 |
 |
 |
| |
Frequent Fibrosis Regression With SVR in HCV/HIV+, Cutting Death Risk
|
| |
| |
Webcast:
https://www.youtube.com/watch?v=pnHtjW_zFtA
IAS 2015, July 19-22, 2015, Vancouver
from Jules: State & federal govts are imposing death sentences on patients by imposing restrictions to access to HCV therapy.
all the patients in this study appeared to have at least relatively advanced disease at baseline and had HCV for over 20 years, with relatively low Cd4 counts. Patients with less advanced HCV disease were more likely to achieve SVR & of course patients with an SVR were more likely to achieve fibrosis regression: 55% of patients with SVR achieved fibrosis regression while only 15% without SVR achieved fibrosis regression, but of note 45% of patients with an SVR did not achieve fibrosis regression; delaying HCV therapy decreases chance for fibrosis regression & early HCV treatment increases chance for regression. Noteworthy study outcomes: Treat HCV EARLY, once a patient has advanced liver disease they face increased risk for disease & death because they were less likely to achieve fibrosis regression. Even patients who achieve fibrosis regression risk liver disease but in this study those rates were low but a few patients who achieved SVR & FR still died or had liver complications. The current restrictions by medicaids are supported by the federal govt because the federal govt is not doing anything to prevent the states from imposing these restrictions, so the federal govt is imposing increased death risk to patients. Of note ALL patients with cirrhosis even if they achieve SVR must have continuous followup with their doctors to look for HCC, liver cancer, even with an SVR there remains a risk for HCC forever, followup with MRI every 6 months forever is required & recommended, is the Standard of Care.
Mark Mascolini
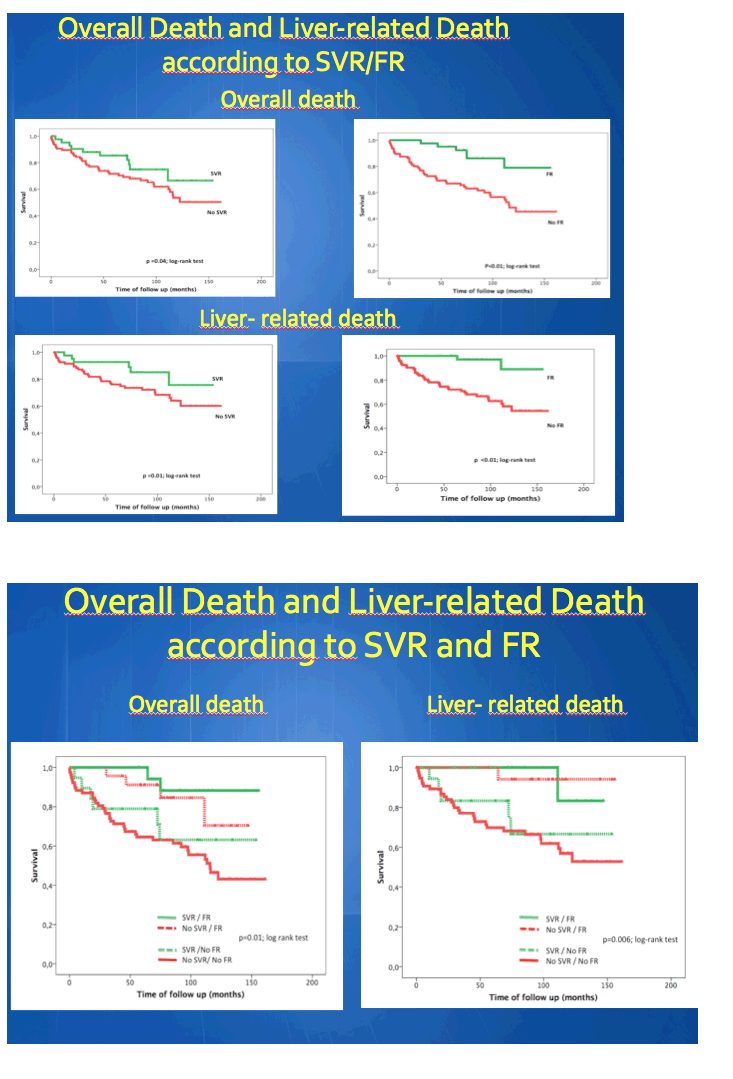
More than half of HCV/HIV-coinfected people with a sustained virologic response (SVR) to anti-HCV therapy achieved fibrosis regression, compared with 15% who did not achieve SVR and experienced fibrosis progression in a study of 133 people in Spain [1]. Through almost 7 years of follow-up, people with SVR and fibrosis regression had significantly lower mortality and liver-related mortality than people with SVR and no regression.
Direct-acting antiviral regimens boosted SVR rates in HCV/HIV-coinfected people, but the impact of SVR on fibrosis regression--and of both factors on liver complications and survival--remains poorly understood. To address these issues, researchers from Madrid's Ramon y Cajal Hospital conducted an observational study of people treated with the pre-DAA standard, pegylated interferon plus ribavirin. The analysis included all coinfected people treated since 2004 and monitored with transient elastography (TE) since 2006. They defined fibrosis regression as a confirmed drop of at least 1 point in the Metavir score, without worsening in a subsequent TE, if available. Follow-up continued until liver transplant, death, or July 2014.
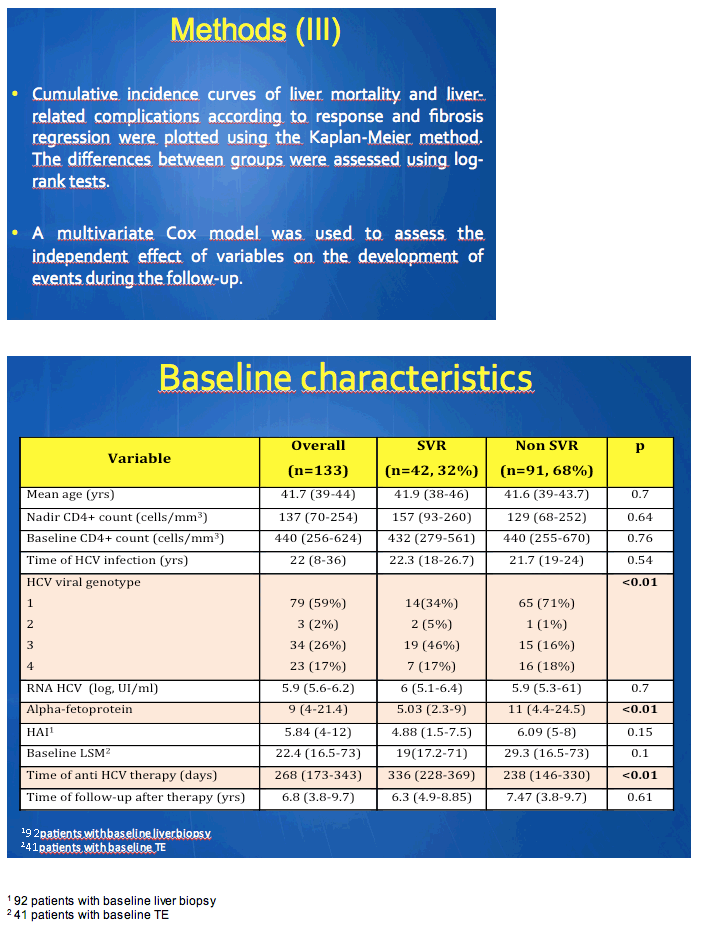
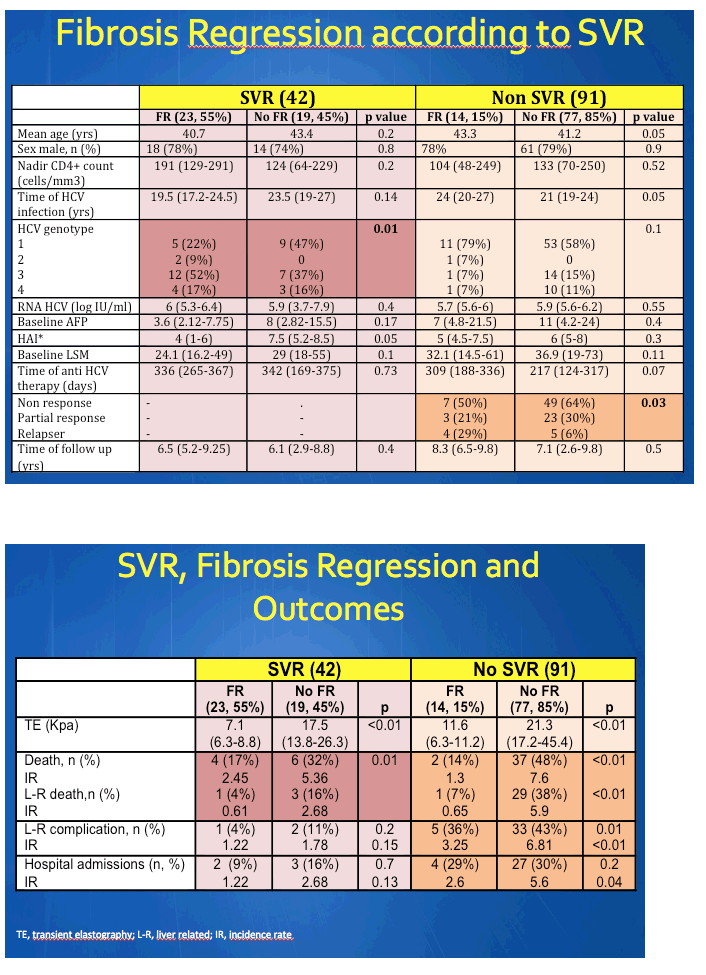
The study involved 133 HCV/HIV-coinfected people with an average age of 41.7 years, an average 22 years of HCV infection, and an average 6.8 years of follow-up since anti-HCV therapy. Forty-two people (32%) achieved SVR and 91 did not. Among the 42 SVR patients, 23 (55%) had fibrosis regression and 19 did not. Among the 91 people without SVR, 14 (15%) had fibrosis regression and 77 did not.
In the SVR group, 4 of 23 with fibrosis regression died, compared with 6 of 19 without regression (17% versus 32%, P = 0.01). Mortality with versus without regression was 2.45 versus 5.36 per 100 person-years. One person with fibrosis regression and 3 without regression had a liver-related death (4% versus 16%, incidence 0.61 versus 2.68, P = 0.01). Liver-related complications and hospital admissions also proved less frequent in the group with fibrosis regression, but not significantly so.
In the 91 people without SVR, 2 (14%) with fibrosis regression versus 37 (48%) without regression died (mortality 1.3 versus 7.6) (P < 0.01). Liver-related deaths numbered 1 (7%) in the fibrosis regression group and 29 (38%) in the no-regression group (incidence 0.65 versus 5.9) (P < 0.01). Liver-related complications were significantly less frequent with than without regression (36% versus 43%, incidence 3.25 versus 6.81, P < 0.01).
Cox regression analysis identified one factor associated with both lower overall mortality and lower liver-related mortality: Fibrosis regression cut the overall death risk 64% (adjusted hazard ratio [aHR] 0.36, 95% confidence interval [CI] 0.15 to 0.86) and the liver-related death risk 85% (aHR 0.15, 95% CI 0.03 to 0.65). Fibrosis regression sliced the risk of liver-related complications more than 90% (aHR 0.09, 95% CI 0.30 to 0.3), while SVR curbed the complication risk more than 75% (aHR 0.24, 95% CI 0.07 to 0.87).
In the entire study group, liver stiffness measure (LSM) after therapy proved a reliable discriminator between outcomes:
-- SVR vs no SVR: LSM Kpa 9.6 versus 18.1, P < 0.01
-- Death vs survival: LSM Kpa 34.8 versus 15.7, P < 0.01
-- Liver-related death versus survival: LSM Kpa 45 versus 15.3, P < 0.01
-- Liver-related complications versus no: LSM Kpa 21.3 versus 15.7, P = 0.03
-- Ascites versus no: LSM Kpa 27.4 versus 15.8, P = 0.08
-- Encephalopathy versus no: LSM Kpa 36.4 versus 15.7, P < 0.01
-- Gastrointestinal bleeding versus no: LSM Kpa 34.8 versus 16.9, P = 0.02
Because LSM can identify cirrhotic patients at high risk of complications, the Madrid team proposed that sequential LSM after therapy could be useful in managing HCV/HIV patients with cirrhosis, regardless of whether they achieve SVR.
Reference
1. Casado JL, Banon S, Quereda C, Moreno A, Perez Elias MJ, Moreno S. Liver fibrosis regression after anti HCV therapy and the rate of death, liver-related death, liver-related complications, and hospital admissions in HIV/HCV co-infected patients with cirrhosis. IAS 2015. 8th Conference on HIV Pathogenesis, Treatment and Prevention. July 19-22, 2015. Vancouver. Abstract TUAB0204.
----------------------



|
| |
|
 |
 |
|
|