 |
 |
 |
| |
Summary from CROI 2016 for Hepatitis C
Feedback from real-life: how is HCV therapy working outside of clinical trials?
|
| |
| |
Jurgen K. Rockstroh M.D., Professor of Medicine
University of Bonn, Germany
Correspondence:
Prof. Dr. J.K. Rockstroh
Department of Medicine I
University of Bonn
Sigmund-Freud-Str. 25
53105 Bonn
Germany
Introduction
At this year CROI, far less new phase-3 clinical studies in hepatitis C mono- or coinfection have been reported than in previous years. Clearly, with various registered all oral DAA combinations in the US and Europe, this year focus was on feedback from implementation of DAA-based therapy in real-life HIV/HCV cohorts. Beyond efficacy and safety in real-life settings, one important question has become how to best retreat patients with previous DAA treatment failure. One session was devoted to discuss the relevance of HCV resistance associated variants (RAVs) in this context. Also very interesting, new insights into practical aspects such as drug-drug interactions between HIV and HCV drugs was presented. In addition, new study results on the treatment of acute HCV with all oral DAA combinations was shown for the first time. Finally in the symposium on global aspects of HCV it was impressively demonstrated how in resource constrained settings huge HCV treatment roll out can take place as part of a broader HCV elimination program underlining if there is sufficient political will and strong support from the medical community access to HCV therapy can become reality. This form of strong political will and leadership is needed in many more regions of the world where HCV DAA treatment access remains just a wish.
DAA Therapy in Acute Hepatitis C
At AASLD several pilot trials and case series were presented which examined the efficacy and safety of shorter treatment durations with sofosbuvir + ribavirin combination for treatment of acute HCV (1-3). The first study was the ACTG study (A5327) also named as SWIFT-C which was an open label, two-cohort clinical trial (N=44). SVR12 rate in the first cohort (n=17) which received sofosbuvir 400mg QD + weight based ribavirin for 12 weeks was surprisingly low with only 59 % (1). In order to better understand which factors were potentially associated with higher risk of relapse the authors performed a multivariate analysis looking at traditional factors associated with relapse (baseline HCV RNA, HCV genotype and IL28B genetics) but also measured ribavirin levels in order to evaluate whether ribavirin levels had an impact on achieving SVR (4). RBV plasma samples were analyzed using a validated LC-MS/MS assay. Most interestingly, traditional factors did not differ in those that achieved SVR vs. relapse; nor did CD4 count or duration of HCV infection. However, ribavirin concentrations were 52% higher in those that achieved SVR (p=0.01). The ribavirin concentrations over time for patients achieving SVR versus developing relapse are shown in figure 1. No statistical differences in those that achieved SVR versus relapsed were observed with regard to factors such as race, weight or inosine triphosphatase which are all known to potentially affect ribavirin pharmacokinetics. In addition, no statistical difference was found between those that achieved SVR versus relapsed in self-reported adherence.
Figure 1: Ribavirin concentrations for patients achieving SVR versus relapse
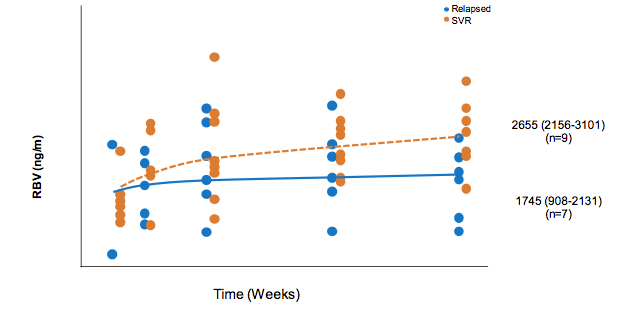
In summary, in the SWIFT-C acute HCV trial, RBV exposures were lower at the end of treatment in HIV-infected participants that relapsed to SOF/RBV therapy for acute HCV compared to those that achieved SVR. The cause of this discrepancy in RBV exposures however, remains unclear, but could relate to pharmacokinetic variability, a chance imbalance between groups, or differences in adherence despite the high levels of self-reported adherence.
Webcast:http://www.croiwebcasts.org/console/player/29625?mediaType=audio&
Although the sofosbuvir + ribavirin 12 week treatment of acute HCV was disappointing clearly the future will be the use of an IFN and RBV free oral DAA combination. First reports, again from AASLD, showed up to 100% and 93,3% for 4 weeks of Sofosbuvir/Ledipasvir (n=14) or 8 weeks of Sofosbuvir/Simeprevir (n=15), respectively (5). All patients had a genotype 1 and were recruited from 6 inner city drug rehabilitation programs in Brooklyn, NY. As much as these results are promising, a full paper is still lacking despite numerous presentations at various conferences and some important questions remain unanswered such as the median duration of HCV diagnosis and the question whether patients with high baseline HCV viral load were also included into this trial. At CROI for the first time data from an acute HCV trial investigating efficacy and safety of an 8 week therapy with Ledipasvir/Sofosbuvir was presented (6). The study design is shown below in figure 2.
APASL:SLAM C study Sofosbuvir and Ledipasvir versus Sofosbuvir and Simeprevir combination therapy in the management of acute hepatitis C: A randomized open label prospective clinical pilot study. (drug rehabs in NYC] - (02/22/16)
Figure 2: Study design
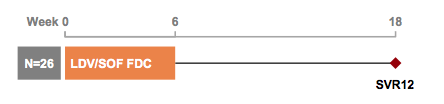
The study included patients with chronic HIV and acute HCV infection with either HCV GT 1 or 4 and ART consistent with LDV/SOF co-administration with HIV <200 copies/mL or not receiving ART with no plans to start. Acute HCV was defined as HCV RNA-positive and negative anti-HCV antibody/HCV RNA test within last 6 months or elevated ALT/AST >2.5 x ULN in past 6 months with normal LFTs in past year, and other causes excluded. Overall 26 subjects were enrolled. All patients were male, 92% were white, 69% had GT1 and 31% GT4. Mean HCV viral load was 5.4 log10 IU/mL (range < LLOQ–7.3). The main SVR results for this study are shown below in figure 3.
Figure 3: Sustained virological response (SVR) 4 and 12 weeks after stopping HCV therapy (SVR 4 and SVR12)
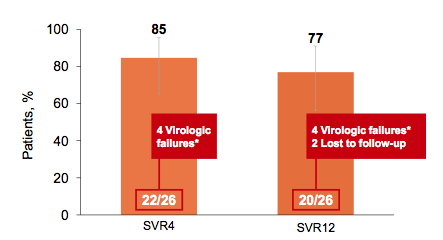
*3 patients relapsed, 1 was reinfected (GT 1a at baseline, 4d in post-treatment).
Error bars represent 95% confidence intervals.
Treatment with LDV/SOF for 6 weeks resulted in a 77% SVR12 rate in HIV-infected patients with acute HCV infection. All of the 3 relapses occurred early between end-of-treatment and SVR4. In the same time window one reinfection occurred with a different GT than detected at baseline (shift from GT1a to 4d). Once again this underlines that DAA therapy alone is not enough for controlling the HCV epidemic among HIV-seropositive men who have sex with men (msm) but in addition behavioral interventions are needed to control the epidemic. 2 patients were lost to follow-up between week 4 and 12 of post-treatment follow-up. Most interestingly only patients with a high baseline HCV viral load above 6.9 log developed a relapse (see figure 4).
Figure 4: Baseline HCV RNA and Treatment Outcome (SVR)
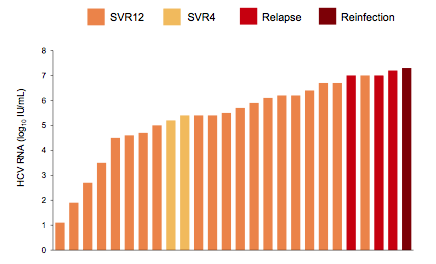
Interestingly, all patients who relapsed had a high viral load between 7.0 and 7.2 log. Remembering, that in management of treatment naïve chronic HCV patients with no advanced fibrosis and HCV viral loads below 6 Mill I.U. 8 weeks of Ledipasvir/Sofosbuvir have been demonstrated to be sufficient in obtaining cure of HCV in >95% the idea becomes reinforced that in the treatment of HCV, regardless whether acute or chronic in patients with favorable host factors (no cirrhosis, and no history of treatment failure) shorter treatment durations are feasible. The favorable impact of very early treatment however, seems not to play out in acute HCV most likely because the higher SVR rates observed with IFN in treatment of acute HCV versus chronic HCV were related to immunologic properties of IFN which may not be exerted by DAAs. The fact though that all patients with HCV viral loads below 7 log responded is very reassuring and can help to guide treatment decisions during acute HCV. If HCV viral load is higher most likely either waiting to see whether HCV viral load drops or longer treatment durations of 12 weeks should be recommended. Of note no new NS5A or NS5B RAVs were observed at relapse suggesting that shorter durations of DAA combination are less likely to end in the development of relevant RAVS than has been reported for longer treatment durations and subsequent relapse. Safety of the short 6 week treatment with Ledipasvir/Sofosbuvir was very well tolerated and no discontinuation because of adverse events occurred. Obviously, with the high costs of modern DAA therapy strategies which help reducing drug costs are urgently needed. Early treatment of HCV may indeed be very cost-effective if shorter treatment durations can reliably be applied.
CROI: Ledipasvir/Sofosbuvir for 6 Weeks in HIV-Infected Patients with Acute HCV Infection - (03/01/16)
Feedback from real-life patient cohorts
An update of the German GECCO cohort was presented at CROI (7). The GECCO cohort is a multicenter cohort from 9 sites in Germany. All patients started on the following DAAs were included in the analysis (Figure 5): Sofosbuvir (SOF), pegylated interferon and ribavirin (RBV); SOF and RBV; SOF and simeprevir (SMV); SOF and daclatasvir (DCV) +/- RBV; SOF and ledipasvir (LDV); paritaprevir/ritonavir (PTV/r), ombitasvir (OBV)+/- RBV and +/- dasabuvir (DSV).
Figure 5: DAA regimes and duration of treatment documented in GECCO
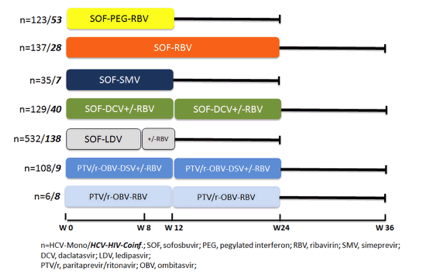
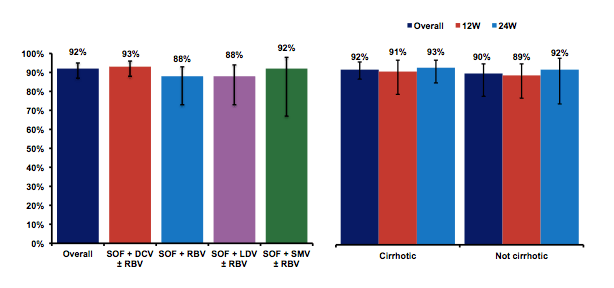
Up to date, 1346 patients have been included into the cohort. The HCV GT distribution was as follows: 996 (74%) GT 1, 51 (4%) GT 2, 221 (16%) GT 3, 78 (6%) GT4. Almost a quarter of patients had advanced fibrosis (F4) based on non-invasive fibrosis markers. N=282 were HIV-coinfected patients with a median CD4+ cell count of 606/mm3. SVR12 rates for the whole cohort according to genotype and DAA regimen are shown in Figure 6.
Figure 6: Treatment efficacy stratified by genotype and DAA regimen (with at least n=10 treated, ITT)
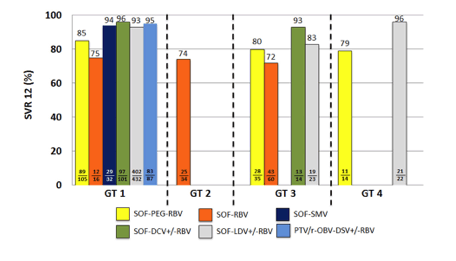
On average very high SVR12 rates around 95% were observed for all oral DAA combinations. Lower response rates were seen for SOF+PEG-IFN+RBV and for GT 3 particularly in patients receiving sofosbuvir and ribavirin. Figure 7a shows SVR12 rates depending on baseline factors. Although some subgroups end up with relatively small numbers it is impressive to see that also in real-life cohorts there is no difference between the cure rates between HIV/HCV coinfected subjects and HCV mono-infected patients. Moreover, it appears even in the traditionally more difficult-to-treat patients including F4 fibrosis or diabetes similar high SVR rates are achieved under DAA therapy than in patients who do not depict these risk factors. In figure 7b efficacy of a shorter treatment duration of 8 weeks of Ledipasvir/sofosbuvir is analyzed in various subgroups again independent of baseline risk factors shows overall strong SVR12 rates
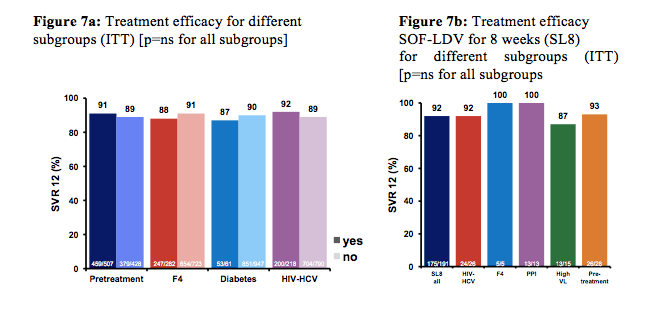
Currently, the AASLD guidelines do not recommend shorter treatment durations in HIV/HCV coinfected patients because of the fear that this may not be sufficient for this specific patient population. In particular, the 8 week arm with sofosbuvir and daclatasvir in the Ally-2 study underperformed, raising corresponding doubts around shorter treatment durations in HIV-patients (8). Noteworthy though, in the 8-week arm of Ally-2 there was no viral load cutoff and secondly because of missing PK interaction data, patients on boosted PIs within this study had all received lower daclatasvir doses of 30mg which may have been under dosed. In consideration of the artificial restrictions in access to HCV antiviral therapy throughout the world shorter treatment durations would come at considerable cost savings and therefore warrant further investigation.
A further real-life DAA cohort was presented from France. HIV-HCV co-infected patients enrolled in the French nationwide ANRS CO13 HEPAVIH cohort were included in this analysis if an oral DAA-based regimen without peg-interferon was initiated before March 1st 2015 (3-month regimen) or before December 1st 2014 (6-month regimen) and if the patients had sufficient follow-up data to evaluate DAA efficacy (9). Treatment success was defined as an undetectable HCV-RNA (<15 UI/mL) at 12 weeks or thereafter (SVR12). Patients with premature treatment stops, detectable HCV-RNA and those who died during treatment were considered as treatment failures. Overall, 215 HIV HCV co-infected patients from the ANRS HEPAVIH cohort were examined. 67% of the patients analyzed were cirrhotics, 71% were treatment experienced. So clearly, this was a very advanced liver disease enriched cohort and therefore difficult to treat. Most reassuringly however, no influence of cirrhosis or pre-treatment upon SVR was found for all oral DAA combinations (see figure 8a and b). Again lowest cure rates were observed for the sofosbuvir and ribavirin only treatment arm. Overall, an impressive 92% of patients achieved SVR12 with all the different DAA based therapies used. No significant difference in outcome was seen between 12 or 24 weeks of DAA therapy.
Figure 8a: SVR rates for the various DAA regimens Figure 8b: Impact of cirrhosis on SVR
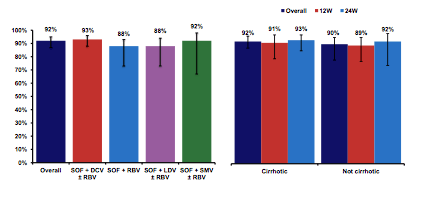
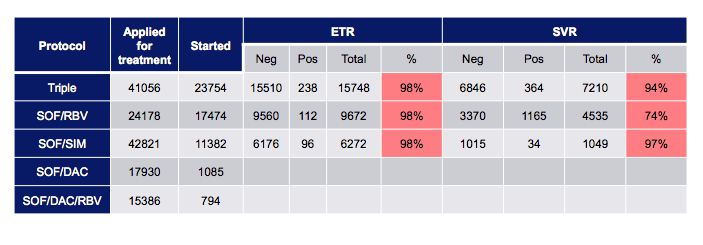
In conclusion, real-life HCV treatment data shows a very impressive HCV cure rate even in much more difficult to treat patient populations. Now obviously this data is largely from countries with access to DAA based therapy. How are these regimens going to play out in more resource limited settings or countries with different levels of medical care? It was most thrilling to listen to the Egyptian status report on their ambitious HCV elimination program. Almost 200.000 HCV patients have already been treated with DAAs in 2015 with response rates being similar to real life data from Europe/US. Table 1 shows end-of-treatment (ETR) and sustained virological responses (SVR) for the various DAA based regimens used. Note that triple therapy still consisted of PEG-IFN + ribavirin and sofosbuvir. Lowest cure rates were achieved with sofosbuvir and ribavirin which is well in line with other data for this combination in GT 4 patients. It is important to remember that in Egypt over 95% of HCV infections are all genotype 4 infections. The overall goal is to treat up to 1.2 Million HCV-infected individuals in the next coming months. By now additional DAA combinations have been approved and are ready to be used enabling simplified regimens without IFN or RBV for the majority of patients. Note that the average price for 12 weeks of treatment for an all oral DAA combination is estimated on average to be around 800 US$. Clearly, similar price arrangements are urgently needed in many other regions of the world.
Table 1: Real Life DAA Data from Egypt
DAA resistance in the real world
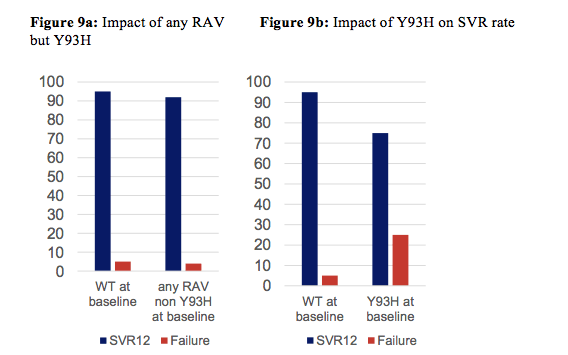
Despite great HCV treatment success rates which have become achievable with modern DAA therapy, on average in real life cohorts 5-10% of patients end up with virological failure prompting the question of which resistance conferring mutations may occur, which impact they subsequently may have on treatment outcome, and whether if present at baseline these mutations may have independently led to relapse. At CROI one whole themed discussion was dedicated towards "rants about HCV resistance associated variants (RAVS)". Interesting data was presented in this context from France, where colleagues analyzed HCV resistance to Daclatasvir/Sofosbuvir across different genotypes in the real life at baseline and after virological failure (11). Patients from this cohort (n=177) were treated for 12 weeks. 44% had cirrhosis, 60% were treatment experienced at baseline. Population sequencing of the NS5A and NS5B genes was performed prior to SOF/DCV treatment in 177 patients (GT 1a, n=44; GT1b, n=63; GT3, n=29; GT4, n=41) and at treatment failure (relapse) in 8 patients who did not achieve SVR12. NS5A sequences were examined at 9 known RAV positions in domain I (residues 28, 29, 30, 31, 32, 58, 62, 92 and 93), while NS5B sequences were examined at 5 known RAV positions (159, 282, 316, 320, 321). Prior to treatment, NS5A RAVs were detected at a frequency of 9% in GT1a-, 32% in GT1b-, 21% in GT3- and 10% in GT4-infected patients. The most frequent RAVs were Y93H in GT1b (11%) and GT3 (3.5%), and L28M in GT4 (10%). No NS5B S282T, L320F or V321A variants were detected, while L159F was found in 12% of GT1b patients at baseline. C316N and C316H were detected in GT1b patients (20% and 2%, respectively) and in GT4 patients (5% and 2%, respectively). RAVs frequency at baseline did not differ between patients with and without cirrhosis. Figure 9a and 9b show the impact of any RAV but non-Y93H, and of the Y93H RAV on SVR rate. Indeed, virological failure tended to be more frequent when an NS5A Y93H substitution was present at baseline (p=0.067). All relapse patients harbored dominant NS5A RAVs post-treatment, with Y93H present in all GT3-infected subjects failing treatment. In addition, a so far undescribed NS5A amino-acid substitution (H85Y) was detected in a GT3-infected patient at relapse.
The patient profile and NS5A as well as NS5B mutations at baseline and at virological failure are summarized in table 2 for the 8 relapsers in this cohort.
Table 2: DAA resistance in patients failing after Daclatasvir/Sofosbuvir
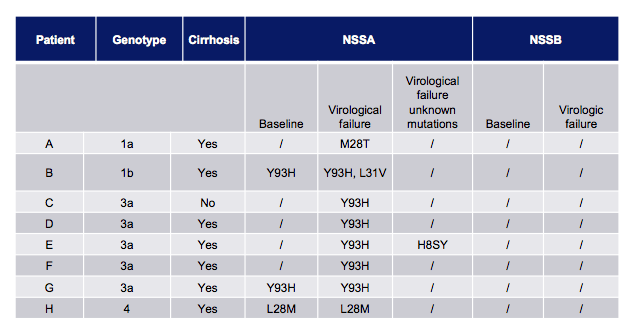
Interestingly, all but one patient who developed failure had cirrhosis. At failure all patients had a NS5A mutation. Only 3 of the relapsers already depicted relevant RAVs at baseline.
A further study looking at resistance associated variants came from the NIAID SYNERGY trial, which was an innovative 8 arm clinical trial that treated over 200 hepatitis C patients with varying sofosbuvir (SOF)-based DAA regimens (see figure 10) (12).
Figure 10: SYNERGY Trial study: Study arms and SVR rates
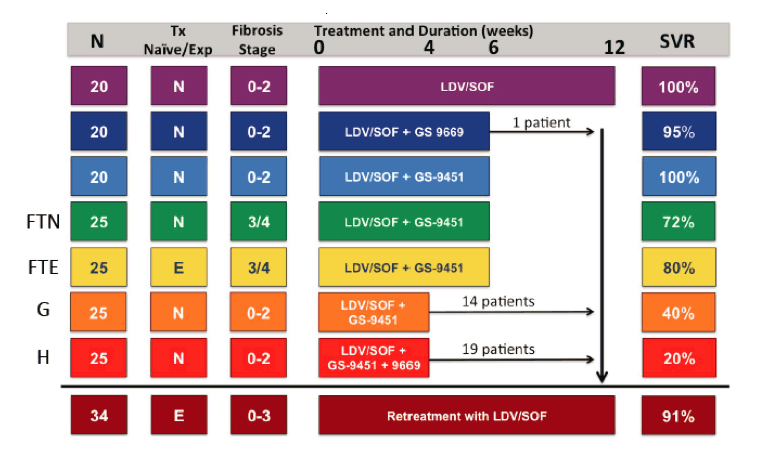
Deep sequencing was performed on plasma at baseline on all patients, at the time of relapse in patients failing therapy, and prior to retreatment in order to differentiate treatment-enriched RAVs (those increasing in proportion to wild type from baseline to relapse) and treatment-emergent RAVs (those which were not present at baseline). Correlation analysis was performed between RAVs and sustained virologic response (SVR). Of 160 patients treated initially, 99 patients had baseline RAV testing available. Figure 11 shows the prevalence for NS5A RAVS and their impact on SVR rates for study arms F (treatment-naïve (TN) and treatment experienced (TE)) G and H.
Figure 11: Baseline RAV prevalence and outcome by study arm
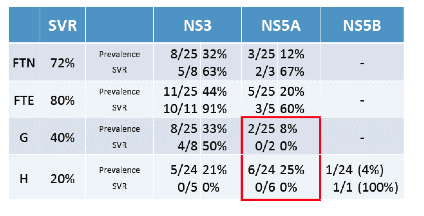
Overall, high level NS5A RAVs were infrequent but did tend to persist over time. High level NS5A RAVs only predicted poorer response to short course therapy (4 week arm G and H). Retreatment with 12 weeks of ledipasvir/sofosbuvir was effective in 91% of patients failing short course therapy, including those with high level NS5A RAVs. These results indicate that increasing treatment duration may help to overcome relevant RAVs at baseline.
CROI: Resistance Associated Variants: Data From the NIAID SYNERGY Trial / Harvoni-Retreating Short Duration Therapy - (03/07/16)
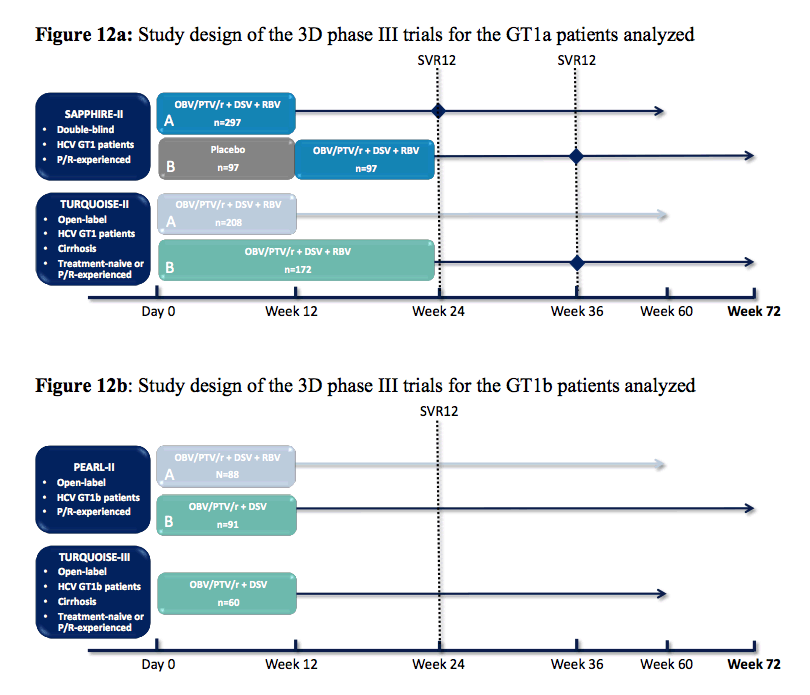
One of the important things to learn in the evolving field of RAVS and what they mean for treatment outcome is that frequency and eventually potential impact of a RAV will crucially depend on the genotypic resistance assay used or the respective next generation sequencing cutoff applied. Deep sequencing and low cutoffs will lead to higher frequency of RAVs and thereby potentially dilute the sample making it more unlikely to find an impact of baseline variants on treatment outcome. Furthermore, definition of what counts as a relevant RAV will also be of importance and potentially impact outcomes, as some RAVs seem to have much more impact than others. Furthermore, having RAVs per se does not preclude treatment success so often it appears more like the combination of RAVs and short treatment duration or unfavorable host factors (cirrhosis, previous treatment failure etc.) add up to risk for relapse. Therefore, in many studies one can now see a separation between NS5A RAVS and then those specific RAVs which have been associated with a greater loss in susceptibility of the respective NS5A inhibitor being studied. In a late breaker Poster Mark Sulkowski assessed the prevalence of baseline RAVs and their impact on the rate of sustained virologic response (SVR) in Phase 3 trials with the 3D regime (paritaprevir/r/ombitasvir + dasabuvir) (13). The study included HCV GT1a-infected patients treated with the 3D regimen + RBV in SAPPHIRE-II (prior P/R treatment experience, no cirrhosis) and TURQUOISE-II (treatment naive and prior P/R treatment experience, compensated cirrhosis) as well as HCV GT1b-infected patients treated with 3D without RBV in PEARL-II (prior P/R treatment experience, no cirrhosis) and TURQUOISE-III (treatment naive and prior P/R treatment experience, compensated cirrhosis). The study designs of these respective trials are summarized in figure 12a and b.
Figure 12a: Study design of the 3D phase III trials for the GT1a patients analyzed
RAVs were determined by next generation sequencing (NGS). Detection thresholds of 1 and 15% were used for the analysis, consistent with the limits of detection for variants by NGS and population sequencing, respectively. The following were considered GT1a NS5A class RAVs: M28(all), Q30(all), L31(all), P32L, H58D/R, and Y93(all). The following were considered 3D regimen-specific RAVs for GT1a: Paritaprevir-specific RAVs: F43L, Y56H, R155G/K/S/T/W, A156S/T/V, and D168A/E/F/H/N/V/Y. Q80K was evaluated separately based on the significance of this variant for the NS3 inhibitor simeprevir. Ombitasvir-specific RAVs: M28T/V, Q30E/K/R/Y, L31V, P32L, H58D, and Y93C/F/H/L/N/S. Dasabuvir-specific RAVs: L314H, C316Y, M414I/T/V, E446K/Q, Y448C/H, C451R, A553T, G554S, Y555H, S556G/R, G557R, G558R, D559G/N, Y561H/N. The following were considered 3D regimen-specific RAVs for GT1b: Paritaprevir-specific RAVs: Y56H, R155K, A156T/V, and D168A/F/H/K/T/V/Y. Ombitasvir-specific RAVs: L28M/T, P29del, R30Q, L31F/V, P32del, P58A/S, A92E, and Y93H/N/S. Dasabuvir-specific RAVs: C316H/N/W/Y, S368T, N411S, M414I/T/V, C445F/Y, Y448C/H, A553V, S556G, and D559G. Figure 13 shows the effect of method and threshold of RAV detection on prevalence of NS5A RAVs.
Figure 13: Prevalence of baseline GT1a NS5A RAVs: Impact of RAV definition and sensitivity of detection
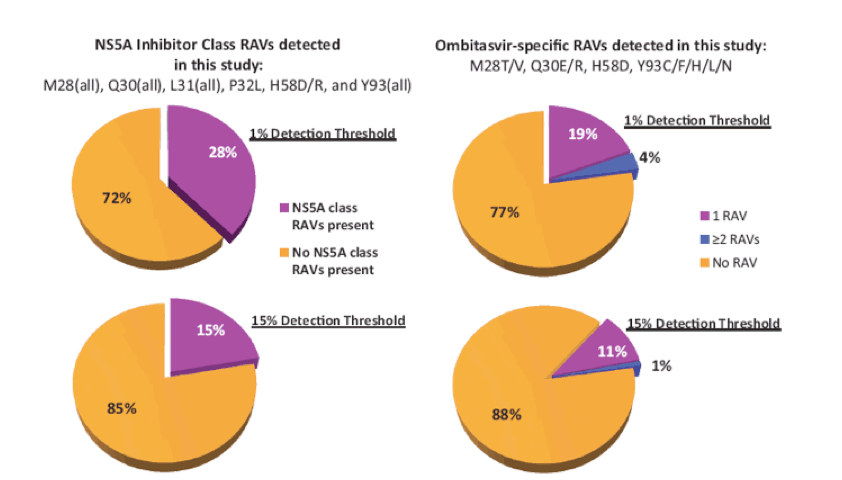
These figures demonstrate clearly that lower detection thresholds substantially increase frequency of RAVS analyzed. Therefore, identification of RAVs using a 1% threshold may underestimate impact. Moreover, frequency of RAVS depends on whether any amino acid variant at resistance-associated positions is included or only inhibitor class RAVs, or compound-specific RAVs.
Obviously the other objective of this analysis was to assess in particular the prevalence of baseline RAVs and their impact on the rate SVR of sustained virologic response (SVR) in In HCV GT1a-infected patients treated with the 3D regimen + RBV. Figure 14 shows the corresponding SVR rates in association with baseline GT1a NS5A Class RAVs and Ombitasvir-specific RAVs.
Figure 14: Impact of Baseline GT1a NS5A Class RAVs and Ombitasvir-specific RAVs on SVR Rate
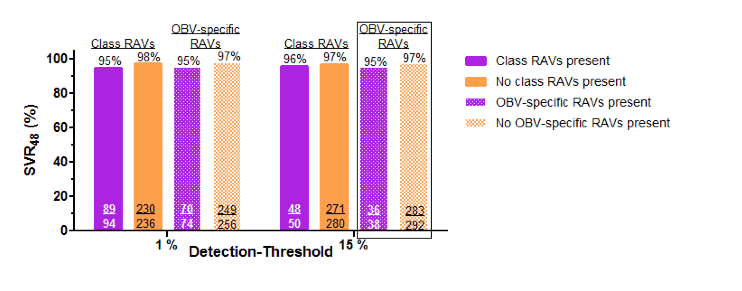
Most interestingly, in the phase III trials of the 3D regimen GT1a patients treated with the label-recommended regimens of 3D + RBV for 12 weeks (no cirrhosis) or 24 weeks (cirrhosis) achieved similarly high SVR rates regardless of the presence or absence of baseline RAVs. Obviously, the addition of ribavirin could be meaningful here as data also from other DAA combinations such as grazoprevir/elbasvir suggest that extending treatment duration from 12 to 16 weeks and adding ribavirin may help to overcome the negative impact of baseline RAVs on SVR rates in GT1a patients. In GT1b the importance of RAVs seems much less prominent and indeed in this analysis all GT1b patients treated for 12 weeks with 3D (no RBV) achieved SVR, regardless of the presence or absence of baseline RAVs.
CROI: Effect of Baseline Resistance-Associated Variants on SVR With the 3D Regimen Plus RBV - (02/24/16)
One of the big questions around RAVs is in case of DAA treatment failure which treatment strategies may help to secure SVR once patients have developed RAVs after relapse. Therefore, it was very interesting to see a presentation on the retreatment of patients who had failed in the ION-4 trial (coinfection study for GT 1 or 4 patients with HIV/HCV coinfection who were treated with 12 weeks of Ledipasvir/Sofosbuvir) (14). Overall, 9 individuals were retreated with the same DAA combination but extended treatment duration of 24 weeks and addition of weight adapted ribavirin. Figure 15 summarizes the NS5A RAVs before primary study as well as after relapse. Most impressively, SVR was achieved in 8/9 subjects with only one relapse 4 weeks after EOT in a GT1a patient who was non-cirrhotic.
Figure 15: Baseline NS5A RAVs and Treatment Outcome (N=9)
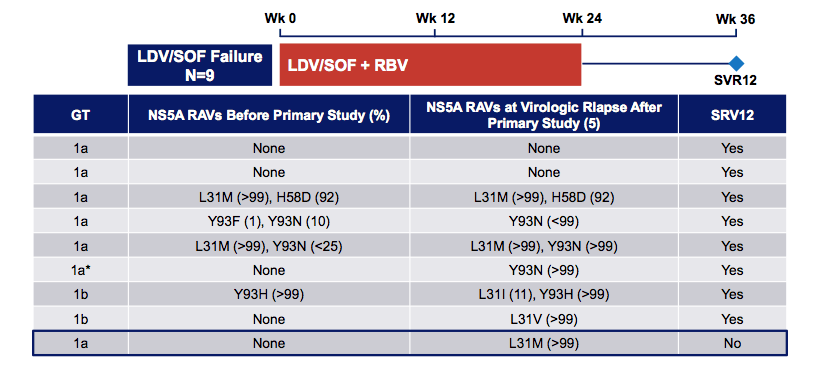
Ledipasvir/sofosbuvir with ribavirin for 24 weeks was well tolerated and demonstrated that successful retreatment is possible in the majority of these genotype 1-infected, NS5A-experienced HCV/HIV co-infected patients. Obviously, extending treatment duration and adding ribavirin can help to achieve SVR despite prior failure on the same DAAs and for the majority development of NS5A RAVs upon relapse.
CROI: Retreatment of HCV/HIV-Coinfected Patients Who Failed 12 Weeks of LDV/SOF - (03/05/16)
New DAA drugs and combinations
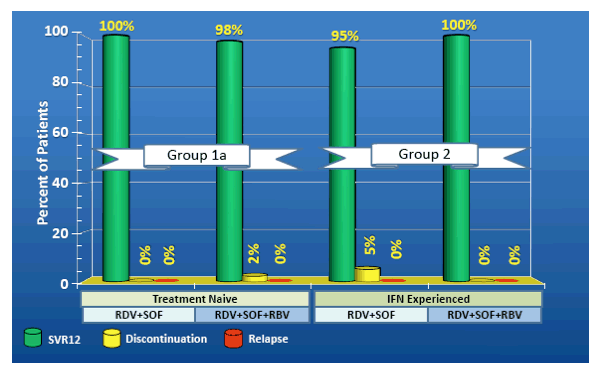
At this year CROI only few new DAA studies were shown. However, numerous studies were presented which examined drug-drug interactions between HIV agents and some of the newer DAAs currently waiting for regulatory approval or further continuation of their clinical development program. One new DAA combination study which was presented at CROI in the oral hepatitis session was a phase 3 registrational trial in Egyptian HCV GT-4 patients, assessing the combination of ravidasvir (RDV), a pan-genotypic HCV NS5A inhibitor, and sofosbuvir (a nucleotide HCV NS5B polymerase inhibitor), with or without ribavirin (RBV). Key inclusion criteria were age 18-65 years, HCV GT-4 infection, serum HCV RNA >4 log10 IU/mL, and absence of decompensated cirrhosis or other causes of liver disease. Egypt has the highest prevalence of hepatitis C infection in the world, of which 90% are due to HCV genotype-4 underlining the urgency of developing DAA combinations particularly for treatment of this otherwise more infrequent GT. Figure 16 shows the study design of this trial which included non-cirrhotic IFN-naïve and experienced (Group 1a and 2) as well as cirrhotic again IFN-naïve or experienced (Group 1b and 3). Within groups 1 and 2 patients were randomized to +/- ribavirin. The most difficult to treat group (cirrhotic with previous treatment failure) was randomized to 12 vs 16 weeks of ravidasvir + sofosbuvir + ribavirin.
Figure 16: Study design of the ravidasvir and sofosbuvir trial in Egypt.
The results for the non-cirrhotic patients in group 1a and 2 are shown below in figure 17. Among the 170 non-cirrhotic patients enrolled, there were three early discontinuations unrelated to safety issues or virological failure with no relapses, therefore resulting in a 100% SVR rate if discontinuations are excluded from the analysis. Clearly, these are extremely impressive results in most likely one of the largest controlled GT4 trials to date.
Figure 17: SVR 12 Outcomes in Non-Cirrhotic Patients (ITT)
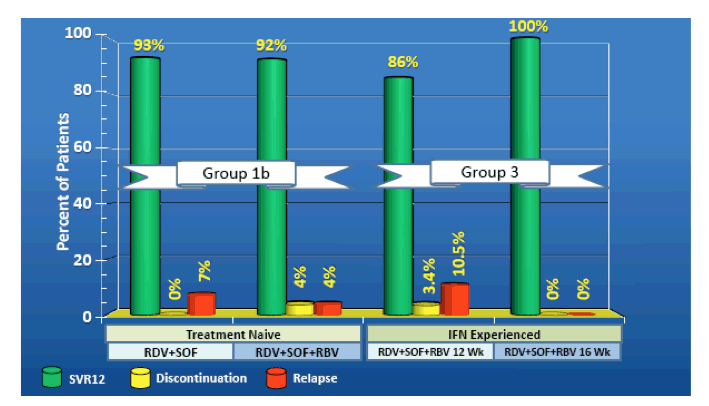
The SVR results for the cirrhotic patient groups is shown in figure 18. Among the 130 cirrhotic patients enrolled there were two premature discontinuations, one being safety related, and six virological relapses. No relapses to date were recorded in the cirrhotic 16 week treatment cohort suggesting that extension of treatment duration may help to lower relapse rate in this more difficult to treat patient group. Per protocol SVR12 rate in this cirrhotic patient group amounted to an impressive 94%.
Figure 18: SVR12 Outcomes in the Cirrrhotic Patients (ITT)
Overall, there was a low incidence of adverse events. Headache and fatigue being slightly more common in the ribavirin treated patients. One SAE was recorded which was a transient episode of symptomatic bradycardia in a cirrhotic patient. In conclusion, RDV/SOF could be an attractive once-daily regimen for people with HCV genotype 4 infections with excellent safety and efficacy results. According to the presenter ravidasvir is undergoing regulatory approval and registration as a HCV DAA in Egypt currently.
CROI: Ravidasvir/Sofosbuvir Yields Quick SVRs in Largest Genotype 4 Trial
WEBCAST:http://www.croiwebcasts.org/console/player/29746?mediaType=audio&
Sofosbuvir/Velpatasvir (SOF/VEL) (400/100 mg) FDC treatment of HCV for 12 weeks in Phase 3 studies resulted in high SVR in patients with HCV GT 1-6. The regimen is currently under regulatory review. At CROI a study evaluating drug-drug interactions between SOF/VEL (400/100 mg) and boosted-ARV regimens was presented. VEL is a substrate of P-gp, BCRP, OATP, CYP2B6, CYP2C8, CYP3A4 and an inhibitor of P-gp, BCRP, and OATP thereby offering at least some potential for DDIs. The figure 19 shows the various study arms which were evaluated.
Figure 19: Phase 1 drug-drug interaction studies in healthy volunteers receiving SOF/VEL and antiretroviral regimens.
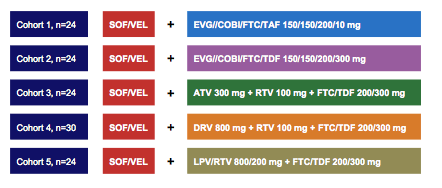
These were multiple-dose, randomized, cross-over DDI studies. Subjects received SOF/VEL and ARV regimens (EVG/COBI/FTC/TDF, FTC/TDF+DRV/r, FTC/TDF+ATV/r, FTC/TDF+LPV/r, and EVG/COBI/FTC/TAF) alone and in combination. Steady-state plasma concentrations of SOF, its predominant circulating nucleoside metabolite GS-331007, VEL, and ARVs were analyzed on the last day of dosing for each treatment and PK parameters were calculated. Geometric least-squares means ratios and 90% confidence intervals (combination vs. alone) for SOF, GS-331007, VEL, and ARV AUCtau, Cmax and Ctau were estimated and compared against lack of PK alteration boundaries of 70-143% for all analytes.
Interestingly, the impact of boosted ARVs on VEL PK parameters was different for the various drugs tested. Figure 20 summarizes the effect of the respective HIV ARVs on VEL PK.
Figure 20: Effect of boosted HIV ARVs on Velpatasvir pharmacokinetics
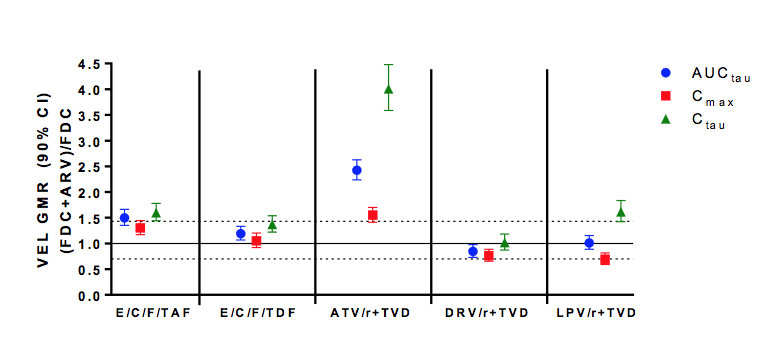
The highest increase in Ctau was seen in patients receiving atazanavir. Some ARVs had almost no change (darunavir/r) and some only a minimal increase in VEL exposure. Changes in VEL exposure were explained by being a substrate of P-gp, BCRP, OATP, CYP2B6, CYP2C8, and CYP3A4. Based on exposure-safety and -efficacy relationships established in Phase 3 SOF/VEL studies, changes in VEL PK did not warrant dose adjustment. With regard to impact of VEL on the boosted ARVs no significant effects of SOF/VEL on EVG, ATV, RTV, DRV, or LPV were noted. There was however an increase in COBI Ctau which however is not expected to introduce further drug interaction potential. Finally, there was also an analysis on the impact of VEL on tenofovir /TDF) or tenofovir alafenamide (TAF) exposure which was very interesting as the NS5A inhibitors ledipasvir has been reported to increase tenofovir exposure particularly in combination with a boosted PI which has led to a restriction in the label for concomitant use. Figure 21 shows the impact of SOF/VEL on tenofovir exposure for the various ARV regimens tested.
Figure 21: Effect of SOF/VEL on TAF and TFV PK
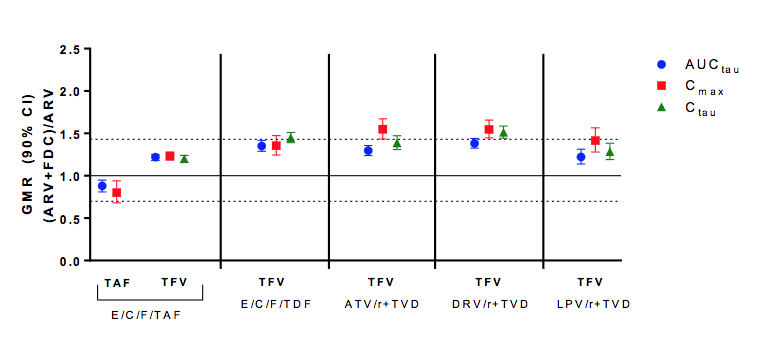
Overall, no significant impact of SOF/VEL on TAF or TFV derived from TAF was found similarly to what has been shown for Ledipasvir/sofosbuvir and TAF. This is important as in case there is a patient with an already reduced GFR or a greater risk for tenofovir induced renal toxicity where higher tenofovir exposure at best should be avoided, TAF would be ideal to use in order to have no change in tenofovir exposure and thereby minimize the risk for renal toxicity. A modest increase in TFV exposure (~20-40%) was observed when administered as TDF (plus PI/r) in presence of SOF/VEL. The mechanism for these slight increases is most likely inhibition of efflux transport (e.g., P-gp). No impact of SOF/VEL on FTC exposure (AUC GMR range 100-105%) was noted.
In summary, VEL/SOF can be safely co-administered with cobicistat boosted Elvitegravir/FTC/TDF or cobicistat boosted Elvitegravir/FTC/TAF as well as ritonavir boosted atazanavir, darunavir or lopinavir. Tenofovir levels do increase 20-30% when boosted PI + TDF/FTC are co-administered with VEL/SOF.
CROI: Drug Interaction Studies Between Sofosbuvir/Velpatasvir and Boosted HIV ARV Regimens - (02/24/16)
In clinical practice there have been concerns about the increased tenofovir levels seen with ledipasvir/sofosbuvir particularly in combination with boosted PI regimens where tenofovir levels are already higher than in non-boosted ARV regimens. Interestingly, one poster presented at CROI from Spain evaluated the renal safety of 12 or 24 weeks of Sofosbuvir/Ledispasvir (SOF/LDV), according to boosted TDF exposure, in a cohort of 159 HIV/HCV co-infected patients at a tertiary center in Madrid (17). In this "real life" population, SOF/LDV did not significantly worsen renal function in patients on "boosted TDF" (EVG/COBI/FTC/TDF or TDF with a RTV-boosted HIV PI). Figure 22 shows the distribution of CKD-EPI eGFR (ml/min) at baseline and at the end of treatment for the TDF no-boosted and TDF-boosted ARV regimens.
Figure 22: GRF changes for TDF non-boosted and TDF-boosted ARV regimens

Overall, more patients on a TDF plus boosted ARV regimen developed a GFR < 70ml/min than on TDF non-boosted ARV regimens but discontinuations of therapy due to renal adverse events were very rare.
Further interesting PK data were presented for the new DAA combination in development of ABT-493 and ABT530 (18). ABT-493 is a pangenotypic protease inhibitor discovered by AbbVie and Enanta and ABT-530 is a new NS5A inhibitor, both being currently developed as a combination regimen for treatment of chronic hepatitis C virus genotype 1-6 infection.
Phase 1 drug-drug interaction (DDI) studies were conducted to evaluate pharmacokinetics, tolerability, and safety of ABT-493 + ABT-530 co-administered with rilpivirine or raltegravir.
Both rilpivirine and raltegravir DDI studies utilized open label, randomized, multiple-dose, non-fasting study designs. In each study, healthy adult subjects received ABT-493 300 mg QD + ABT-530 120 mg QD and rilpivirine 25 mg QD (N=24) or raltegravir 400 mg BID (N=12) alone or in combination. Intensive pharmacokinetic assessments were performed for ABT-493, ABT-530, rilpivirine, and raltegravir on multiple days throughout the study. For ABT-493 and ABT-530 as victims of drug-drug interactions, exposures of both DAAs were minimally affected (≤13% change in exposures) when co-administered with rilpivirine or raltegravir. Compared to raltegravir alone, co-administration with the DAAs increased raltegravir Cmax and AUC12 central values by 34% and 47%, respectively, and C12 central values by 164%, compared to administration of raltegravir alone. Compared to rilpivirine alone, co-administration with ABT-493 + ABT-530, increased rilpivirine Cmax, AUC24, and C24 by 105%, 84%, and 77%, respectively. Despite the considerable increases in rilpivirine and raltegravir exposure however, consistent with rilpivirine and raltegravir label recommendations for coadministration with other drugs that caused similar magnitude of increase in rilpivirine and raltegravir exposures, no dose adjustment is needed when ABT-493 and ABT-530 are co-administered with rilpivirine or raltegravir.
CROI: Interactions Between ABT-493 plus ABT-530 combination and Rilpivirine or Raltegravir - (03/01/16)
Summary
⋅ 12 weeks of Sofosbuvir + ribavirin for treatment of acute HCV Ledipasvir/sofosbuvir only led to a disappointing 59% SVR12 rate; lower ribavirin levels were associated with a higher probability of relapse.
⋅ Treatment with LDV/SOF for 6 weeks resulted in a 77% SVR12 rate in HIV-infected patients with acute HCV infection No relapse among patients with a baseline HCV RNA ≤6.9 log10 IU/mL. Reinfection in 1/26 patients followed through post-treatment Week 12 and underlines the need for reinfection counselling. Treatment with LDV/SOF for 6 weeks was well tolerated.
⋅ In the German GECCO cohort real-life DAA-based treatment regimens were highly effective in HCV-mono- as well as HIV-HCV-coinfected patients. Relapse occurred in only 4% of the patients. All DAA combinations were generally well tolerated. SOF/LDV for 8 weeks appeared highly effective in selected patients in this population.
⋅ Treatment of large numbers of patients with effective therapy is the only option for control of HCV in Egypt. Political will and dedication on all levels of medical care have led to an impressive start with almost 200.000 individuals already having received HCV therapy with response rates being similar to real life data from Europe/US.
⋅ In a French real-life cohort the prevalence of NS5A RAVs at baseline varied considerably across genotypes 1a, 1b, 3 and 4. Virological failure tended to be more frequent when an NS5A Y93H substitution was present at baseline.
⋅ HCV NS5A RAVs do not predict relapse when using sofosbuvir-based treatment for at least 6 weeks or during re-treatment with standard 12 week regimens of LDV/SOF. RAVs do appear to impact treatment response when treating with ultra-short, 4 week duration therapies.
⋅ In general, analysis of RAV impact on treatment outcome is influenced by choice of analysis parameters, such as: Separation by subtype, i.e. GT1a or pooled GT1a/GT1b, threshold for detection of variants, i.e. 1-20% sensitivity cut-off and variants included in the analysis.
⋅ Ledipasvir/sofosbuvir with ribavirin for 24 weeks in genotype 1-infected, NS5A-experienced HCV/HIV co-infected patients was safe and highly successful. Obviously, extending treatment duration and adding ribavirin can help to achieve SVR despite prior failure on the same DAAs and for the majority development of NS5A RAVs upon relapse.
⋅ Among treatment-naive people without cirrhosis, 100% taking ravidasvir/sofosbuvir (RDV/SOF) and 98% taking RDV/SOF plus ribavirin attained SVR12 in intention-to-treat analyses Among interferon-experienced people without cirrhosis, SVR12 rates were 95% with RDV/SOF and 100% with RDV/SOF plus ribavirin. Nobody without cirrhosis had a treatment relapse.
⋅ VEL/SOF can be safely co-administered with cobicistat boosted Elvitegravir/FTC/TDF or cobicistat boosted Elvitegravir/FTC/TAF as well as ritonavir boosted atazanavir, darunavir or lopinavir. Tenofovir levels do increase 20-30% when boosted PI + TDF/FTC is co-administered with VEL/SOF.
⋅ No dose adjustment is needed when ABT-493 and ABT-530 are co-administered with rilpivirine or raltegravir.
References
1. Naggie S et al.: Sofosbuvir plus ribavirin without interferon for treatment of acute hepatitis C virus infection in HIV-1.infected Individuals (SWIFT-C). 66th Annual Meeting of the American Association for the Study of Liver diseases, November 13-17, 2015, San Francisco, USA; abstract 1094
2. Martinello M et al. Sofosbuvir and ribavirin for six weeks is not effective among people with acute and recently acquired HCV infection: The DARE-C II Study. 66th Annual Meeting of the American Association for the Study of Liver diseases, November 7-11, 2015, San Francisco, USA; abstract 1083
3. Fierer D et al. Sofosbuvir in the treatment of acute HCV infection in HIV-infected men. 66th Annual Meeting of the American Association for the Study of Liver diseases, November 13-17, 2015, San Francisco, USA; abstract 1090
4. MacBrayne CE et al.: Lower Ribavirin Exposures in HIV+ Patients That Relapsed to Acute HCV treatment. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 99
5. Basu P et al.: Sofosbuvir and ledipasvir versus sofosbuvir and simeprevir combination therapy in the management of acute hepatitis C: A randomized open label prospective clinical pilot study. SLAM C study. Interim data. 66th Annual Meeting of the American Association for the Study of Liver diseases, November 13-17, 2015, San Francisco, USA; abstract 1074
6. Rockstroh JK et al.: Ledipasvir/Sofosbuvir for 6 Weeks in HIV-Infected Patients With Acute HCV Infection. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract LB154
7. Christensen S et al.: Directly Acting Agents Against HCV - Results From the German Hepatitis C Cohort (GECCO) 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 584
8. Wyles DL, et al.: ALLY-2 Investigators. Daclatasvir plus Sofosbuvir for HCV in Patients Coinfected with HIV-1. N Engl J Med. 2015 Aug 20;373(8):714-25.
9. Piroth L et al. for the ANRS CO13 HEPAVIH Study Group. Response to DAA-Based Regimens in HIV-HCV Coinfected Patients in Real Life, France. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 582
10. El-Sayed M. Hepatitis C in Egypt: A National Approach to Treatment. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; Symposium Global Impact of Hepatitis Symposium, abstract 65
11. Fourati S et al.: HCV Resistance to Daclatasvir/Sofosbuvir Across Different Genotypes in the Real Life. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 577
12. Wilson EM et al.: Resistance Associated Variants: Data From the NIAID SYNERGY Trial. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 580
13. Sulkowski M et al.: Effect of Baseline Resistance-Associated Variants on SVR with the 3D Regimen Plus RBV. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 539LB
14. Cooper C et al.: Retreatment of HCV/HIV-Coinfected Patients Who Failed 12 Weeks of LDV/SOF. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 573
15. Waked I et al.: High Response Rate in HCV-Genotype 4 Patients Treated With Ravidasvir and Sofosbuvir. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 153
16. Mogalian E et al.: Drug Interaction Studies Between Sofosbuvir/Velpatasvir and
Boosted HIV ARV Regimens. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 100
17. Vivancos-Gallego MJ et al.:Real-Life Renal Safety of Boosted TDF in HIV/HCV Patients on SOF/LDV. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 452
18. Oberoi RK et al.: Interactions between ABT-493 plus ABT-530 combination and Rilpivirine or Raltegravir. 23rd Conference on Retroviruses and Opportunistic Infections, February 22-25, 2016, Boston; abstract 453
|
| |
|
 |
 |
|
|