 |
 |
 |
| |
No Pharmacokinetic Interaction Between HCV NS5A Inhibitor Elbasvir and
Buprenorphine/Naloxone in Healthy Volunteers
|
| |
| |
Reported by Jules Levin
AASLD Liver Meeting 2015 San Francisco, CA
November 13-17
William L. Marshall1; Ted Marenco2; Hwa-Ping Feng1; April M. Barbour1; Jocelyn Gilmartin1; Fang Liu1; Deborah Panebianco1; Angela Mirzac2; Mike Di Spirito2; Daria Stypinski2; Ziv Machnes2; Michael Gartner2; Marian Iwamoto1; Joan R. Butterton1; Wendy W. Yeh1
1Merck & Co., Inc., Kenilworth, NJ, USA; 2Celerion, Lincoln, NE, USA







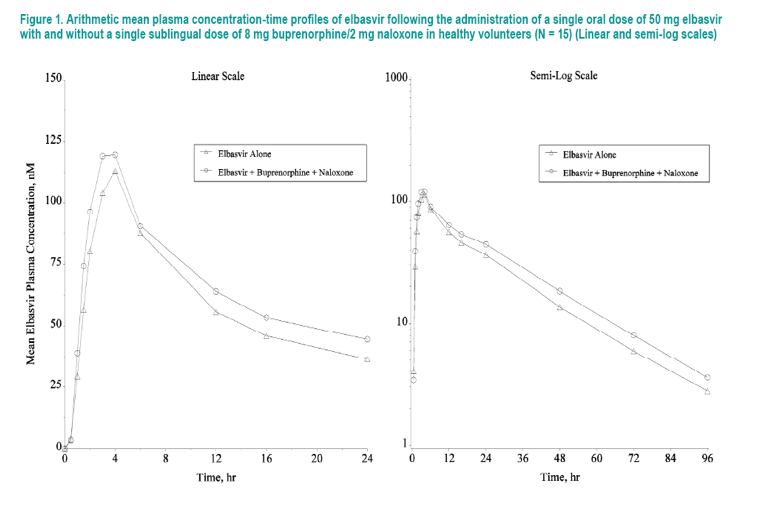
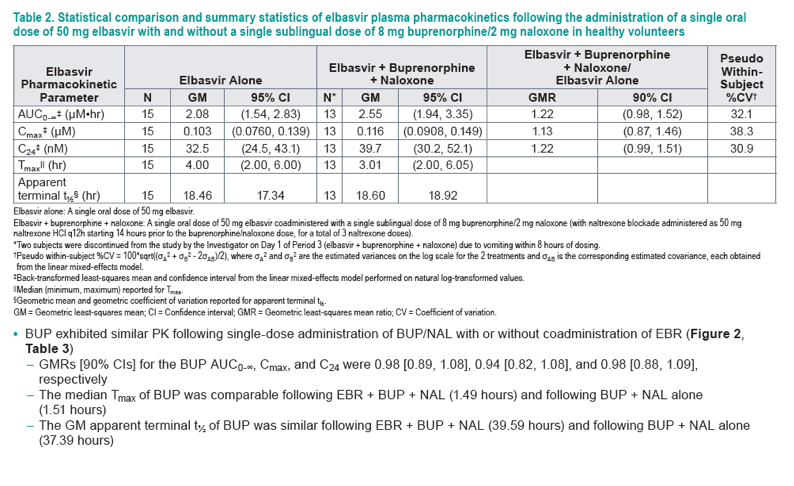
Figure 2. Arithmetic mean plasma concentration-time profiles of buprenorphine following the administration of a single sublingual dose of 8 mg buprenorphine/2 mg naloxone with and without a single oral dose of 50 mg elbasvir in healthy volunteers (N = 16 for buprenorphine + naloxone alone; N = 15 for elbasvir + buprenorphine + naloxone) (Linear and semi-log scales)
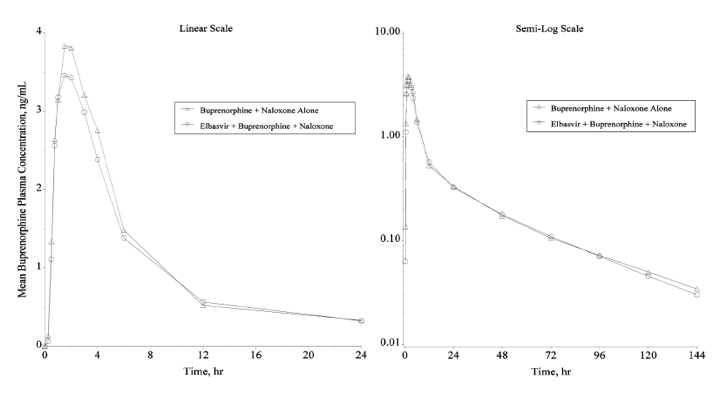
Table 3. Statistical comparison and summary statistics of buprenorphine plasma pharmacokinetics following the administration of a single sublingual dose of 8 mg buprenorphine/2 mg naloxone with and without a single oral dose of 50 mg elbasvir in healthy volunteers
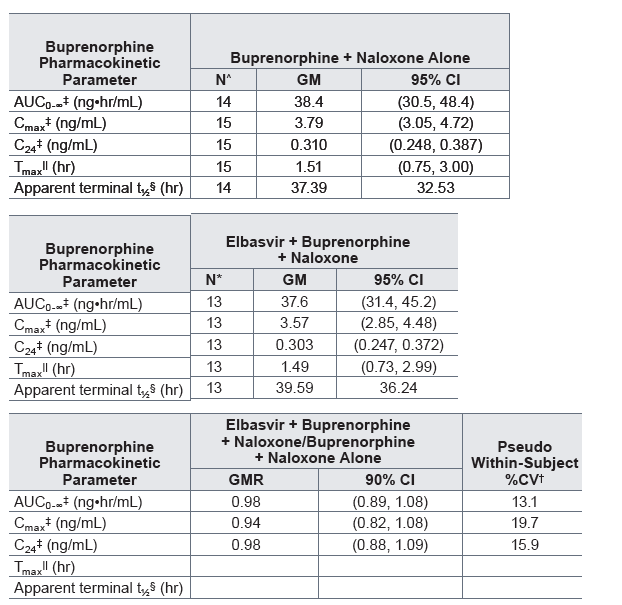
Buprenorphine + naloxone alone: A single sublingual dose of 8 mg buprenorphine/2 mg naloxone (with naltrexone blockade administered as 50 mg naltrexone HCl q12h starting 14 hours prior to the buprenorphine/naloxone dose, for a total of 3 naltrexone doses).
Elbasvir + buprenorphine + naloxone: A single oral dose of 50 mg elbasvir coadministered with a single sublingual dose of 8 mg buprenorphine/2 mg naloxone (with naltrexone blockade administered as 50 mg naltrexone HCl q12h starting 14 hours prior to the buprenorphine/naloxone dose, for a total of 3 naltrexone doses).
One subject was discontinued by the Investigator on Day 1 of Period 1 (buprenorphine +naloxone alone) due to vomiting within 3 hours of dosing.
* Two subjects were discontinued from the study by the Investigator on Day 1 of Period 3 (elbasvir + buprenorphine + naloxone) due to vomiting within 8 hours of dosing.
Pseudo within-subject %CV = 100*sqrt((σA2 + σB2 - 2σAB)/2), where σA2 and σB2 are the estimated variances on the log scale for the 2 treatments and σAB is the corresponding estimated covariance, each obtained from the linear mixed-effects model.
Back-transformed least-squares mean and confidence interval from the linear mixed-effects model performed on natural log-transformed values.
Median (minimum, maximum) reported for Tmax.
Geometric mean and geometric coefficient of variation reported for apparent terminal t.
GM = Geometric least-squares mean; CI = Confidence interval; GMR = Geometric least-squares mean ratio; CV = Coefficient of variation.
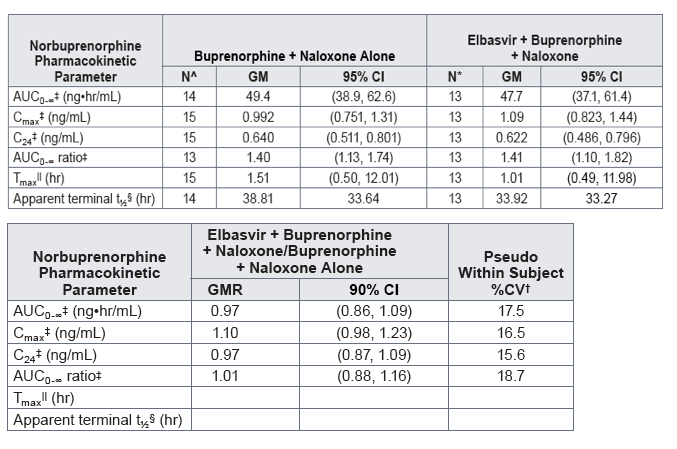
NOR exhibited similar PK following single-dose administration of BUP/NAL with or without coadministration of EBR (Figure 3, Table 4)
- GMRs [90% CIs] for the NOR AUC0-∞, Cmax, C24, and the molecular weight-adjusted parent/metabolite AUC0-∞ ratio were 0.97 [0.86, 1.09], 1.10 [0.98, 1.23], 0.97 [0.87, 1.09], and 1.01 [0.88, 1.16], respectively
- Relative to when BUP + NAL was administered alone, coadministration with EBR resulted in a median Tmax of NOR that occurred approximately 0.5 hours earlier, ie, 1.01 hours postdose compared to 1.51 hours postdose
- The GM apparent terminal t1/2 of NOR was comparable following EBR + BUP + NAL (33.92 hours) and following BUP + NAL alone (38.81 hours)
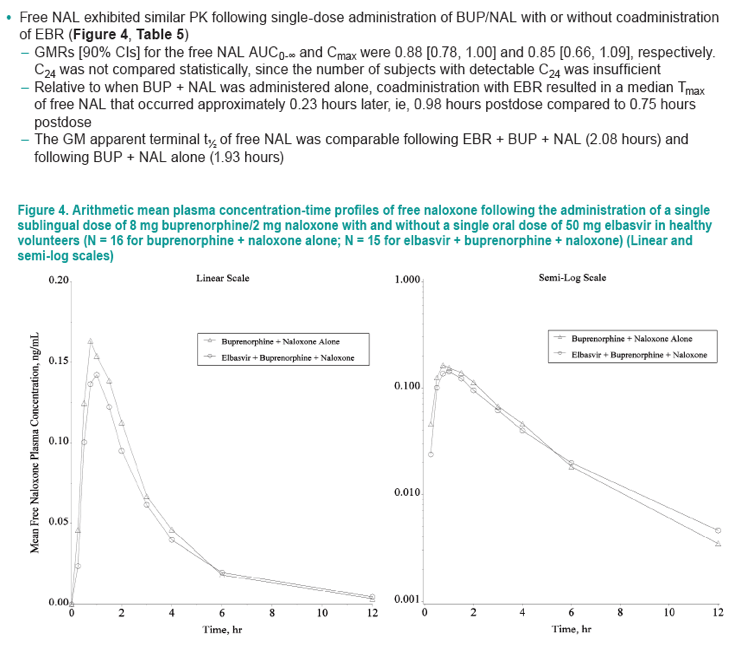
Figure 3. Arithmetic mean plasma concentration-time profiles of norbuprenorphine following the administration of a single sublingual dose of 8 mg buprenorphine/2 mg naloxone with and without a single oral dose of 50 mg elbasvir in healthy volunteers (N = 16 for buprenorphine + naloxone alone; N = 15 for elbasvir + buprenorphine + naloxone) (Linear and semi-log scales)
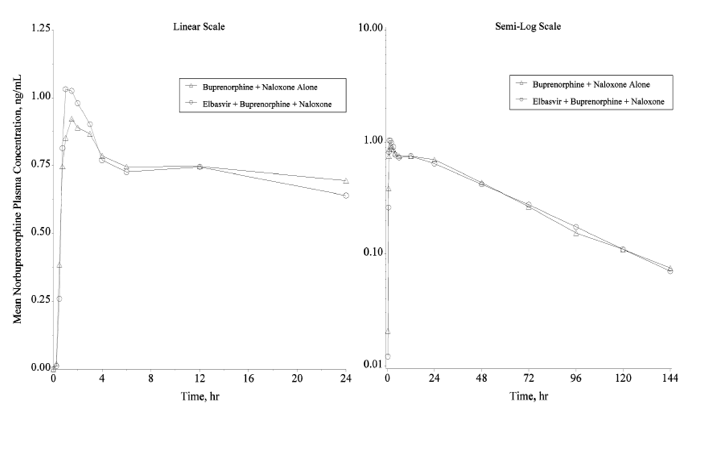
Table 4. Statistical comparison and summary statistics of norbuprenorphine plasma pharmacokinetics following the administration of a single sublingual dose of 8 mg buprenorphine/2 mg naloxone with and without a single oral dose of 50 mg elbasvir in healthy volunteers
Buprenorphine + naloxone alone: A single sublingual dose of 8 mg buprenorphine/2 mg naloxone (with naltrexone blockade administered as 50 mg naltrexone HCl q12h starting 14 hours prior to the buprenorphine/naloxone dose, for a total of 3 naltrexone doses).
Elbasvir + buprenorphine + naloxone: A single oral dose of 50 mg elbasvir coadministered with a single sublingual dose of 8 mg buprenorphine/2 mg naloxone (with naltrexone blockade administered as 50 mg naltrexone HCl q12h starting 14 hours prior to the buprenorphine/naloxone dose, for a total of 3 naltrexone doses).
^One subject was discontinued by the Investigator on Day 1 of Period 1 (buprenorphine +naloxone alone) due to vomiting within 3 hours of dosing.
*Two subjects were discontinued from the study by the Investigator on Day 1 of Period 3 (elbasvir + buprenorphine + naloxone) due to vomiting within 8 hours of dosing.
Pseudo within-subject %CV = 100*sqrt((σA2 + σB2 - 2σAB)/2), where σA2 and σB2 are the estimated variances on the log scale for the 2 treatments and σAB is the corresponding estimated covariance, each obtained from the linear mixed-effects model.
Back-transformed least-squares mean and confidence interval from the linear mixed-effects model performed on natural log-transformed values.
Median (minimum, maximum) reported for Tmax.
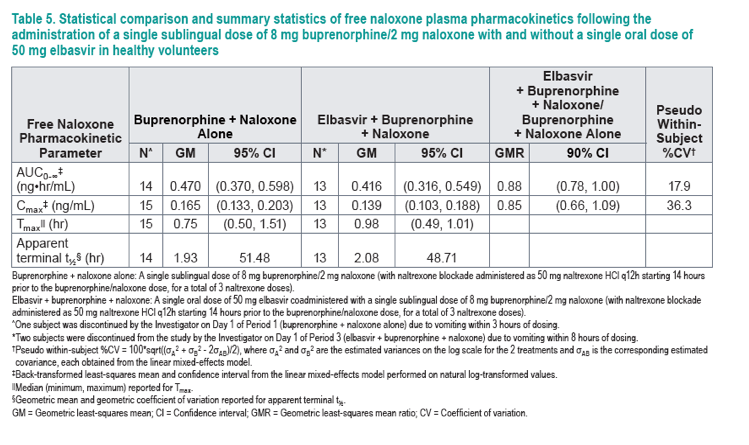
Geometric mean and geometric coefficient of variation reported for apparent terminal t.
GM = Geometric least-squares mean; CI = Confidence interval; GMR = Geometric least-squares mean ratio; CV = Coefficient of variation;
AUC0-∞ ratio = Molecular weight-adjusted AUC0-∞ ratio of
norbuprenorphine/buprenorphine.
|
| |
|
 |
 |
|
|