| |
Cocrystal Pharma Updates New HCV Drugs Development -
|
| |
| |
in announcement:
Announces Filing 2016 Second Quarter Financial Statements and Provides Company Update
http://ir.cocrystalpharma.com/press-releases/detail/27
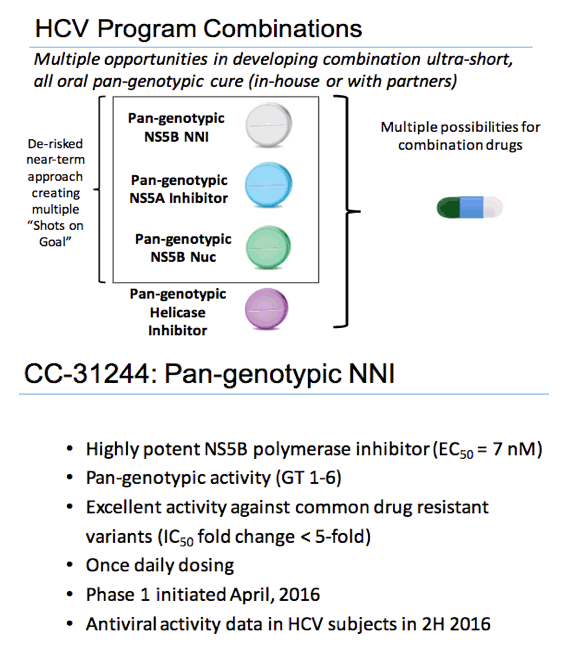
During the last three months, the Company focused on its research and development efforts and commenced clinical trials for our Non-Nucleoside HCV Polymerase Inhibitor (NNI) CC-31244.
⋅ Hepatitis C -- CC-31244. Our first in-human study began in April, 2016. This is a Phase 1a safety study. Four of the five dose groups have completed and dosing in the final group has commenced.
⋅ Hepatitis C -- CC-2850. Our former back-up nucleoside inhibitor, CC-2850, has a better pre-clinical profile than our previous lead candidate, CC-1845. Therefore, we have initiated scaling-up synthesis of CC-2850 for further preclinical evaluation.
⋅ Hepatitis C -- CC-2069. We have obtained additional preclinical in vitro data for CC-2069, our NS5A pan-genotypic inhibitor. We have initiated non-GMP and GMP synthesis of CC-2069.

|
|
| |
| |
|
|
|