 |
 |
 |
| |
Drug Interactions Between Direct-Acting
anti-HCV Antivirals Sofosbuvir and Ledipasvir and HIV Antiretrovirals
|
| |
| |
Reported by Jules Levin
15th International Workshop on Clinical Pharmacology of HIV & Hepatitis Therapy, Wash DC, 2014 May 19-21
Polina German, Philip S Pang, Steve West, Lingling Han, Karim Sajwani and Anita Mathias
Gilead Sciences, Foster City, CA

Program abstract:
Drug interactions between direct acting anti-HCV antivirals Sofosbuvir and Ledipasvir and HIV antiretrovirals
Introduction: A once daily fixed-dose combination tablet composed of NS5A inhibitor ledipasvir (LDV) 90 mg and NS5B inhibitor sofosbuvir (SOF) 400 mg is in Phase 3 clinical trials for the treatment of chronic HCV infection. We conducted a Phase 1 study in healthy volunteers to evaluate potential DDIs between LDV and an integrase inhibitor raltegravir [RAL], and LDV/SOF and NNRTI-based ARV regimens efavirenz (EFV)/emtricitabine (FTC)/tenofovir DF (TDF) [Atripla®; ATR] or FTC/rilpivirine (RPV)/TDF [Complera®; CPA] before recommending their co-use in HIV/HCV co-infected patients.
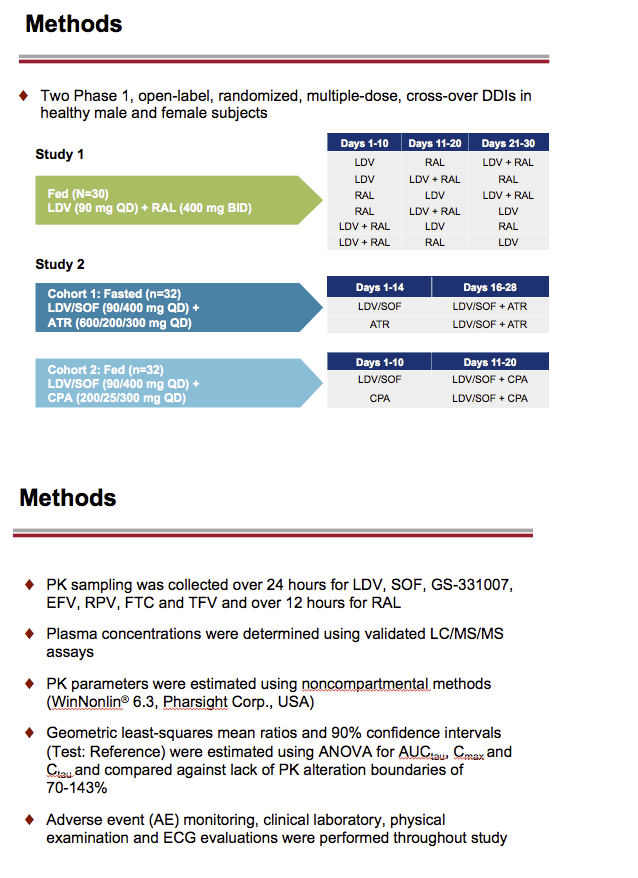
Materials & Methods; This was a multiple-dose, randomized, cross-over DDI study. Group 1 subjects (N=30, fed) received LDV 90 mg QD, RAL 400 mg BID and LDV+RAL each for 10 days. Group 2 subjects (N =32, fasted) received LDV/SOF 90/400 mg alone followed by co-administration with ATR (600 mg/200 mg/300 mg) QD or ATR alone followed by co-administration with LDV/SOF, for 14 days. Group 3 subjects (N=32, fed) received LDV/SOF alone followed by co-administration with CPA (200 mg/25 mg/300 mg) QD or CPA alone followed by co-administration with LDV/SOF, for 10 days.
LDV, SOF, GS-331007 (predominant circulating nucleoside metabolite) and ARV plasma concentrations were analyzed on the last day of dosing for each treatment and PK parameters were calculated. Geometric least-squares means ratios % and 90% confidence intervals (combination vs. alone) for LDV, SOF, GS-331007, and ARV AUCtau, Cmax and Ctau were estimated using a linear mixed effect model with fixed treatment, period and sequence effects, and random subject effects, and compared against pre-specified lack of PK alteration boundaries of 70-143%. Safety assessments were conducted throughout the study.
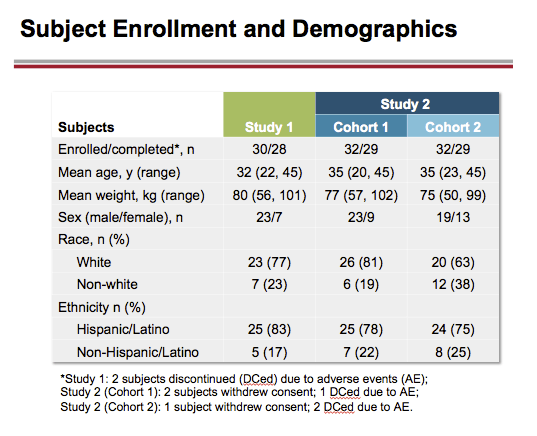
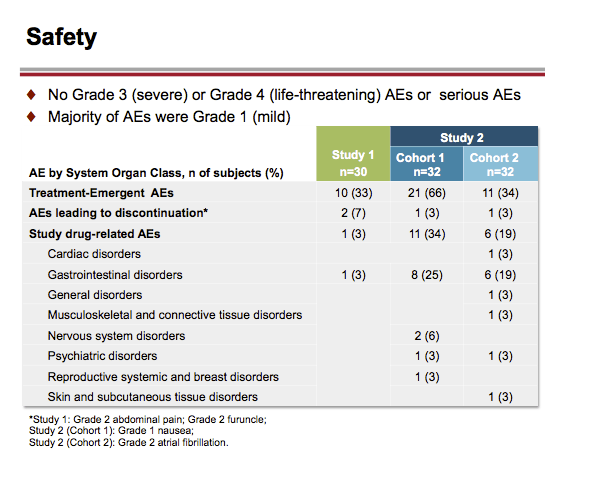
Results: Twenty-eight of 30 subjects (Group 1) and 29 of 32 subjects (Groups 2 and 3) completed the study. The majority of adverse events (AEs) were Grade 1. The most frequent AEs were constipation (Group 1), headache and dizziness (Group 2), and nausea (Groups 2 and 3). Two subjects in Group 1, 1 subject in Group 2, and 1 subject in Group 3 discontinued due to an AE.
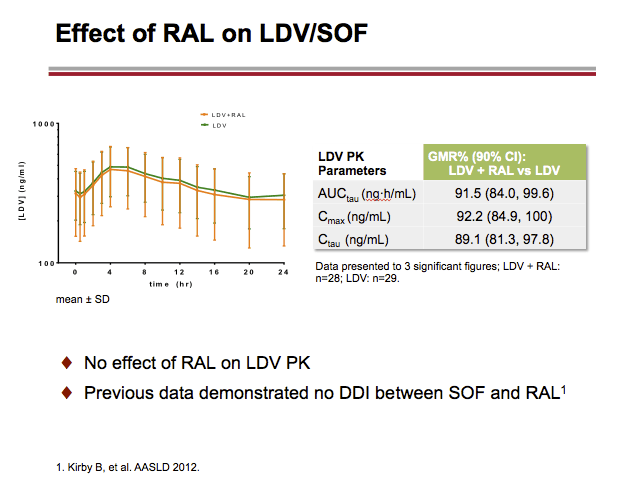
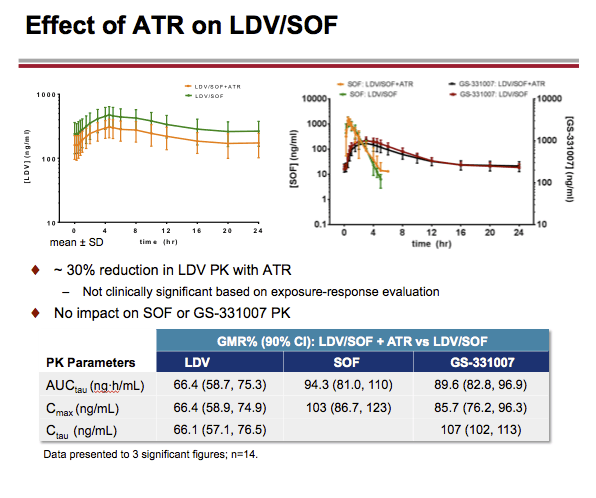
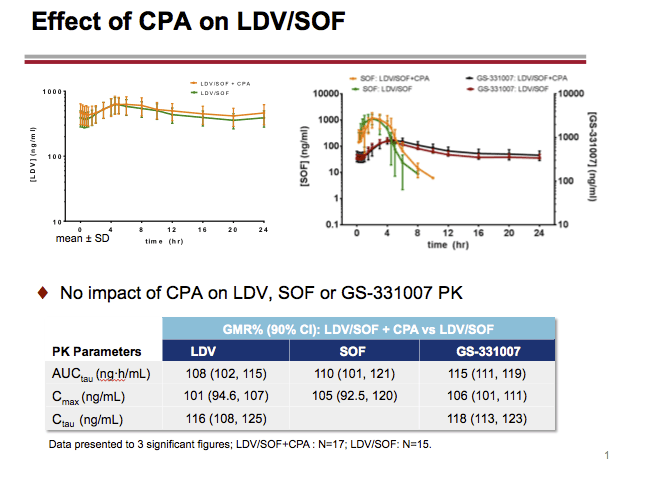
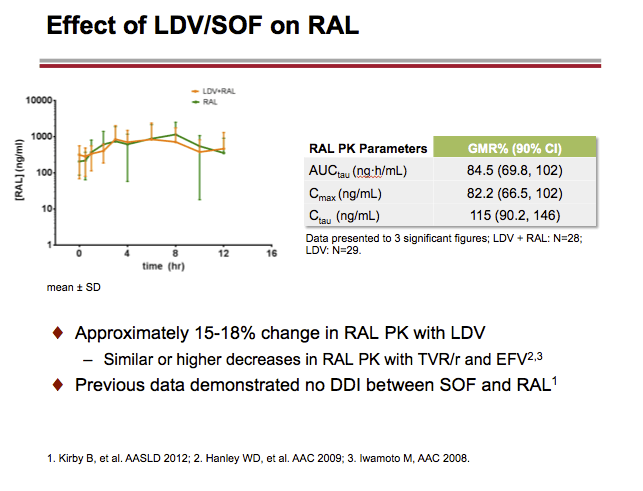
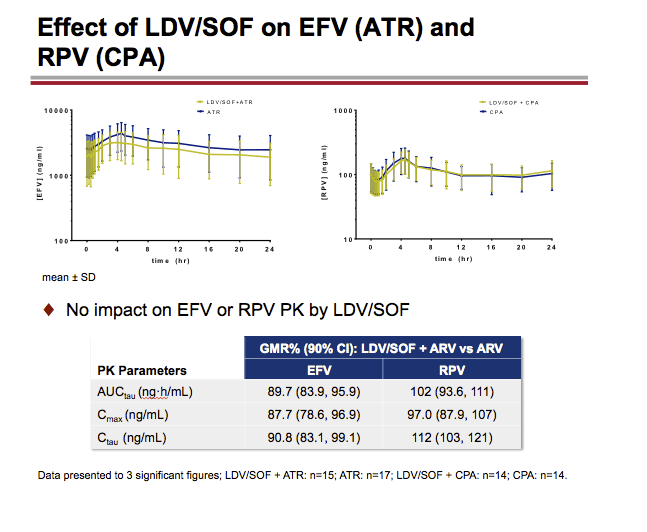
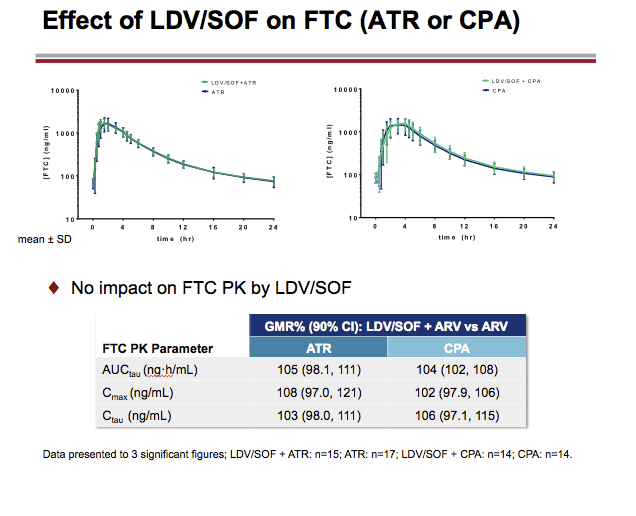
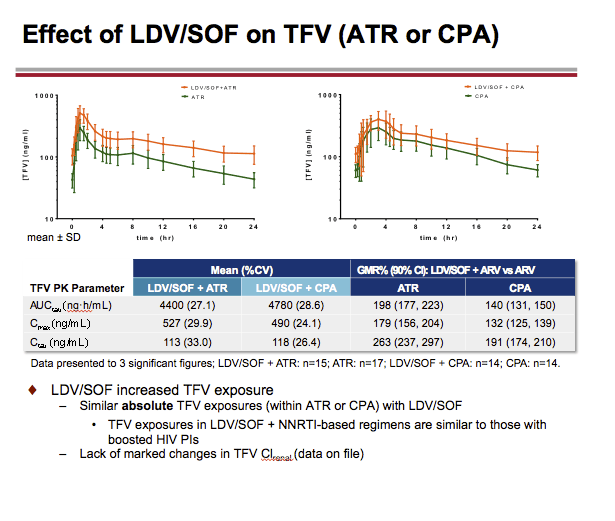
LDV or LDV/SOF PK was unaffected by RAL or CPA, respectively. A modest (~34%) decrease in LDV exposure with no impact on SOF or GS-331007 PK with ATR, and small (< 20%) changes in RAL PK with LDV, were observed on co-dosing. These changes were not deemed clinically relevant. EFV, RPV, or FTC PK was not affected by LDV/SOF. TFV exposure increases (ATR: ~1.8-2.6-fold; CPA: ~1.3-1.9-fold) were observed with LDV/SOF; overall, TFV absolute AUC values in the test (combination) treatments were comparable to those achieved when FTC/TDF is administered with ritonavir-boosted PIs, which do not warrant dose adjustment.
Conclusion: Study treatments were generally well tolerated. Cumulatively, results from this study and a previous DDI study between SOF and ARVs demonstrate that LDV/SOF may be administered with ATR, CPA, RAL, with a backbone of FTC/TDF.



|
| |
|
 |
 |
|
|