6% (VT), 47.9% to 35.4% (AE) and 43.4% to 34.6% (FA). Finally, compared to patients' baseline, all items related to CF improved (all p< 0.0015) while only 2 items related to PF showed moderate improvement (p=0.0202 and 0.0133, respectively).
Conclusions: SVR-12 is associated with improvement of fatigue. Additionally, central fatigue is primarily affected by HCV clearance. Longer follow up may be needed to show any potential improvement in peripheral fatigue.



|
| |
|
 |
 |
|
|