 |
 |
 |
| |
Switch from PI/rtv +2 nucleos(t)ides to RPV+DRV/rtv
maintains HIV suppression and is well tolerated (PROBE study)
|
| |
| |
Reported by Jules Levin
IAS 2015 Vancouver July 19-23
Franco Maggiolo, Daniela Valenti, Annapaola Callegaro, Elisa Di Filippo, Giampietro Gregis, Marco Rizzi
AO Papa Giovanni XXIII, Bergamo, Italy

-------------------------
Program Abstract
Switch from PI/rtv +2 nucleos(t)ides to RPV+DRV/rtv maintains HIV suppression and is well tolerated
Presented by Franco Maggiolo
F. Maggiolo1, D. Valenti1, A. Callegaro2, E. Di Filippo1, G. Gregis1, M. Rizzi1
1AO Papa Giovanni XXIII, Infectious Diseases, Bergamo, Italy, 2AO Papa Giovanni XXIII, Microbiology and Virology Lab, Bergamo, Italy
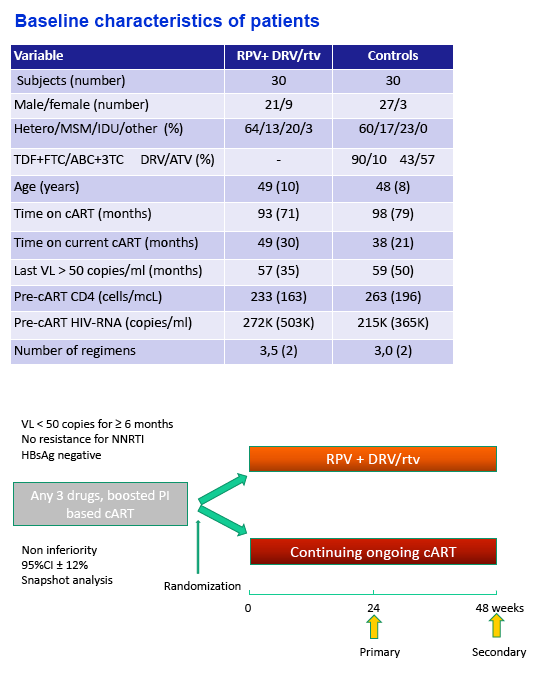
Background: A nucleos(t)ides (NRTI) free regimen may be useful in selected patients who developed NRTI related toxicity. We report the week (W) 24 results of a pilot, prospective, randomized, ongoing trial of switch to a dual regimen rilpivirine + ritonavir-boosted darunavir (RPV+DRV/r).
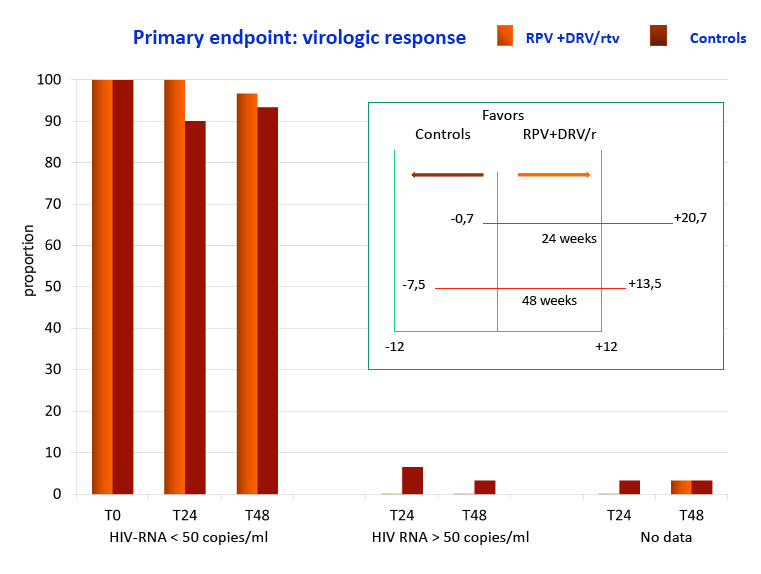
Methods: Virologically suppressed subjects on PI/r + FTC/TDF or 3TC/ABC for > 6 months were randomized (1:1) to switch to RPV+DRV/r or remain on their baseline 3 drugs regimen (control). Eligibility criteria included no documented resistance to RPV and no HBV co-infection. The primary endpoint was the proportion of subjects who maintained HIV-RNA < 50 copies/ml at W24 by FDA snapshot algorithm (12% noninferiority margin).
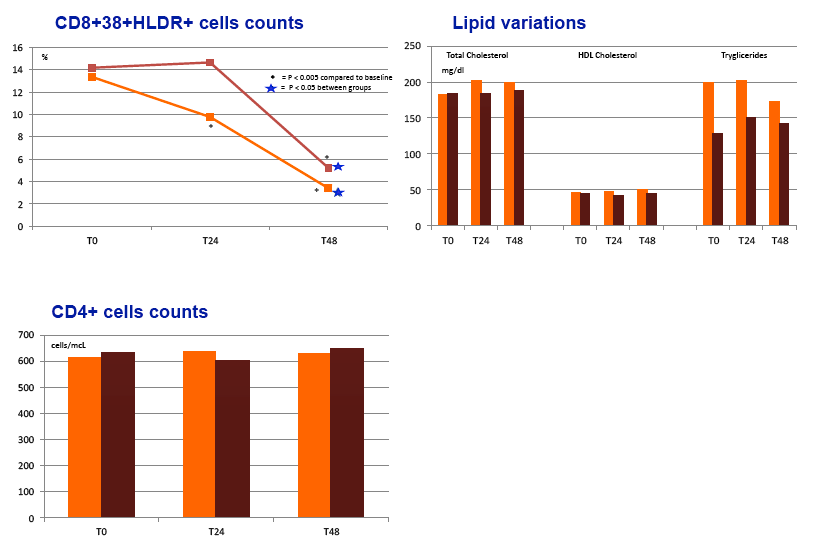
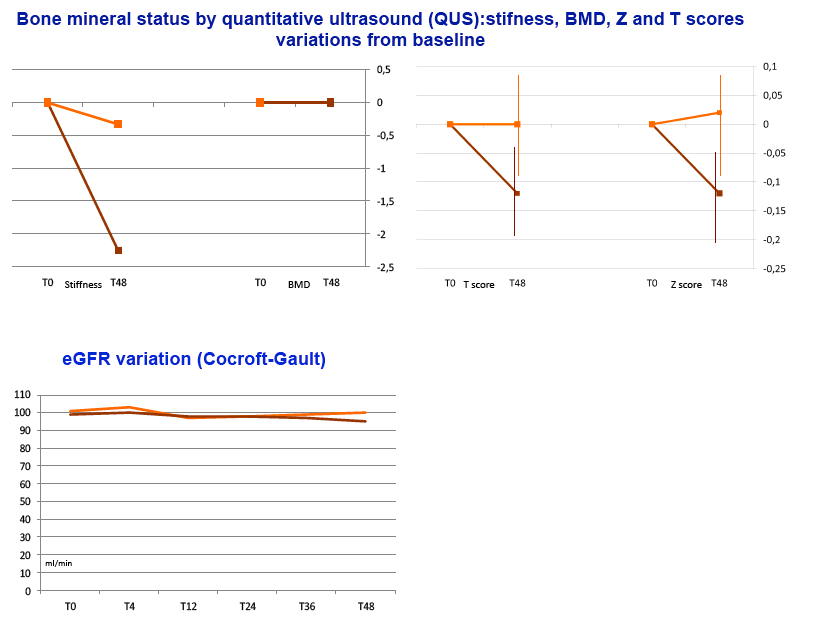
Results: Sixty subjects (80% male, 22% with a previous diagnosis of AIDS, mean age 48 years) were randomized and treated (30 RPV+DRV/r; 30 controls). At randomization FTC/TDF was used in 83% of subjects and the most commonly used PIs were ritonavir-boosted atazanavir (57%) and ritonavir-boosted darunavir (35%). Median years since first ARV use was 6.2 and median number of regimens 3. The mean CD4 count was 623 cells/mcL and baseline characteristics were similar between the groups. At W24 100% of subjects who switched to RPV+DRV/r maintained HIV-RNA < 50 copies/ml compared to 86.7% of controls (difference 13.3%, 95% CI -1.1% to +25.4%). There was no confirmed virologic failure and, at W24, HIV-RNA was < 3 copies/ml in 66.7% of subjects in either group. Compared to controls, subjects on RPV+DRV/r experienced a greater CD4+ increment (mean +24 vs -13 cells/mcL ), CD8+ decrement (mean -4 vs +17 cells/mcL) and CD8+CD38+HLA*DR+ decrement (mean -3.3 vs +1.2%). There was no AE leading to drug discontinuation. At W24 there was a lower increment in fasting triglycerides for RPV+DRV/r (+10 mg/dl vs +23mg/dl) and a larger increment in both total (+14 mg/dl vs -0.7 mg/dl) and HDL cholesterol (+0.6 mg/dl vs -4.2 mg/dl).
Conclusions: Switching to RPV+DRV/r compared to continuing a PI/r + FTC/TDF or 3TC/ABC demonstrated virologic non inferiority. RPV+DRV/r presented slight immunologic advantages and was well tolerated with a favorable safety profile. Switching to this NRTI free regimen may be an option for patients experiencing NRTI related toxicity.
--------------

|
| |
|
 |
 |
|
|