| |
4 weeks/4 Drugs-Daclatasvir/Sofosbuvir/Asunaprevir/BMS325
|
| |
| |
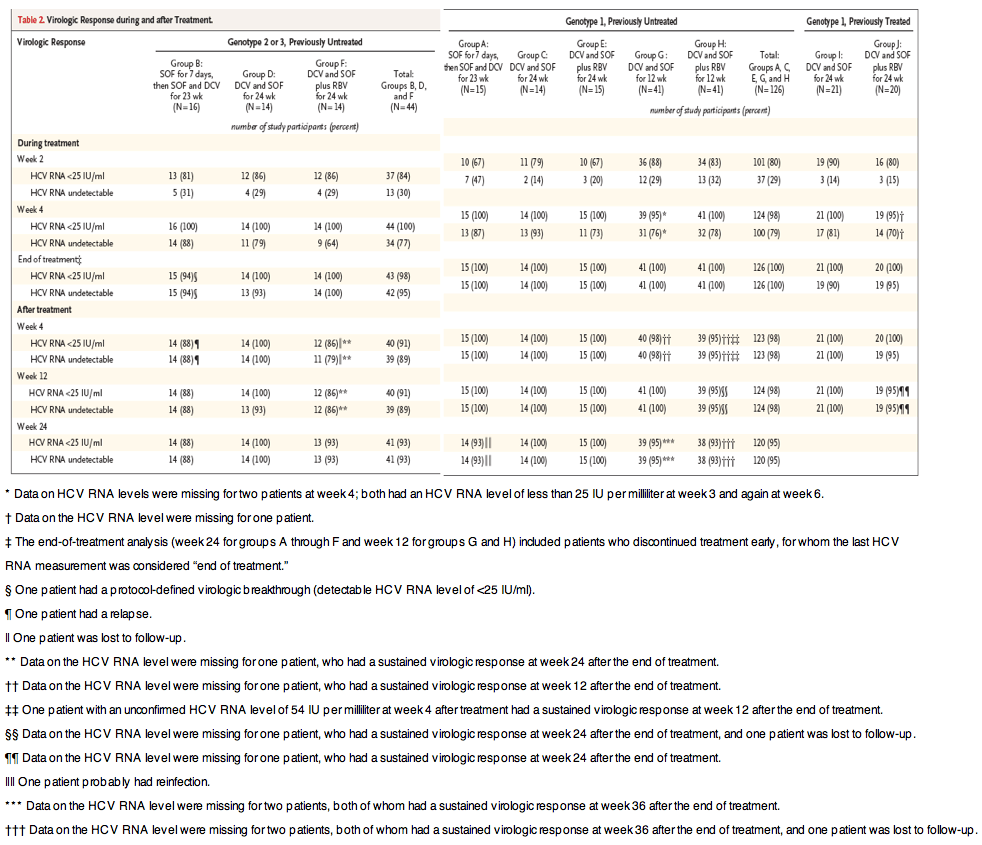
In light of the news this week that announced publicly for the 1st time that BMS is starting a study of sofosbuvir+daclatasvir in combination (4-drug regimen) with other BMS drugs asunaprevir+BMS325 for 4 and 6 weeks, here is the recently published data on Daclatasvir+sofosbuvir for 12 weeks, note arms G & H in the table:
AND here were the original presentations of 24 weeks before the expansion to an arm of 12 weeks for which the results were reported in the NEJM earlier this year--
EASL/2012: Potent Viral Suppression With the All-Oral Combination of Daclatasvir (NS5A Inhibitor) and GS-7977 (Nucleotide NS5B Inhibitor), +/- Ribavirin, in Treatment-Naive Patients With Chronic HCV GT1, 2, or 3 (100% SVR gt1, 91% gt2) - (04/19/12)
EASL: Sustained Virologic Response With Daclatasvir Plus Sofosbuvir ± Ribavirin (RBV) in Chronic HCV Genotype (GT) 1-Infected Patients Who Previously Failed Telaprevir (TVR) or Boceprevir (BOC) - (04/27/13)
Daclatasvir plus Sofosbuvir for Previously Treated or Untreated Chronic HCV Infection, NEJM
http://www.natap.org/2014/HCV/011714_04.htm
"In this open-label study, we initially randomly assigned 44 previously untreated patients with HCV genotype 1 infection and 44 patients infected with HCV genotype 2 or 3 to daclatasvir at a dose of 60 mg orally once daily plus sofosbuvir at a dose of 400 mg orally once daily, with or without ribavirin, for 24 weeks. The study was expanded to include 123 additional patients with genotype 1 infection who were randomly assigned to daclatasvir plus sofosbuvir, with or without ribavirin, for 12 weeks (82 previously untreated patients) or 24 weeks (41 patients who had previous virologic failure with telaprevir or boceprevir plus peginterferon alfa-ribavirin). The primary end point was a sustained virologic response (an HCV RNA level of <25 IU per milliliter) at week 12 after the end of therapy"
|
|
| |
| |
|
|
|