 |
 |
 |
| |
TAF easier on kidney and bone signals than TDF in 48-week trials
|
| |
| |
CROI 2015, February 23-26, 2015, Seattle, Washington
Mark Mascolini
Tenofovir alafenamide (TAF) appeared to fulfill its promise of greater kidney and bone safety than tenofovir disoproxil fumarate (TDF) in pooled results of two 48-week randomized double-blind trials [1]. Clinicians in the CROI hall where Paul Sax spelled out these findings voiced hopeful concern that the 1-year differences in renal and bone signals would translate into fewer cases of chronic kidney disease or bone loss with TAF than with TDF.
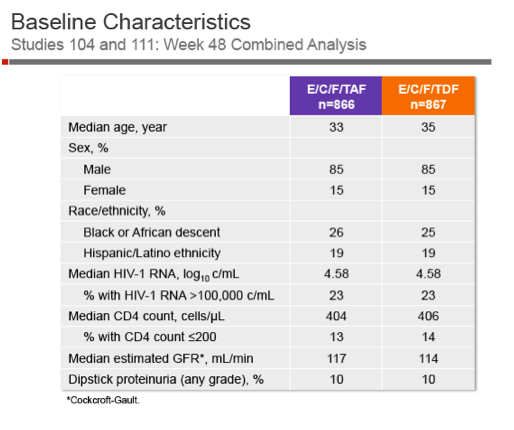
Two identical phase 3 trials at sites around the world randomized 1733 antiretroviral-naive adults to TAF or TDF, each as part of a once-daily formulation also containing elvitegravir, cobicistat, and emtricitabine. The researchers planned from the beginning to pool results of these double-blind, double-dummy trials for this 48-week analysis. All study participants had an estimated glomerular filtration rate (eGFR) at or above 50 mL/min, and none had hepatitis B or C. An earlier CROI report summarized separately by NATAP established the virologic noninferiority of the TAF formulation to the TDF formulation [2]. Prespecified safety endpoints were serum creatinine, proteinuria, and hip and spine bone mineral density (BMD).
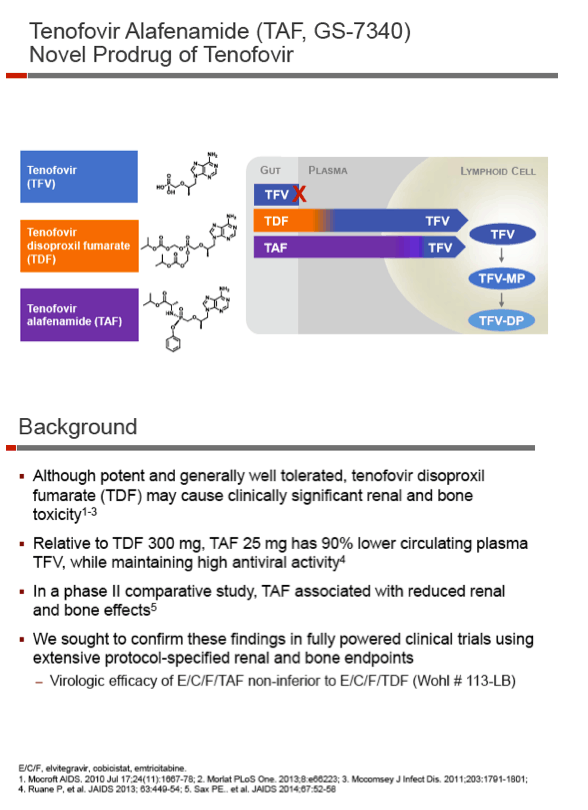
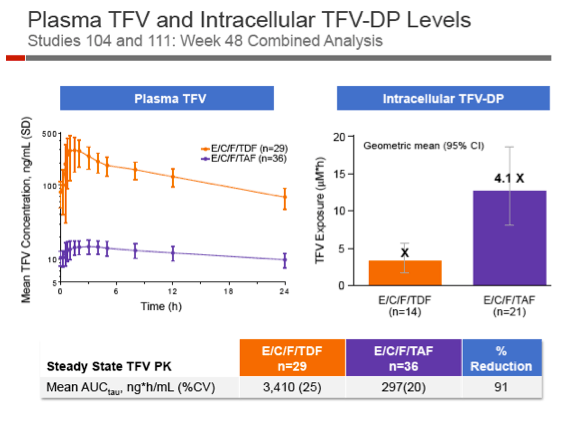
Study participants had a median age around 34, 85% were men, about 25% were black, and 19% were Hispanic. Median pretreatment CD4 count stood around 405 and median pretreatment viral load at 4.58 log (about 38,000 copies). Tenofovir in plasma--where it can cause trouble for patients--was 91% lower with TAF than TDF. But tenofovir in cells--where it can cause trouble for HIVŃwas 4 times higher with TAF.
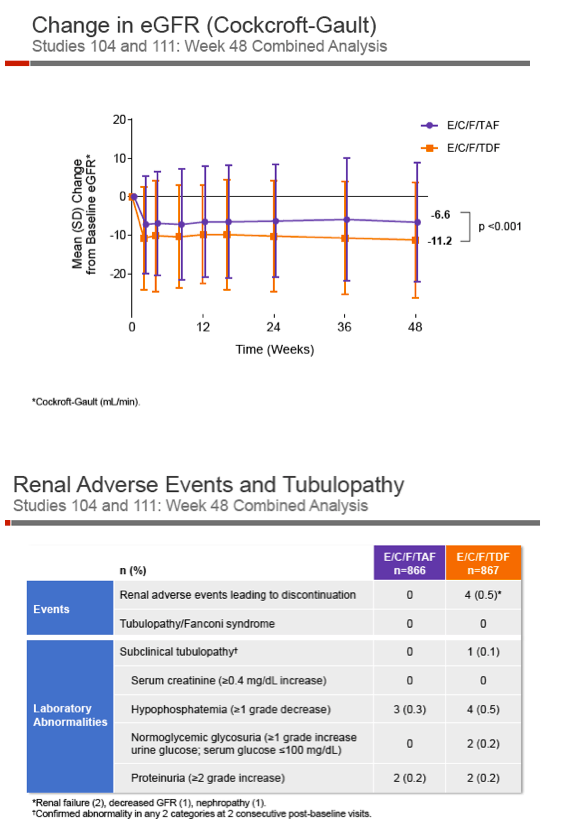
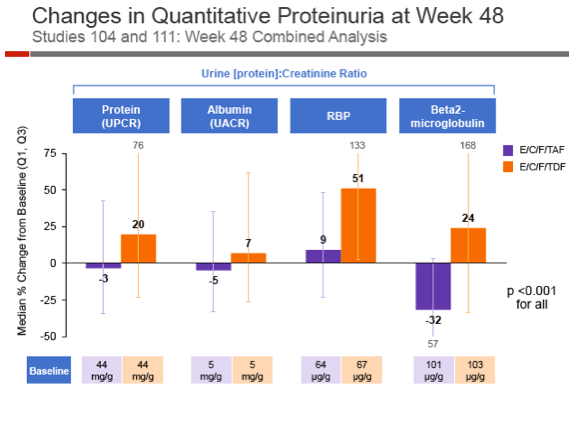
At week 48 serum creatinine rose an average 0.08 mg/dL with TAF versus 0.11 mg/d with TDF, a significant difference (P < 0.001). eGFR was fell by an average 6.6 mL/min with TAF versus 11.2 mL/min with TDF (P < 0.001). No one taking TAF and 4 people taking TDF (0.5%) left the study because of renal adverse events (2 renal failures, 1 decreased eGFR, 1 nephropathy). Three people in the TAF arm (0.3%) and 4 in the TDF arm (0.5%) had hypophosphatemia, and 2 people in each arm (0.2%) had a grade 2 or greater increase in proteinuria. Proximal tubulopathy developed in no one. Measures of urine protein, albumin, and beta2-microglobulin through 48 weeks all favored the TAF arm (P < 0.001 for all).
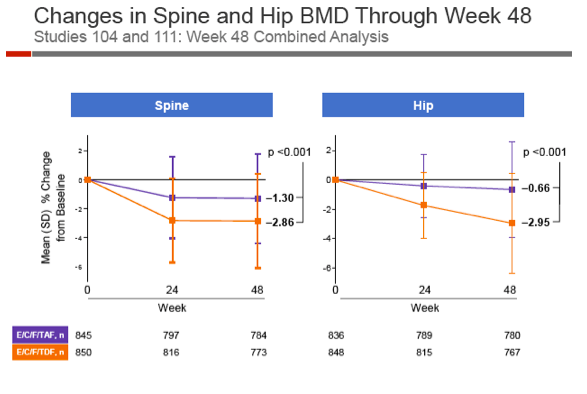
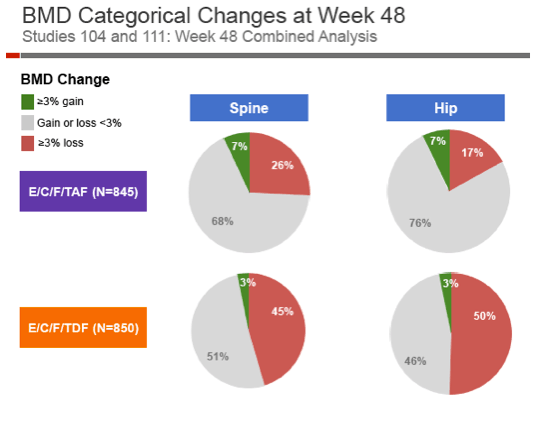
After 48 weeks of treatment, hip BMD fell slightly with TAF (-0.66%) while dropping 2.95% with TDF (P < 0.001). Spine BMD fell more than twice as much with TDF than TAF (-2.86% versus -1.30%, P < 0.001). Spine BMD dropped 3% or more in 45% on TDF versus 26% on TAF. Hip bone density fell 3% or more in 50% on TDF versus 17% on TAF. Levels of parathyroid hormone and two bone turnover markers--C-telopeptide and P1NP--and rose significantly less with TAF than TDF.
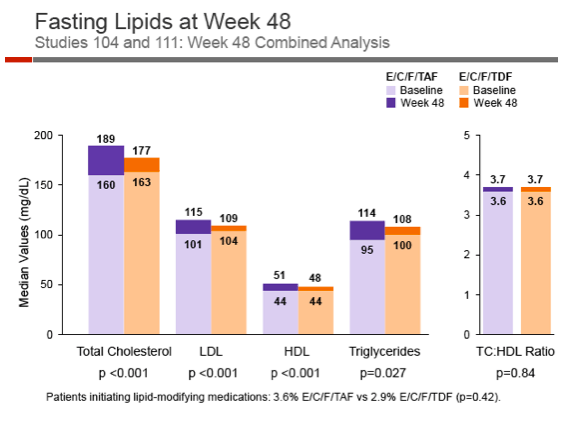
Fasting lipids (triglycerides and total, LDL, and HDL cholesterol) all rose significantly more in the TAF arm than the TDF arm. But total-to-HDL ratio changed hardly at all in either arm.
Referring to all renal and bone differences between the drugs, Sax and colleagues proposed that "the most likely explanation for these findings is the 90% lower tenofovir plasma exposure with TAF versus TDF."
References
1. Sax PE, Saag MS, Yin MT, et al. Renal and bone safety of tenofovir alafenamide vs tenofovir disoproxil fumarate. CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 143LB.
2. Wohl D, Pozniak A, Thompson M, et al. Tenofovir alafenamide (TAF) in a single-tablet regimen in initial HIV-1 therapy. CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 113LB.


|
| |
|
 |
 |
|
|