 |
 |
 |
| |
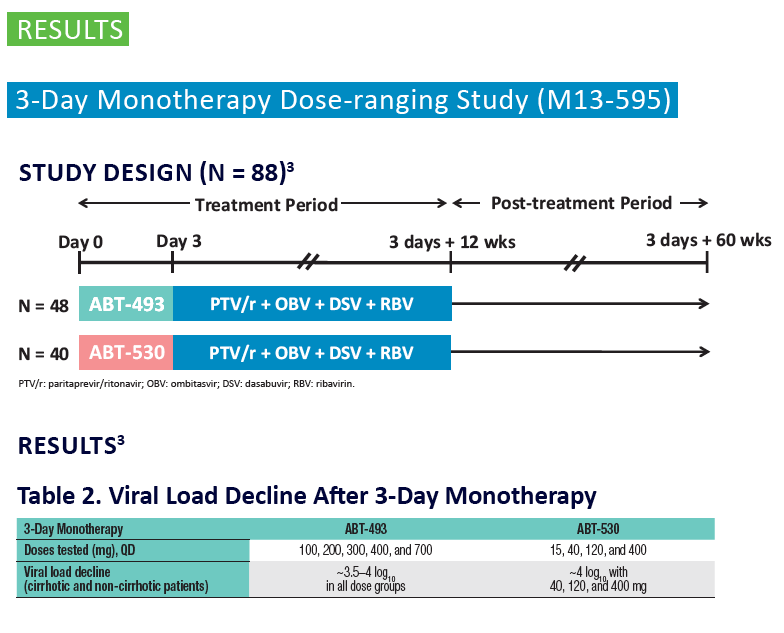
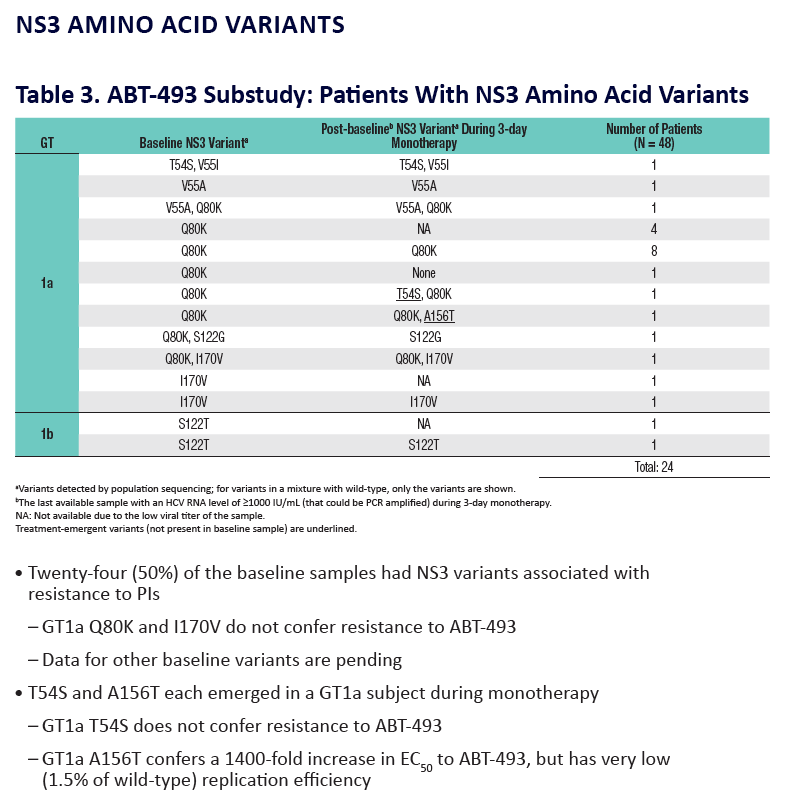
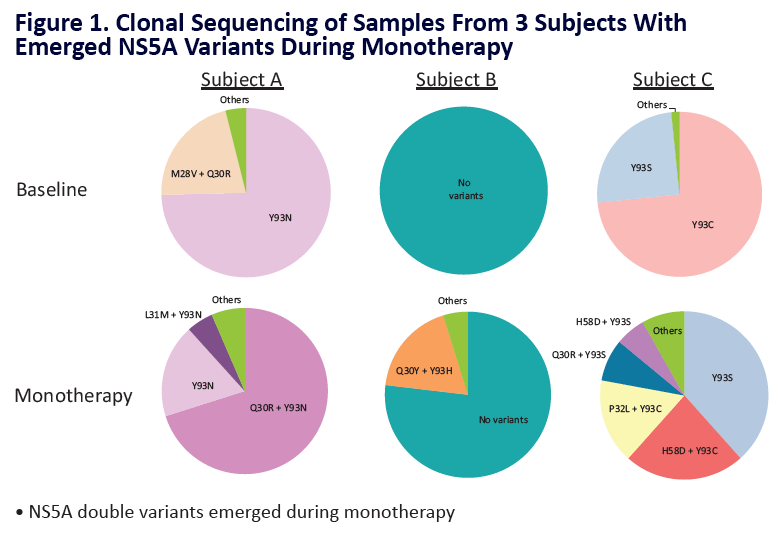
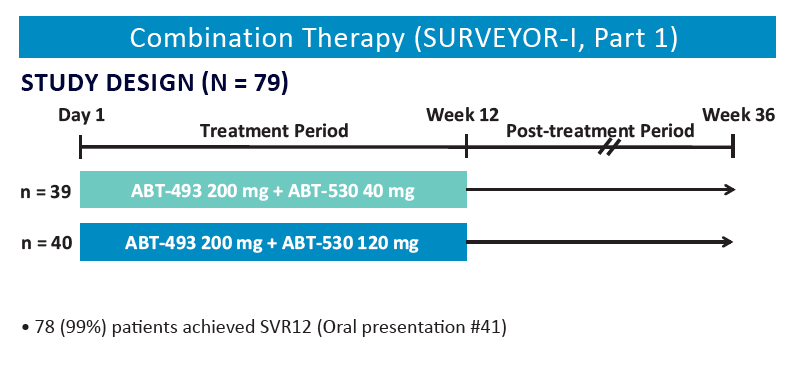
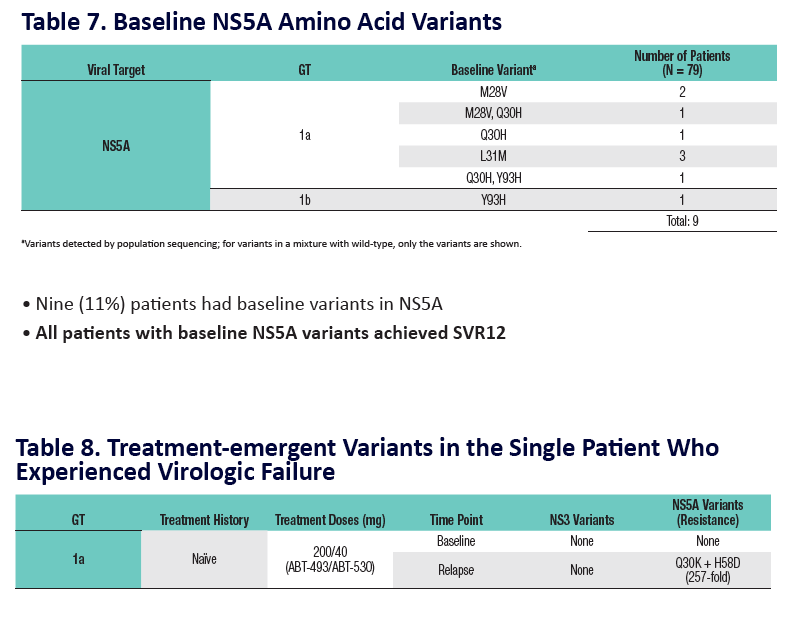
Analysis of HCV Genotype 1 Variants Detected During Monotherapy and Combination Therapy With Next Generation HCV Direct-Acting Anti viral Agents ABT-493 and ABT-530 "........COMBINATION THERAPY (SURVEYOR-I PART 1, PHASE 2 DOSE-RANGING STUDY) .....All patients with baseline NS3 or NS5A variants achieved SVR12 (Study SVR12: 99%)"
|
| |
| |
Reported by Jules Levin
AASLD 2015 Nov 13-17 San Francisco, CA
Teresa Ng, Tami Pilot-Matias, Rakesh Tripathi, Gretja Schnell, Thomas Reisch, Jill Beyer, Tanya Dekhtyar, Armen Asatryan, Federico Mensa, Andrew Campbell, Jens Kort, Christine Collins
AbbVie Inc., North Chicago, Illinois, United States









|
| |
|
 |
 |
|
|