 |
 |
 |
| |
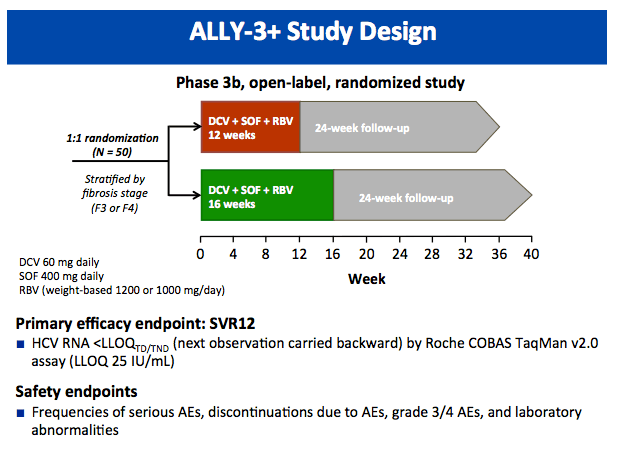
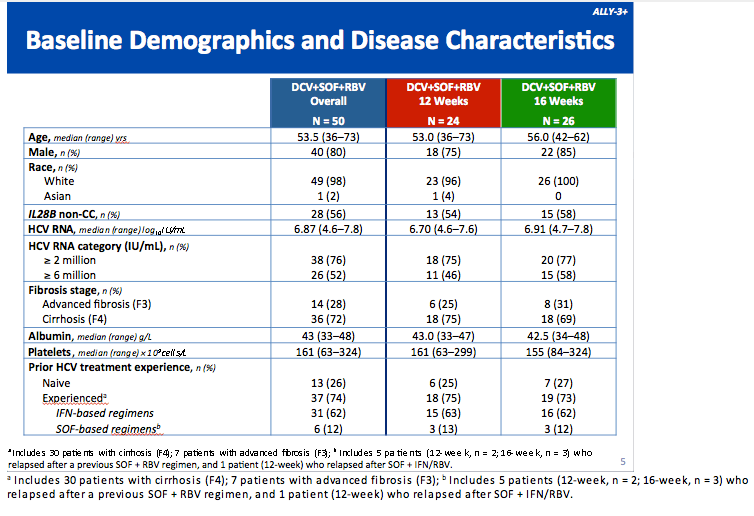
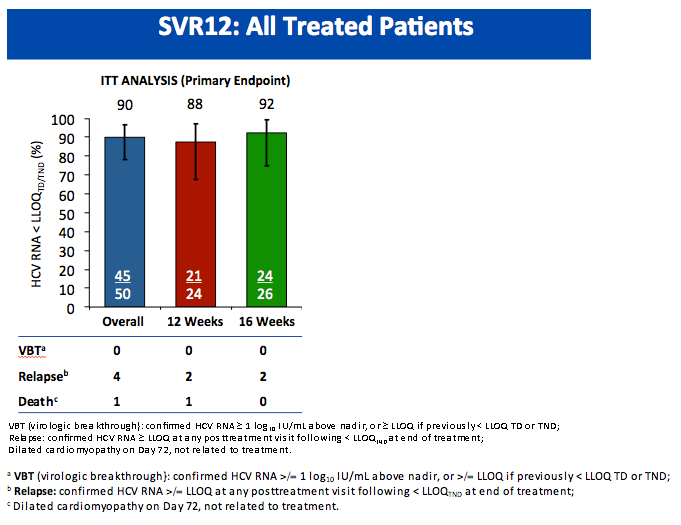
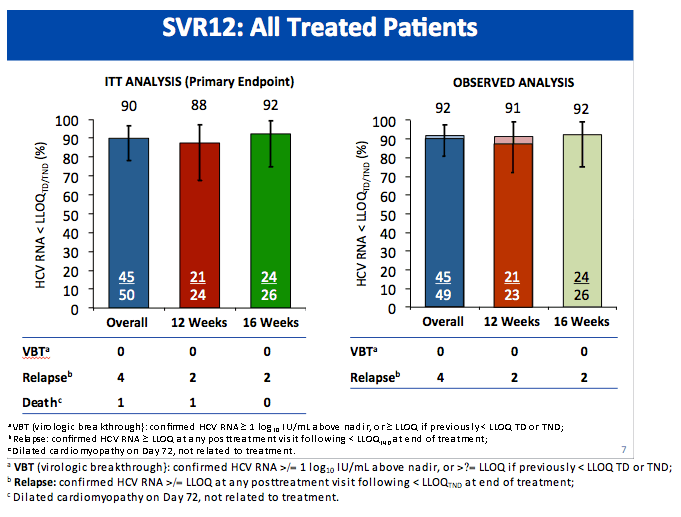
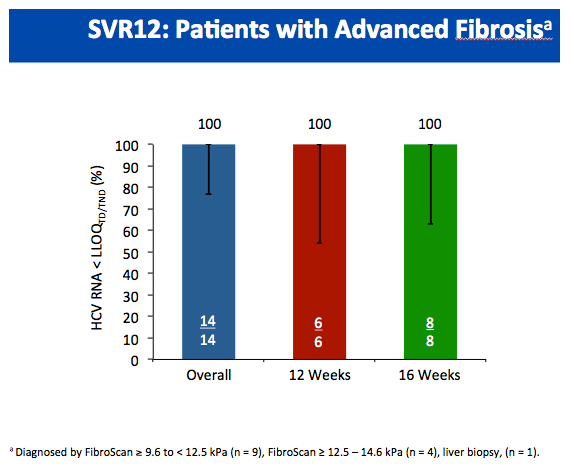
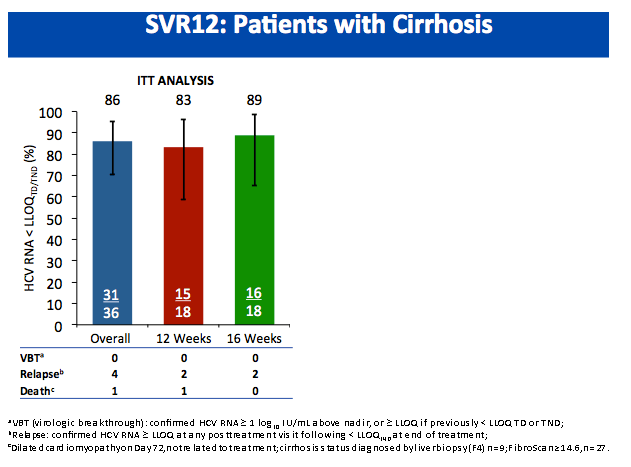
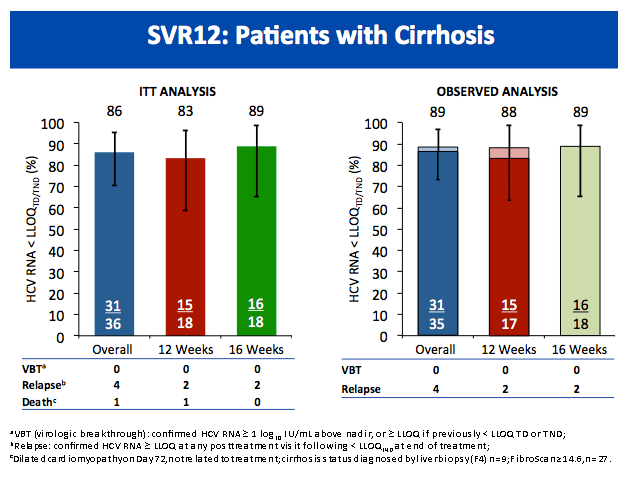
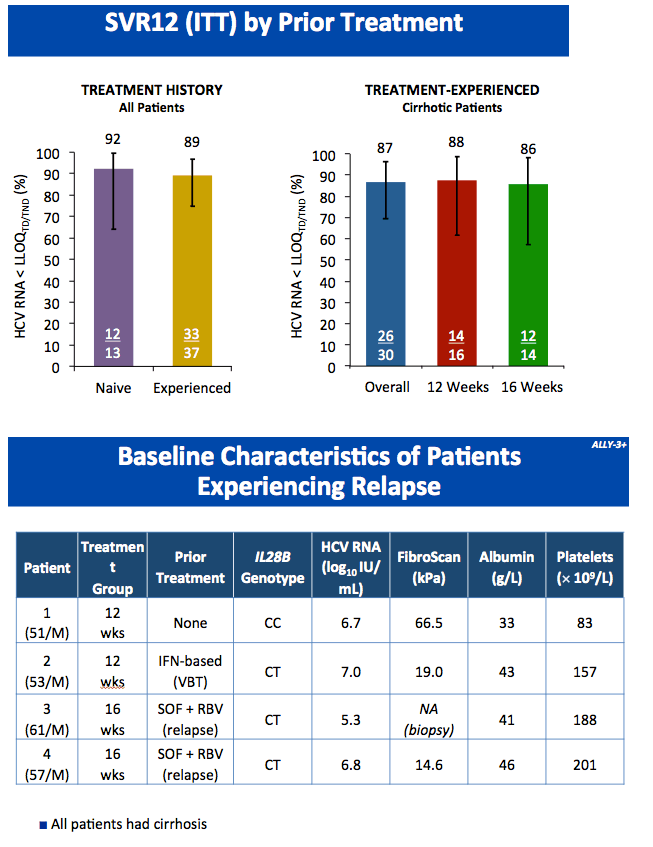
All-Oral Treatment With Daclatasvir Plus Sofosbuvir Plus Ribavirin for 12 or 16 Weeks in HCV Genotype 3-Infected Patients With Advanced Fibrosis or Cirrhosis: The ALLY-3+ Phase 3 Study
|
| |
| |
Reported by Jules Levin
AASLD 2015 Nov 13-17 San Francisco
Leroy V,1 Angus P,2 Bronowicki JP,3 Dore G,4 Hézode C,5 Pianko S,6 Pol S,7
Stuart K,8 Tse E,9 McPhee F,10 Bhore R,11 Jimenez-Exposito MJ,11 Thompson A4
1CHU de Grenoble, La Tronche, France; 2Austin Hospital, Heidelberg, Australia; 3CHU Nancy & Lorraine University, Nancy, France; 4St. Vincent's Hospital and Kirby Institute, Sydney, Australia; 5CHU Henri Mondor, Créteil, France; 6Monash Medical Centre, Clayton, Australia; 7Hôpital Cochin, Paris, France; 8Gallipoli Medical Research Foundation, Greenslopes, Australia; 9Royal Adelaide Hospital, Adelaide, Australia; 10Bristol-Myers Squibb Research & Development, Wallingford, CT; 11Bristol-Myers Squibb Research & Development, Princeton, NJ.



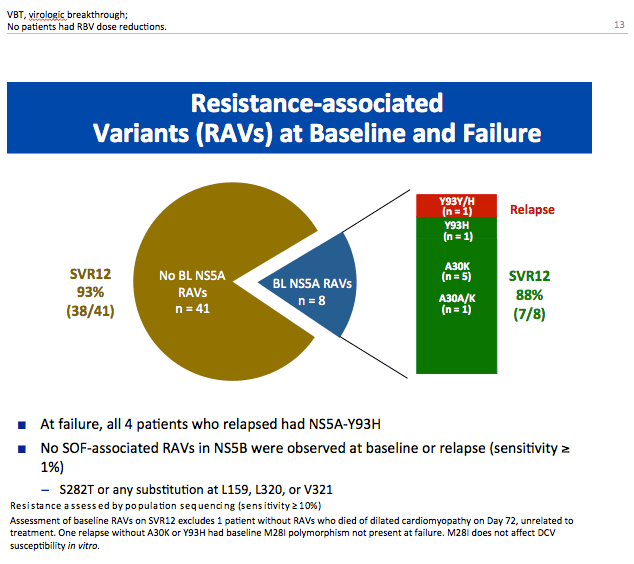
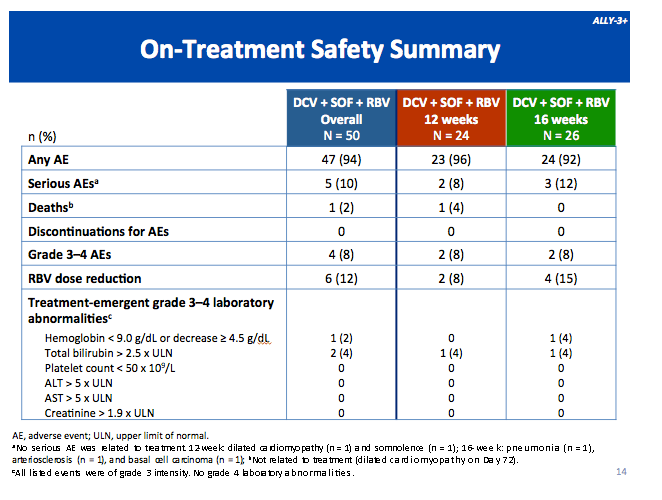
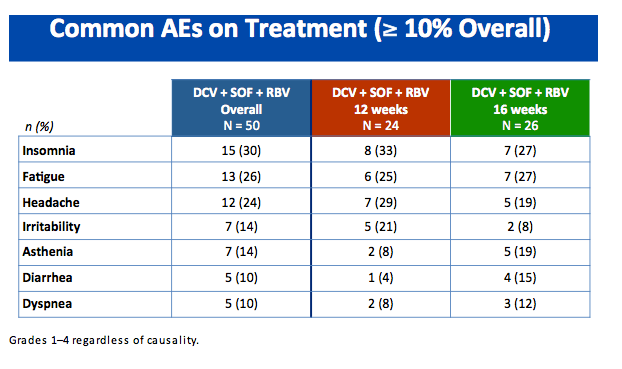
|
| |
|
 |
 |
|
|