 |
 |
 |
| |
Drug-Drug Interaction Between HCV Inhibitors Grazoprevir/Elbasvir With Dolutegravir
|
| |
| |
Reported by Jules Levin
CROI 2015 Feb 23-26, Seattle, WA
Wendy W. Yeh1; Ted Marenco2; Hwa-Ping Feng1; Zifang Guo1; Daria Stypinski2; Lisa Ross3; Ivy Song4; Patricia Jumes1; Charles Tomek2; Joan R. Butterton1
1Merck & Co., Inc., Kenilworth, NJ, USA; 2Celerion, Lincoln, NE, USA; 3ViiV Healthcare, Research Triangle Park, NC, USA; 4GlaxoSmithKline, Research Triangle Park, NC, USA
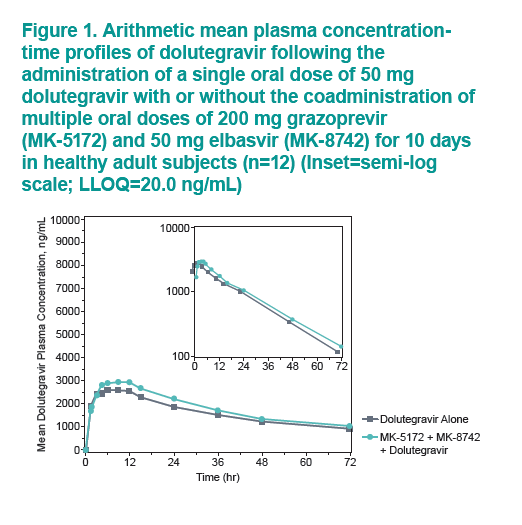
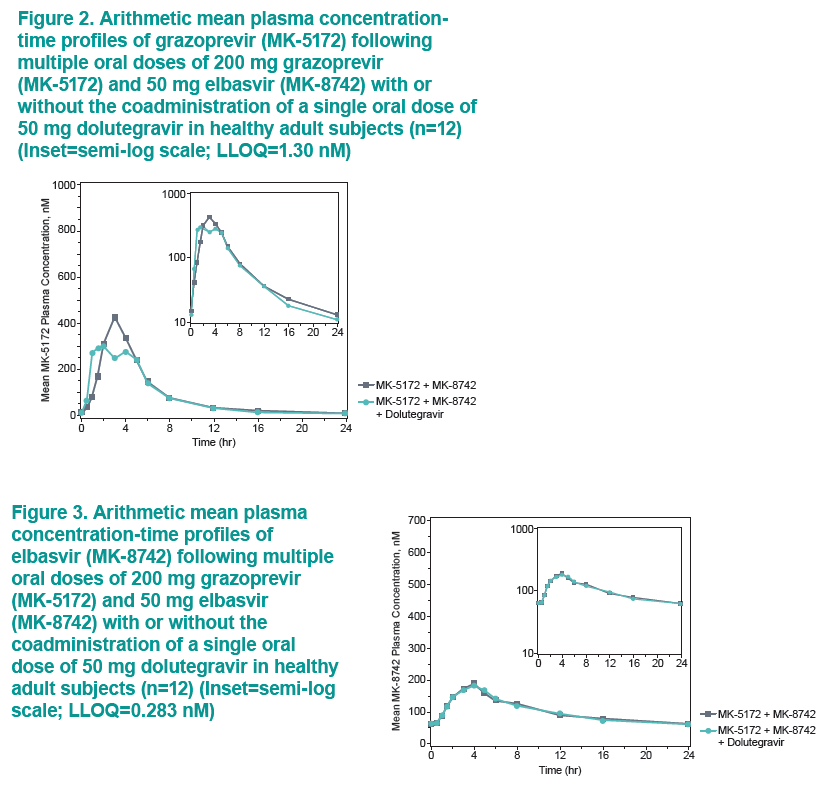
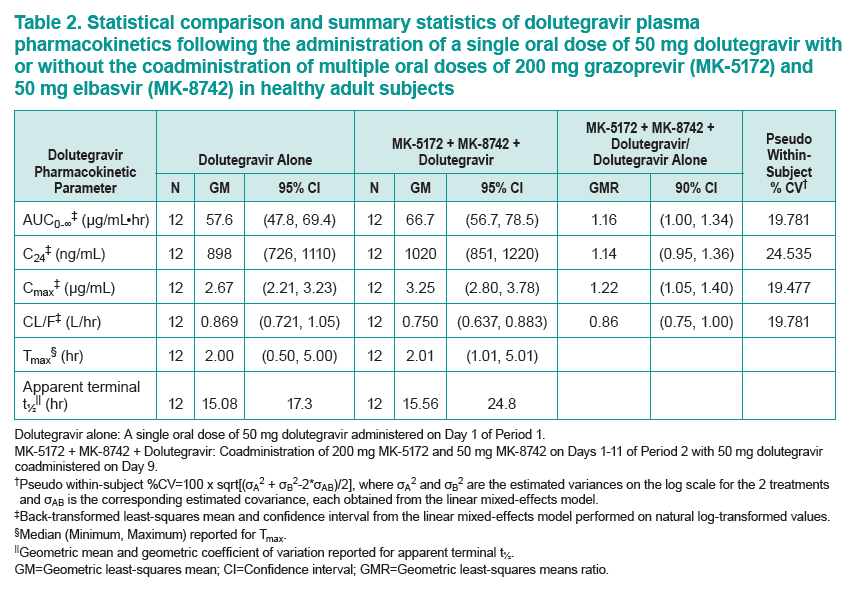
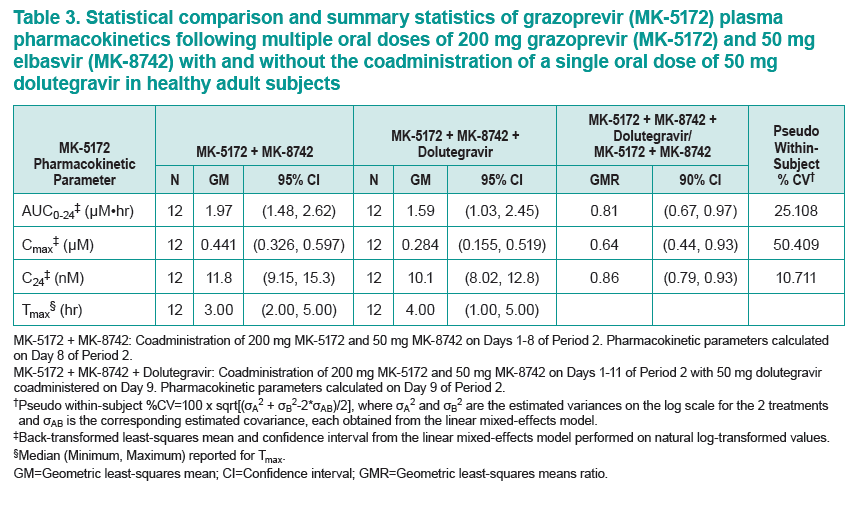
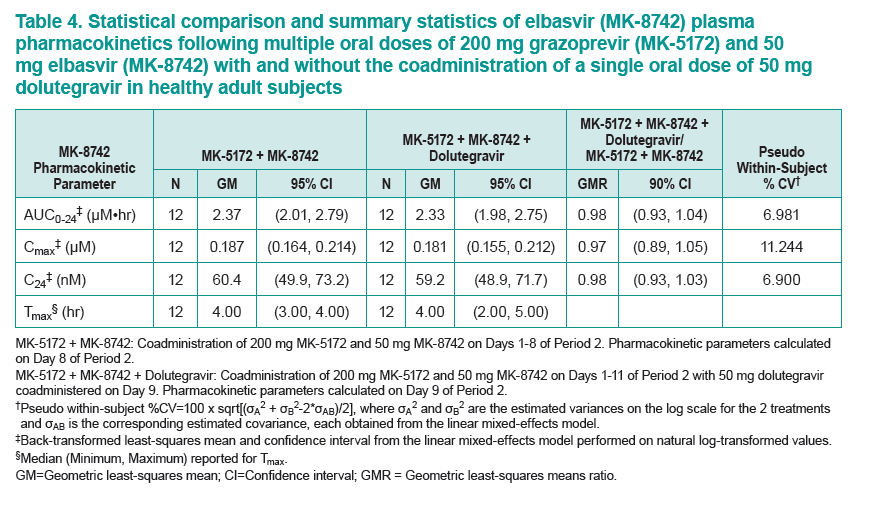
....."Grazoprevir (GZR, MK-5172), a potent once-daily inhibitor of the hepatitis C virus (HCV) NS3/4A protease, and elbasvir (EBR, MK-8742), a potent once-daily inhibitor of the HCV NS5A replication complex. are being developed as a fixed-dose combination therapy for the treatment of chronic HCV infection in monoinfected and HCV/human immunodeficiency virus (HIV)-coinfected patients. This study evaluated the safety and pharmacokinetic (PK) interactions of GZR and EBR when coadministered with dolutegravir (DTG), an HIV-1 integrase strand transfer inhibitor, in healthy volunteers......Coadministration of GZR + EBR with DTG had no clinically meaningful effect on the PK of DTG or EBR. Coadministration of DTG with GZR + EBR decreased GZR exposure. However, the decreases in GZR exposure are within the therapeutic window currently defined for GZR based on available clinical data. These results suggest that no dose adjustments in GZR, EBR, or DTG are needed for coadministration in HCV/HIV-coinfected patients.....DTG did result in decreased GZR exposure, with AUC0-24, Cmax, and C24 GMRs [90% CIs] of 0.81 [0.67, 0.97], 0.64 [0.44, 0.93], and 0.86 [0.79, 0.93], respectively"

from Jules: Merck acquired Idenix & their HCV nukes [http://www.natap.org/2014/HCV/060914_01.htm]....."company currently has three HCV drug candidates in clinical development: two nucleotide prodrugs (IDX21437 and IDX21459) and a NS5A inhibitor (samatasvir). These novel candidates are being evaluated for their potential inclusion in the development of all oral, pan-genotypic fixed-dose combination regimens."







|
| |
|
 |
 |
|
|