| |
Isentress FDA Product Label - Drug Interactions with ARTs
|
| |
| |
Download PDF here
HARNESS study: ritonavir-boosted atazanavir
(ATV/r)+raltegravir (RAL) switch study in virologically suppressed, HIV-1-infected patients......http://www.natap.org/2014/IAC/IAC_42.htm

ISENTRESS can be administered with or without food (2.1).
Effect of Food on Oral Absorption
ISENTRESS may be administered with or without food. Raltegravir was administered without regard to food in the pivotal safety and efficacy studies in HIV-1-infected patients. The effect of consumption of low-, moderate- and high-fat meals on steady-state raltegravir pharmacokinetics was assessed in healthy volunteers administered the 400 mg film-coated tablet. Administration of multiple doses of raltegravir following a moderate-fat meal (600 Kcal, 21 g fat) did not affect raltegravir AUC to a clinically meaningful13 degree with an increase of 13% relative to fasting. Raltegravir C12hr was 66% higher and Cmax was 5% higher following a moderate-fat meal compared to fasting. Administration of raltegravir following a high-fat meal (825 Kcal, 52 g fat) increased AUC and Cmax by approximately 2-fold and increased C12hr by 4.1-fold. Administration of raltegravir following a low-fat meal (300 Kcal, 2.5 g fat) decreased AUC and Cmax by 46% and 52%, respectively; C12hr was essentially unchanged. Food appears to increase pharmacokinetic variability relative to fasting.
Administration of the chewable tablet with a high fat meal led to an average 6% decrease in AUC, 62% decrease in Cmax, and 188% increase in C12hr compared to administration in the fasted state. Administration of the chewable tablet with a high fat meal does not affect raltegravir pharmacokinetics to a clinically meaningful degree and the chewable tablet can be administered without regard to food. The effect of food on the formulation for oral suspension was not studied.
7 DRUG INTERACTIONS
7.1 Effect of Raltegravir on the Pharmacokinetics of Other Agents
Raltegravir does not inhibit (IC50>100 μM) CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6 or CYP3A in vitro. Moreover, in vitro, raltegravir did not induce CYP1A2, CYP2B6 or CYP3A4. A midazolam drug interaction study confirmed the low propensity of raltegravir to alter the pharmacokinetics of agents metabolized by CYP3A4 in vivo by demonstrating a lack of effect of raltegravir on the pharmacokinetics of midazolam, a sensitive CYP3A4 substrate. Similarly, raltegravir is not an inhibitor (IC50>50 μM) of UGT1A1 or UGT2B7, and raltegravir does not inhibit P-glycoprotein-mediated transport. Based on these data, ISENTRESS is not expected to affect the pharmacokinetics of drugs that are substrates of these enzymes or P-glycoprotein (e.g., protease inhibitors, NNRTIs, opioid analgesics, statins, azole antifungals, proton pump inhibitors and anti-erectile dysfunction agents).
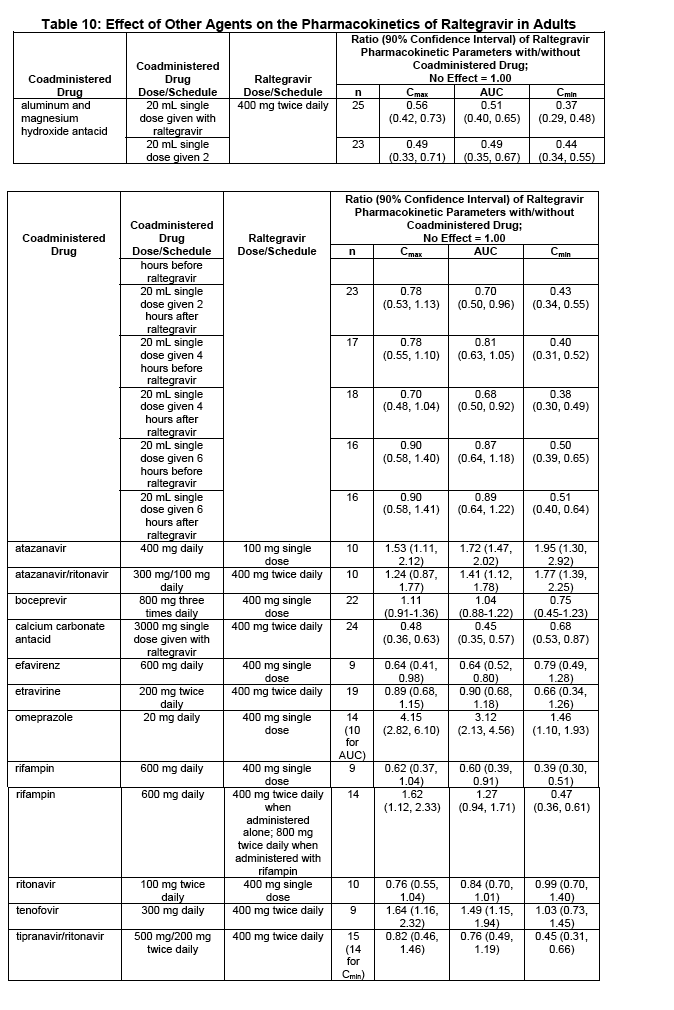
7.2 Effect of Other Agents on the Pharmacokinetics of Raltegravir
Raltegravir is not a substrate of cytochrome P450 (CYP) enzymes. Based on in vivo and in vitro studies, raltegravir is eliminated mainly by metabolism via a UGT1A1-mediated glucuronidation pathway. Coadministration of ISENTRESS with drugs that inhibit UGT1A1 may increase plasma levels of raltegravir and coadministration of ISENTRESS with drugs that induce UGT1A1, such as rifampin, may reduce plasma levels of raltegravir. The impact of other inducers of drug metabolizing enzymes, such as phenytoin and phenobarbital, on
UGT1A1 is unknown.
Selected drug interactions are presented in Table 8 [see Clinical Pharmacology (12.3)].
7.3 Drugs without Clinically Significant Interactions with ISENTRESS
In drug interaction studies, raltegravir did not have a clinically meaningful effect on the
pharmacokinetics of the following: hormonal contraceptives, methadone, lamivudine, tenofovir, etravirine,
darunavir/ritonavir, or boceprevir. Moreover, atazanavir, atazanavir/ritonavir, boceprevir, calcium
carbonate antacids, darunavir/ritonavir, efavirenz, etravirine, omeprazole, or tipranavir/ritonavir did not
have a clinically meaningful effect on the pharmacokinetics of raltegravir. No dose adjustment is required
when ISENTRESS is coadministered with these drugs.
|
|
| |
| |
|
|
|