 |
 |
 |
| |
Neurocognitive Decline is Associated with Antiretroviral Concentrations in Cerebral Spinal Fluid and Blood
|
| |
| |
Reported by Jules Levin
CROI 2015 Feb 23-26, Seattle, WA
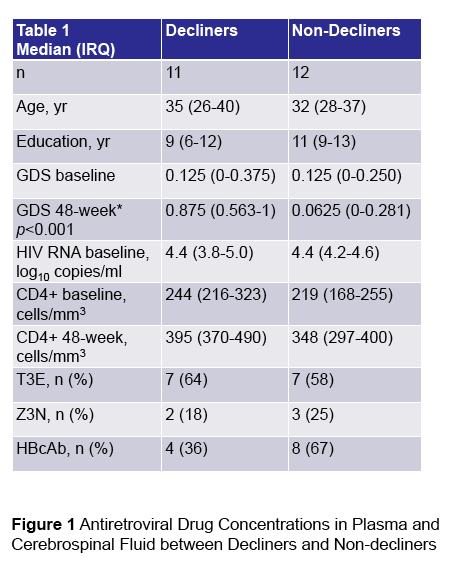
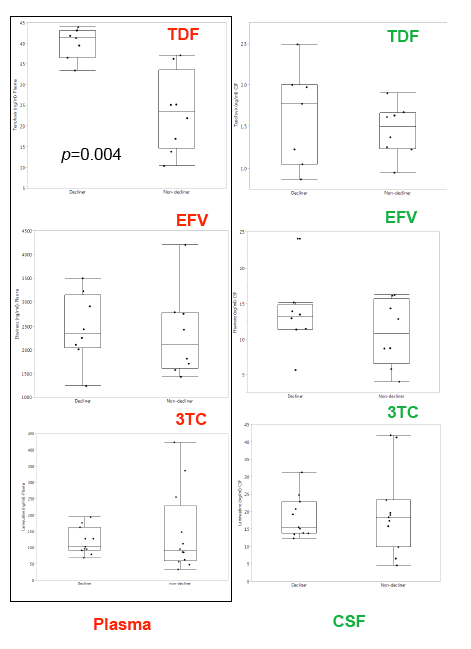
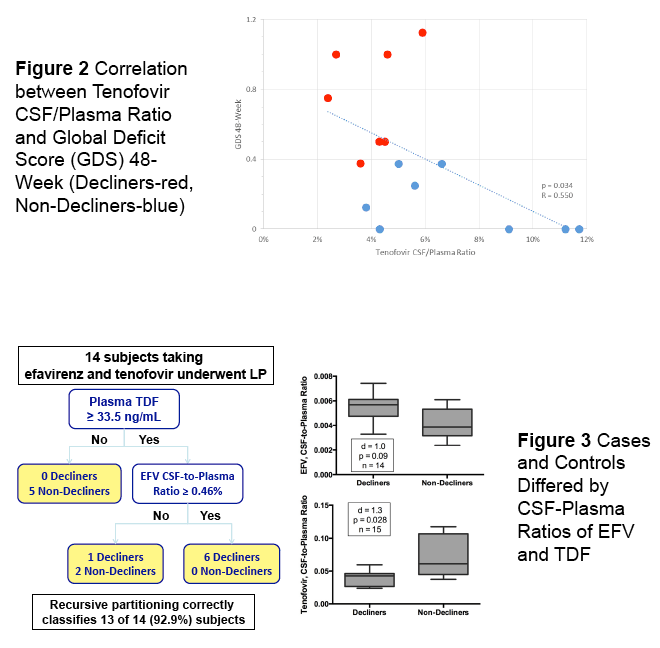
In the poster which is the subject of this report (below) the study finds CSF drug concentrations associated with neurocognitive decline, looking at EFV & TDF in the main study "Randomized Clinical Trial of Antiretroviral Therapy for Prevention of HAND" presented in an oral talk at CROI by Scott Lentendre [http://www.croiwebcasts.org/console/player/25628?mediaType=slideVideo&] reported on a trial in China in ART treatment-naives: "To date, clinical trials have focused on treatment of existing NCI and have been largely inconclusive. We conducted a clinical trial to determine whether one ART regimen would better prevent NCI (neurocognitive impairment) than another". ...."250 HIV+, ART-naive adults were randomized to either open-label zidovudine-lamivudine-nevirapine (ZLN) or tenofovir-lamivudine-efavirenz (TLE) at two hospitals in Beijing. All subjects were NC normal and had CD4+ T-cell counts below 350/mm3 at entry"....."GDS (global deficit scores) values at entry were also similar.....A high proportion of subjects attained virologic suppression: 92% had plasma HIV RNA below 50 c/mL in both groups at 48 weeks"....."TLE (EFV regimen) was associated with greater risk of incident NCI over 96 weeks than ZLN"....authors concluded: "Possible explanations include differences between the regimens in drug distribution into the CNS and neurotoxicity. The substantial difference in other adverse events dictates additional research to identify a less toxic alternative to ZLN."
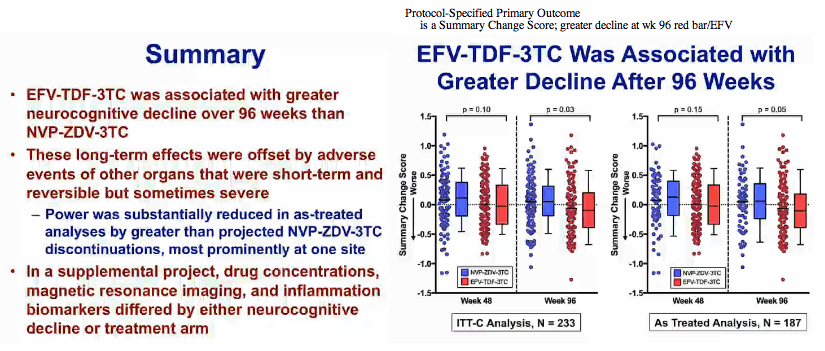
Protocol-Specified Primary Outcome
is a Summary Change Score; greater decline at wk 96 red bar/EFV
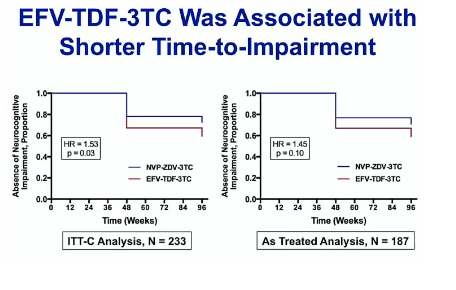
Secondary Outcome-Global Neurocognitive Impairment
people who took EFV had a 53% increased hazard of progressing to impairment over the period of thetrial

Reported by Jules Levin
CROI 2015 Feb 23-26, Seattle, WA
Ma Q1,3, Liu X2, Heaton R1, Zhang F2, Jin H1, Wu H2, Crescini M1, Zhao H2, Zeng H2, Letendre S1 and the China neuroAIDS Team
1University of California San Diego, San Diego, USA; 2. Chinese Center for Disease Control and Prevention, Beijing, China; 3. University at Buffalo, Buffalo, NY, USA
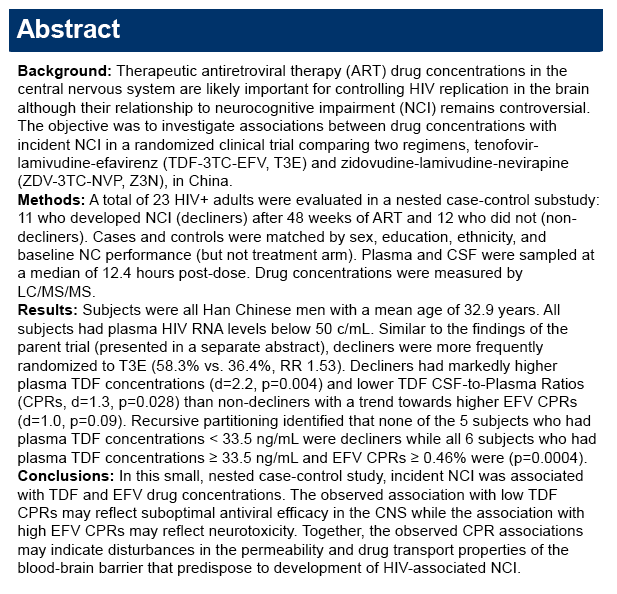


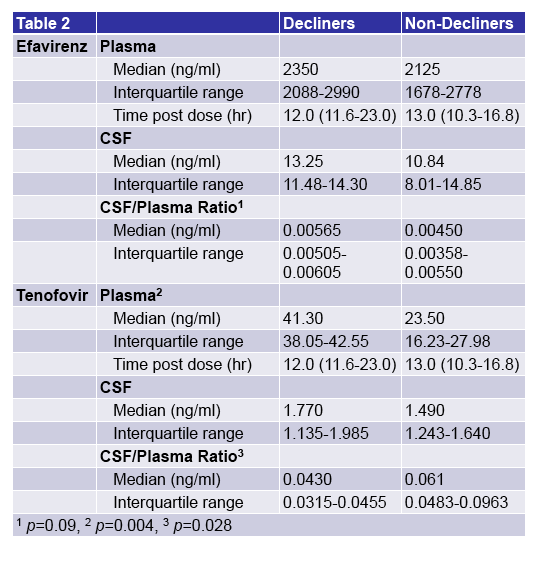

|
| |
|
 |
 |
|
|