| |
Report on new HCV drugs from Optum,
a consulting subsidiary of United Healthcare
|
| |
| |
"Can drugs as expensive as these really be cost-effective? The answer is yes......A study conducted soon after Victrelis and Incivek were introduced concluded that the new regimens were cost effective using quality-adjusted life-year (QALY) measures.15 In addition, the UK National Institute for Health and Clinical Excellence reported "robust cost-effectiveness" for treating hepatitis C with Victrelis and Incivek14.... Based on this experience, it is certainly possible that the combination of higher cure rates and reduced side effects, combined with reduced treatment times may mean even greater cost-effectiveness in the future...... Some experts believe that smaller manufacturers, or those who are slightly later to market with their new drugs, will have an incentive to negotiate aggressively on price with large health plans and PBMs"
------------------------------------
The debate over drug prices is heating up as insurers clash with pharmaceutical companies over a wave of expensive new products......Sovaldi.....A report from Optum, a consulting subsidiary of United Healthcare, agrees. Even very expensive drugs can be cost-effective if they work well enough, Optum said in a report (BELOW) issued before Sovaldi's price was released......."It is certainly possible that the combination of higher cure rates and reduced side effects, combined with reduced treatment times, may mean even greater cost-effectiveness in the future," Optum's analysts wrote.
........http://www.nationaljournal.com/health-care/is-an-84-000-hepatitis-drug-too-expensive-20140529
------------------------------------
http://www.consultantpharmacyinfocus.com/article/hepatitis-c-big-changes-coming-soon

Hepatitis C is the most common chronic blood-borne infection in the U.S. and is one of the leading causes of liver disease and liver cancer.
This article reviews the quickly evolving research and development for new hepatitis C treatments. By short-circuiting the progression of the disease they will have a significant impact both on the way hepatitis C is treated, and on employer plan costs.
Also with this topic we are proud to introduce a new feature called 2 Minute Drill. 2 Minute Drill is a reader-friendly infographic-style format that most readers should need only a few minutes to scan.
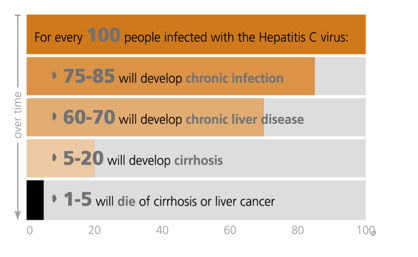
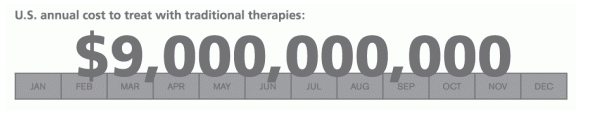
Until relatively recently, hepatitis C had been difficult to treat and nearly impossible to cure with existing therapies. Without the new and pending wave of highly effective treatments, is was estimated that the cost to treat the currently infected group of Americans over their estimated survival span could reach $360 billion in today's dollars.3 That translates into an annual health care cost as high as $9 billion for the affected U.S. population.3
The threat of hundreds of billions in pending future costs began to lighten considerably in May of 2011. That's when the US Food and Drug Administration (FDA) announced approval for a new class of drugs for treating hepatitis C. Two new drugs, Incivek® (telaprevir) and Victrelis® (boceprevir), for the first time offered a true clinical cure to more than 50 percent of those infected - regardless of whether they had received treatment for hepatitis C before.
Even more important for employer plan costs, these new drugs promised to significantly reduce the long-term health and cost consequences of chronic hepatitis C by short-circuiting the progression of the disease. [Read more here.]
But this is not the end of the story. In fact, the research effort around hepatitis C continues to accelerate. Additional new products and medicines are on the way, both immediately and in the near term. These will have a significant impact on the way hepatitis C is treated, and on employer plan costs.
Targeted care
Historically, one of the barriers to successfully treating hepatitis C is that there are at least 6 main hepatitis C genotypes (genetic variations) of the virus.1 Since each variation responds differently to available drug therapies, ideally a doctor would want to know exactly which genotype she needed to attack. But existing methods to identify virus genotypes have been slow and inexact, complicating treatment.4
Now a new test will make it easier to identify the exact form of the virus in a given patient. In June, the FDA approved a new, fully automated test that identifies the specific genotype of the hepatitis C virus.4 Produced by Abbott, this is the first FDA-approved hepatitis C virus genotyping test for patients in the United States.5
This is a vital step forward in treating hepatitis C. Now doctors can quickly determine which type of virus their patient has and use that knowledge to create a personalized, targeted diagnosis and treatment path for improved clinical outcomes.4
Even newer drugs
The research pipeline for hepatitis C also includes new drugs currently at various stages of seeking FDA approval. Several drug makers, including AbbVie Inc. (formerly Abbott), Gilead Sciences Inc., Medivir AB, Janssen and Bristol-Myers Squibb Co. are expected to bring a new generation of hepatitis C drugs to market in 2013 and 2014.6
The common goal for this new group of drugs is achieve a therapy that is not only clinically more effective, but that is simpler and easier to tolerate for large numbers of patients. One indication that this goal may be within reach is that the FDA has designated several of these experimental drugs as breakthrough therapies.7 This is an expedited development and review process for faster possible FDA approval. These include Gilead Sciences' sofosbuvir, Janssen's simeprevir and AbbVie's interferon-free combination therapy.7
Growing effectiveness
It is useful to briefly review the major advances in clinical efficacy for hepatitis C medications over just a few years. Traditional therapies (prior to 2011) commonly reported cure rates in the range below approximately 50 percent.8 The current state-of-the-art therapies, Incivek and Victrelis, (in combination with injected interferon and ribavirin pills) produce cure rates as high as 63%- 79% in first-time treatment patients.9
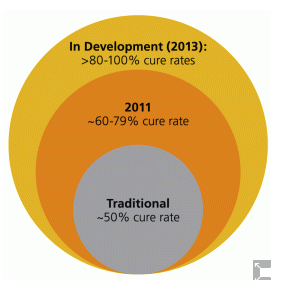
The experimental new drugs and combinations of drugs being tested now are reporting much higher cure rates. At least one has reported cure rates up to 100 percent in trials and several other combinations are reporting cure rates above 80 and 90 percent, albeit still in small studies.10
Simpler, more tolerable regimens
Both Incivek and Victrelis must be used in combination with interferon, an injectable drug that sometimes can cause serious side effects ranging from flu-like symptoms to depression.11 In fact, some people compare the experience to cancer chemotherapy.11
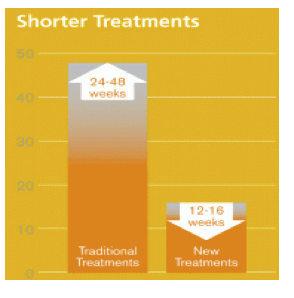
By contrast, almost all of the new drugs are being tested without the use of interferon, or any other injectable drug. As noted above, not only do the new drugs seem to be more effective, but they also work much more quickly, reducing the standard therapy duration from 24 to 48 weeks to only 12 to 16 weeks.10
While only a few of the new drugs have even been submitted for FDA approval at this time, there is still considerable anticipation building. Many now hope that within a few years we will see simplified, all-oral, interferon-free therapies with high cure rates for a wide range of the hepatitis C population.10
Benefits implications
Two factors will bear heavily on how the new wave of hepatitis C drugs will affect employer costs: the number of patients who receive the new drugs, and how the manufacturers price their products.
Number of patients
One manufacturer estimates that as many as 150,000 patients may seek treatment in the U.S. once the new class of all-oral drugs is on the market, compared with 85,000 patients in 2012.12 That's an increase of 76 percent.
The reason for the large increase is that many patients - even those who are currently aware that they are infected - are waiting to receive the new drugs. A survey taken in the spring of 2013 found that in the previous six months 60 percent of hepatitis C specialists had begun warehousing patients (i.e., intentionally delaying treatment) in hopes of using the next generation of treatments.13
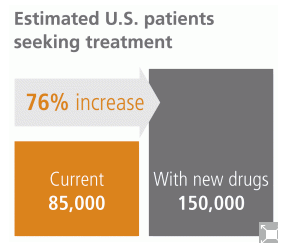
While the exact numbers of patients seeking treatment will vary by employer group, it seems clear that overall there could be a considerable influx of claims soon after a new FDA approval is announced.
Pricing
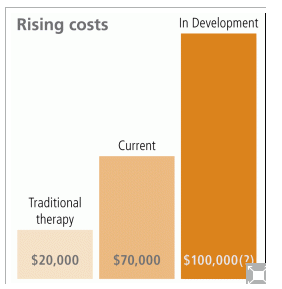
At this time we simply do not know what prices will be for the new drugs, since none of the manufacturers has committed to a number. This hasn't prevented great speculation, including predictions that the new Gilead drug (sofosbuvir) may cost up to $100,000 per patient for a course of treatment.6
While we wait for these details, it may be worthwhile to recall the pharmaco-economic analyses which support treatment of hepatitis C - even with expensive therapies. As it happens, we faced very similar questions when the previous class of new drugs arrived in 2011, so it's possible to draw on that experience.
Cost-benefit analysis
In addition to their improved clinical effectiveness, Victrelis and Incivek are also very expensive. Recall that they must be used in combination with the previously existing therapy (injected interferon and ribavirin pills) which costs approximately $15,000 to $20,000 for 48 weeks of therapy.8
Adding Victrelis and Incivek means an additional cost of nearly $50,000 per course of treatment.9 Can drugs as expensive as these really be cost-effective?
Can drugs as expensive as these really be cost-effective?
The answer is yes.
Hepatitis C-infected patients incur higher health care costs compared with a non-infected population matched by sex, age, and healthcare enrollment.1 Because costs are driven largely by end-stage liver disease, liver transplants and cancer, total hepatitis C- related health care expenses can be expected to decrease over time as successfully treated patients avoid progressing to more severe forms of the disease.14
A study conducted soon after Victrelis and Incivek were introduced concluded that the new regimens were cost effective using quality-adjusted life-year (QALY) measures.15 In addition, the UK National Institute for Health and Clinical Excellence reported "robust cost-effectiveness" for treating hepatitis C with Victrelis and Incivek.14
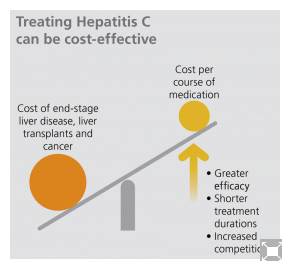
Based on this experience, it is certainly possible that the combination of higher cure rates and reduced side effects, combined with reduced treatment times may mean even greater cost-effectiveness in the future.
In addition, there is reason to be optimistic that with this new class of drugs, the hepatitis C marketplace may finally have matured enough to achieve a critical mass of competing manufacturers.16
Some experts believe that smaller manufacturers, or those who are slightly later to market with their new drugs, will have an incentive to negotiate aggressively on price with large health plans and PBMs.16
What can be done?
As with any drug class, OptumRx will work to obtain the best possible prices with our market leverage. In addition, we recognize that to ensure new specialty treatments for hepatitis C are being effectively managed, they require intensive management and member support.
We recommend:
· Prior authorization so that that only patients who can benefit from certain drugs receive coverage for them.
· Support: Patients taking these medications can benefit from ongoing education, adherence and clinical management programs like those in our Specialty Pharmacy program.
· Use formulary tiering to promote drugs of demonstrated cost-effectiveness (e.g., for OptumRx, Incivek is Tier 2 while Victerlis is Tier 3).
· Supply limits
· Our Price Protection program applies to drugs in this class.
Talk to your OptumRx representative to learn more about these creative ways to help your clients manage the rising costs in the hepatitis C category.
Conclusion
In short, this is a busy time for the entire hepatitis C community of stakeholders - patients, researchers, regulators, manufacturers, health plans and employer benefit plans. We will work to keep you informed as this rapidly-evolving scenario unfolds.
Notes
1. Centers for Disease Control and Prevention. Hepatitis C Information for Health Professionals. Updated May 7, 2013.
2. Centers for Disease Control and Prevention. Hepatitis C Information for the Public. Updated May 6, 2013.
3. The C. Everett Koop Institute, Dartmouth Medical School. Associated Health Costs - United States. Accessed June 24, 2013.
4. FDA NEWS RELEASE. FDA approves first genotyping test for patients with hepatitis C virus. June 20, 2013.
5. Abbott Press Release. Abbott Announces New Test to Help Doctors Determine Personalized Treatment Path for Patients with Hepatitis C. June 20, 2013
6. Bloomberg News. Drugs Go From Hit to Dud in Two Years in Hepatitis Race. Jun 3, 2013.
7. HepMag.com. The FDA Makes Hepatitis C a Priority. June 14, 2013
8. U.S. Department of Veterans Affairs. Hepatitis C for Health Care Providers: Hepatitis C Treatment. Reviewed/Updated Date: January 10, 2013.
9. American Journal of Managed Care. Initial Prescription Utilization of the New Hepatitis C Agents. April 04, 2012.
10. HepMag.com. The Hepatitis C Drug Pipeline Report: A Groundswell to Become a Sea Change. April 15, 2013.
11. Inside Stanford Medicine Benefits of hepatitis C treatment outweigh costs for patients with advanced disease, study shows. Feb. 20, 2012.
12. Bloomberg. AbbVie Says It Can Be First With New Hepatitis C Drugs. Jun 13, 2013.
13. BioTrends Research Group. The Majority of Physicians that Treat Hepatitis C Virus (HCV) Have Begun "Warehousing" and Preparing Their HCV Patients for the Next Generation of HCV Treatments May 23, 2013.
14. Forbes. A Public Health and Healthcare Spending Time Bomb: Hepatitis C. June 19, 2013
15. Pharmacy Times. Triple Therapy for Hepatitis C Can Be Cost-Effective. February 27, 2012.
16. HepMag.com. Hep C's Blockbuster Season. May 15, 2013.
|
|
| |
| |
|
|
|