 |
 |
 |
| |
Efficacy of Elbasvir (EBR) and Grazoprevir (GZR) in Black HCV-Infected
Patients: Results of a Pooled Analysis of Phase 2/3 Studies
|
| |
| |
Reported by Jules Levin
AASLD 2015 Nov 13-17 San Francisco
Zamor P1; McCone J2; Vierling J3; Ghalib R4; Luketic V5; Pearlman B6;
Ravendhran N7; Balart L8; Robertson M9; Hwang P9; Nguyen B-Y9; Wahl J9;
Barr E9; Talwani R9
1Carolinas Medical Center, Charlotte, NC, USA; 2Mount Vernon Endoscopy and
Liver Center, Alexandria, VA, USA; 3Baylor St. Luke's Medical Center, Houston, TX, USA; 4Texas Clinical Research Institute, Dallas, TX, USA; 5Virginia Commonwealth University, Richmond, VA, USA; 6Atlanta Medical Center, Atlanta, GA, USA;
7Digestive Disease Associates, Baltimore, MD, USA; 8Tulane University School of Medicine, New Orleans, LA, USA; 9Merck & Co., Inc., Kenilworth, NJ, USA.
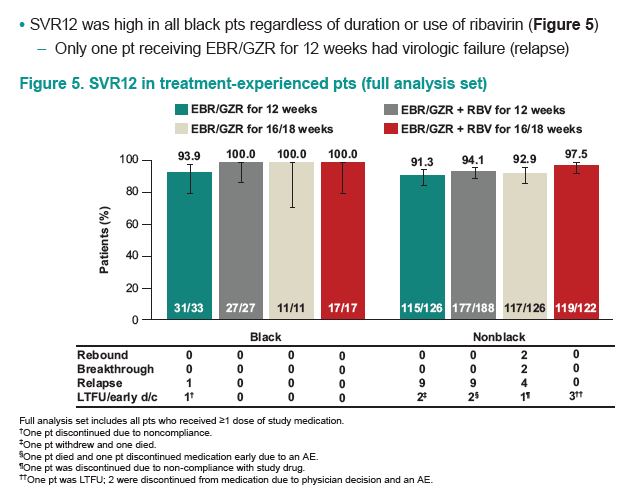
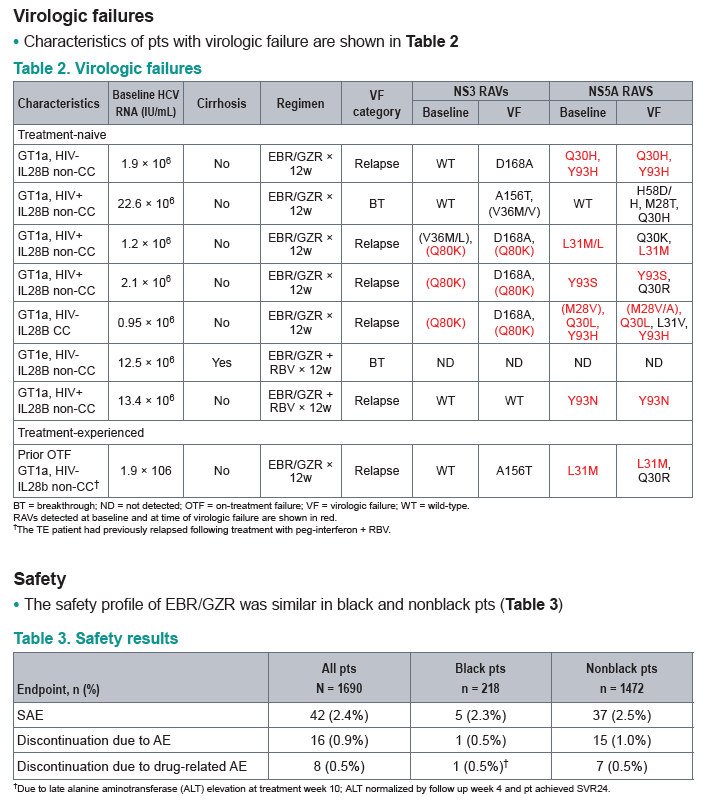
AASLD: The Combination of Elbasvir and Grazoprevir ± RBV Is Highly Effective for the Treatment of GT1a-Infected Patients - (11/17/15)




|
| |
|
 |
 |
|
|