| |
Can Five HIV Prevention Strategies Bring the Epidemic to Its Knees?
|
| |
| |
Download the PDF here
Mark Mascolini

Hard thinking, hard work, and hard numbers have yielded the tools to stop the HIV epidemic.1 Now. But hard policy choices, hard funding strictures, and plain hard luck may mean the epidemic will linger for decades with only a modest dip in HIV incidence. And even if policy choices all favor AIDS care and funding pipelines gush ample cash, the End of AIDS may remain a distant prospect.
Why the gloomy forecast? Large, long trials in diverse populations establish past quibbling that biomedical strategies like voluntary medical male circumcision, preexposure prophylaxis (PrEP), antiretrovirals to prevent mother-to-child transmission (PMTCT) of HIV, and prompt antiretroviral therapy (ART) slash the risk of picking up or passing on HIV. The antiretroviral interventions-when practiced faithfully-offer virtually airtight protection from HIV. Circumcision trims HIV risk 60% in heterosexual men, and condoms protect both men and women, straight and gay, at even higher rates.
With five precision tools in hand, you would think the epidemic could be hammered into submission in a decade or two. But that probably won't happen-even if governments across the globe decide they want these tools in health workers' kits today and pay to put them there. Even in a utopian political and funding climate, at least three things have to happen to make a tool cut HIV risk substantially (Figure 1): (1) The intervention must work well. (2) The intervention must be adopted by a big majority of people with or at risk for HIV, and adopters have to stick with the intervention. (3) If adoption of the intervention falls short or wanes with time, tactics to change those trends must be devised and deployed.
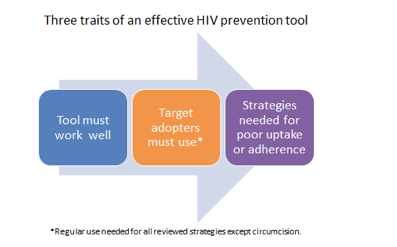
Figure 1. To have a meaningful impact on HIV incidence, a prevention strategy must be highly effective, must be adopted and used faithfully by a high proportion of at-risk people, and must be backed by strategies that address poor uptake or adherence.
This analysis completely skirts the hugely entrammeled questions of policy and funding and asks three questions about five HIV prevention tools: Does it work? Do people use it? And can use be improved? The tools are condom use, circumcision, PrEP, PMTCT, and Test-and-Treat with ART.
The Not-So-Impenetrable Condom
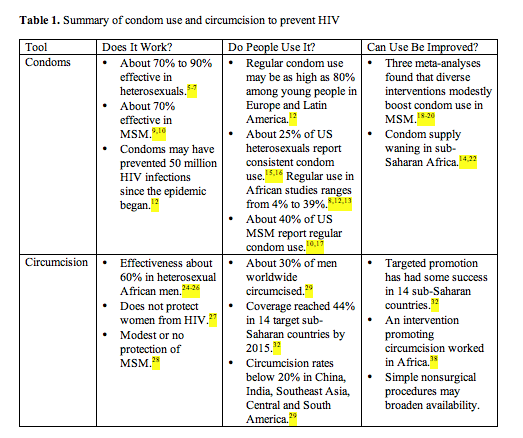
If used properly and if they don't break, condoms provide a nearly impenetrable barrier to HIV.2 As part of the ABC approach to HIV prevention (abstain, be faithful, use condoms), condoms may have contributed to dwindling HIV rates in Uganda and Botswana.3,4 Analysis of HIV seroconversion studies published from 1987 to 1992 concluded that consistent condom use virtually eliminated HIV risk at a protection rate of 90% to 95% during penile-vaginal sex (Table 1).5 A 2002 Cochrane Database meta-analysis involving mostly partners of people infected with blood products figured that consistent condom use cut HIV incidence 80% in heterosexuals.6 A 2015 meta-analysis of 25 studies involving 10,676 HIV-discordant heterosexual couples set the protection rate somewhat lower with consistent condom use, at 71%.7 In 2001 a 30-month study of 17,264 sexually active heterosexuals in Rakai, Uganda, a region with high HIV prevalence, determined that consistent condom use curbed HIV incidence 63% while inconsistent use offered no protection.8
In a Multicenter AIDS Cohort Study of 2915 gays and other men who have sex with men (MSM) in the late 1980s, always using a condom cut HIV risk 71% compared with never using a condom.9 CDC analysis of 3490 MSM in two 1998-2001 trials linked consistent condom use to a 70% lower HIV risk in men reporting any anal sex.10 But a modeling study of Ontario MSM painted a grimmer picture of condom reliability in MSM.11 The researchers estimated a much higher number of anal sex encounters with a condom than without a condom in 2009 (1,184,343 versus 117,133). But of the 693 new HIV infections recorded in 2009, 51% involved men having anal sex with a condom, 33% involved men having anal sex without a condom, and 16% involved men having oral sex. The Ontario team proposed that "the proportion of HIV infections related to condom failure appears substantial and higher than previously thought."11 And the 51% transmission rate may be an underestimate because the model used a higher estimate of condom effectiveness (87%) rather than the 70% estimate from the recent CDC study.10
As unprepossessing as the Ontario modeling estimates may be,11 they do offer insight into why purportedly impenetrable condoms may in fact allow HIV transmission even if worn consistently. Condoms break, condoms leak, and condoms slip, the Ontario team observed. Condoms donned after initial penetration but before ejaculation may give HIV-laden semen some wiggle room. And of course a zone of uncertainly surrounds a study participant's recollection of "consistent condom use" and what really happened. Yet together, all these studies and meta-analyses5-11 give a believable answer to the first critical question, does it work? Yes, about 75% of the time, because HIV appears to slip from a condom-clad penis to anus or vagina about once per four penetrations.
A 75% effective intervention cannot by itself end the HIV epidemic, but it could bring HIV to its knees-if lots of HIV-positive and at-risk people use it regularly. Do they? The World Health Organization (WHO) and UNAIDS say 80% of 15- to 24-year-olds in some European and Latin American countries report using condoms the last time they had sex, but fewer than 30% in some West African countries claim they wear condoms.12 Among sexually active adults starting HIV care at two urban clinics in Uganda, 39% said they always used condoms.13 But in the general-population rural Rakai cohort in Uganda, where HIV rates run high, only 4.4% of sexually active 15- to 59-year-olds reported consistent condom use.8 In its 2013 Global Report, UNAIDS charted falling condom use in Côte d'Ivoire, Niger, Senegal, and Uganda.14
In heterosexual adults attending a Chicago sexually transmitted disease (STD) clinic and judged to run a high risk of HIV infection, 84% rated themselves at low or no risk and fewer than 20% consistently wore condoms.15 In a nationally representative sample of 1645 sexually active heterosexuals in the US National Survey of Sexual Health and Behavior, only 27.5% of men and 22% of women used a condom the last time they had sex.16 In the 1998-2001 CDC study of 3490 high-risk MSM enrolled in two US trials, 40% reported never using condoms for receptive or insertive anal sex in the past 6 months.10 And a 2014 Kaiser Foundation survey found that fewer than half of US MSM (46%) said they wore condoms all or most of the time during sex, while 16% used condoms only some of the time, 14% rarely, and 24% never.17
Although the WHO and UNAIDS suggest 80% of young Latin Americans and Europeans usually put on a condom for sex, the other studies indicate that fewer than half of Africans and US residents-sometimes far fewer than half-rely on condoms to protect themselves and their sex partners from HIV and other STDs. A 75% effective intervention used by fewer than half of at-risk people can have only a modest impact on the HIV epidemic.
Can condom use be improved? Three large meta-analyses-one of them in China-figured that diverse interventions can at least modestly cut rates of condom-free sex, but all three meta-analyses involved MSM, usually HIV-negative MSM.18-20 Two mathematical models credited wider condom use with a drop in HIV incidence among 15- to 49-year-olds in South Africa's largely heterosexual epidemic from 2000 through 2007.21 Condom use accounted for about 30% of declining HIV incidence in both models, while antiretroviral therapy had a smaller impact during these earlier years of the antiretroviral rollout. But condom supply seems to be waning in some countries battling HIV. UNAIDS warned that donors trimmed supplies of both male and female condoms after 2011.14 Even in 2011, UNAIDS figured a condom supply of only 9 condoms for every man per year in sub-Saharan Africa and just 1 female condom yearly for every 10 sub-Saharan women.22
A modeling study presented in 2014 reckoned that condoms have spared 50 million people from HIV infection since the epidemic began.12 That's more than the estimated 37 million people living with HIV today.23 But condoms alone could not stop HIV in the years before antiretroviral therapy because they are not 100% effective and not enough people use them. Whether they continue to limit the epidemic depends on aggressive promotion and provision of this inexpensive tool.
Circumcision: Limited Effect and Reach?
Three similar and rigorous randomized trials proved that voluntary medical male circumcision cuts the risk of HIV infection in heterosexual men about 60% (Table 1).24-26 Male circumcision does not shield a circumcised man's female sex partner from the retrovirus,27 and this simple surgery offers modest or no protection to MSM.28 The 60% protective effect in heterosexual men falls short of the 70% to 90% impact of consistent condom use. But circumcision has a big advantage over condoms and all other HIV-bridling strategies: One procedure lasts for life.
In 2007, just after results of three circumcision trials became known,24-26 UNAIDS estimated that only 30% of men across the world had their foreskin clipped.29 Circumcision rates measured only about 15% in some sub-Saharan countries (Botswana, Burundi, Namibia, Swaziland, Zambia, and Zimbabwe) but higher in others, especially those with large Muslim populations. In 2007 circumcision rates lay below 20% in most of China, India, Southeast Asia, and Central and South America.29
By 2011, UNAIDS reported slow early uptake of circumcision in 13 sub-Saharan target countries.30 Eight of the 13 countries counted only about 100,000 circumcised men by 2009. A year later those eight countries had more than 350,000 circumcised men, but tens of millions remained uncircumcised. By 2015 UNAIDS tallied 10 million circumcised men in 14 sub-Saharan countries-up from 1.5 million in 2011.31
Of course those 10 million still make up a minority of sexually active men at risk in countries with high HIV rates. In 2015 the WHO estimated 44% coverage in 14 sub-Saharan countries based on the 20.8 million circumcisions needed to reach 80% coverage.32 The WHO figures that 80% coverage would spare 3.4 million men from HIV infection by 2025. Not coincidentally, however, modeling in southern Africa indicates that men most likely to be circumcised are those who already run a lower risk of HIV infection, including men with more education, white-collar workers, and the richest quintile.33
Circumcision prevalence remains low in other parts of the world struggling with HIV. In 2012 Chinese health authorities calculated that, aside from two Muslim minorities, fewer than 3% of male Chinese migrant workers were circumcised.34 A 2014 cross-sectional survey of 570 MSM in China found only 18% circumcised, though more than half of uncircumcised men said they would consider the procedure.35 A 2010 report of heterosexual Thai men at high risk for HIV found only 12% circumcised.36 A 2008-2009 study of 551 Thai MSM and transgender persons figured that only 8% had been circumcised.37 Only 30% of these men were willing to participate in circumcision trials, compared with 86% interested in PrEP trials and 70% willing to consider HIV vaccine trials.
A 2012-2014 cluster-randomized trial showed that an intervention can encourage HIV-negative African men to get circumcised.38 And the success of targeted circumcision promotion in 14 African countries31,32 demonstrates that men can be persuaded to undergo the operation. Simpler, cheaper circumcision procedures like PrePex and ShangRing may expand the volume of men who can be circumcised and encourage more men to forsake their foreskin. But the relatively modest 60% effectiveness of circumcision in heterosexual men coupled with the huge number of at-risk uncircumcised men worldwide argue that this HIV prevention tool will make only a small contribution to global HIV control.
PrEP Works But Still Sees Little Use
Unlike circumcision, which reliably shields only heterosexual men from HIV,24-28 PrEP protects anyone who takes it faithfully-heterosexual men and women,39,40 MSM,41,42 and people who pick up HIV when injecting drugs (Table 2).43 Among trial participants who took coformulated tenofovir/emtricitabine (TDF/FTC) regularly in these placebo-controlled studies, PrEP cut HIV risk up to 92% in MSM,41 up to 90% in heterosexual couples,39 and up to 73.5% in people who inject drugs.43
Also unlike circumcision-which lasts for life after a single procedure-the PrEP pills available today require regular dosing. Just how regular remains an open question. US PrEP guidelines call for daily dosing with TDF/FTC.44 Researchers who ran the iPrEx PrEP trial in MSM used data from that study and another to calculate that four TDF/FTC doses weekly can cut HIV risk 96%, compared with 99% with daily dosing.45 The IPERGAY trial in French and Canadian MSM achieved 86% protection with two TDF/FTC doses before sex and two after sex.42
But the as-needed dosing approach in IPERGAY remains unverified in heterosexual men and women-and it may not work in a large portion of MSM. HPTN 067/ADAPT is testing three TDF/FTC PrEP dosing patterns in (1) MSM in Bangkok, (2) MSM in Harlem (New York City), and (3) women in Cape Town.46 Researchers randomized them to (1) daily dosing, (2) twice-weekly dosing plus one dose after sex, or (3) one dose before sex and two after sex. In all three study groups, adherence proved best with daily dosing, fell off with twice-weekly dosing, and dwindled further with before-and-after-sex dosing. Protection from HIV accordingly proved best with once-daily dosing (85% in Bangkok, 66% in Harlem, 75% in Cape Town) and lower with the other two strategies (84% and 74% in Bangkok, 47% and 52% in Harlem, and 56% and 52% in Cape Town).
For now, US guidelines specifically advise against as-needed dosing.44 But dosing frequency will drop greatly if ongoing trials find that monthly or quarterly injectable antiretroviral nanosuspensions protect people from HIV. Agents under study include the investigational integrase inhibitor cabotegravir,47-49 the nonnucleoside rilpivirine (possibly combined with cabotegravir),48,49 and the CCR5 antagonist maraviroc.49 Other long-acting strategies that could improve adoption and simplify adherence involve vaginal rings and subdermal implants.
Something will have to change before PrEP helps curb the HIV epidemic because early uptake of TDF/FTC PrEP looks less than overwhelming. UNAIDS told the New York Times in October 2015 that an estimated 20,000 people in the United States use TDF/FTC PrEP, along with fewer than 10,000 people in other countries.50 The estimated 20,000 US residents on PrEP represent fewer than 2% of the 1.2 million Americans the CDC calculates could benefit from PrEP today.51 Only three countries in Central and South American have PrEP access through demonstration projects, as do eight in Africa and two in Asia.52 Other countries in those regions have no formal access to PrEP, but motivated providers can prescribe TDF/FTC for PrEP.
Among recently published reports, one in a convenience sample of high-risk MSM in Washington State appears to reflect the biggest gain in PrEP adoption, rising from 5% in 2012 to 31% in 2015.53 Uptake and interest are lower among MSM in San Francisco, where PrEP is free (estimated 15% uptake50), San Diego (0.5% uptake among 416 high-risk MSM offered PrEP54), or Amsterdam (13% of 488 MSM intended to use PrEP55). And at least in mid-2013, few infectious disease providers in the United States had prescribed PrEP: Among 573 survey respondents, 74% supported prescribing PrEP, but only 9% had done so.56 Another 43% claimed they plan to prescribe PrEP.
Modeling studies in Botswana, the United States, and Australia indicate that wider adoption of PrEP in these countries would have a meaningful but moderate impact on the epidemics. With a robust antiretroviral rollout, Botswana has already reached viral suppression levels equivalent to US national goals for 2020. Adding TDF/FTC PrEP to Botswana's prevention program may reduce new HIV infections by 39% in women and 40% in men over 10 years.57 If 20% of all US MSM 13 to 64 years old started TDF/FTC PrEP-which would seem the high end of uptake expectations based on current findings-PrEP could cut the new-HIV rate by 13% over 20 years.58 This estimate is probably low, though, because the model uses the 44% effectiveness rate found among all MSM (faithful users and nonusers) in iPrEx.41 If 50% of only high-risk MSM started PrEP (a highly optimistic adoption rate), HIV incidence could drop 29% over 20 years (using the 44% effectiveness rate). TDF/FTC PrEP use by 30% of the general MSM population in New South Wales, Australia may cut the new infection rate 30% over 10 years.59 This analysis figured PrEP effectiveness of 95% against wild-type virus and 40% against drug-resistant virus.
Everyone would welcome a 30% to 40% drop in HIV incidence over the next 10 or 20 years. But that impact depends on higher PrEP adoption and consistent use-both highly optimistic scenarios. A one-third drop in HIV incidence would match the one third overall decline calculated by UNAIDS across the world from 2000 to 2015.23 But even this sanguine outcome, by itself, would leave more than a million new global HIV infections yearly at current rates.

Test-and-Treat Hobbled by Testing Gap
Before everyone could agree that antiretroviral therapy (ART) can control chronic HIV infection, US and French researchers proved that it can prevent HIV transmission. Early in 1994 PACTG 076 investigators reported that zidovudine taken by mothers and their infants sliced the risk of mother-to-child transmission by two thirds.63 More recent randomized trials showed that triple ART virtually eliminates the risk of HIV transmission to infants, at protection rates of 95% or higher (Table 3).64-66 Then the randomized HPTN 052 trial confirmed that immediate ART and viral control for an HIV-positive person nearly abolishes the risk of infecting a sex partner.67 As powerful as antiretrovirals are in shielding an HIV-negative person taking PrEP, they seem even stronger in protecting infants and sex partners of HIV-positive people.
Results of studies testing ART to prevent mother-to-child transmission of HIV led the WHO to affirm that such transmission "can be nearly fully prevented if both the mother and the child are provided with ARV drugs throughout the stages when infection could occur."23 The power of ART to prevent AIDS, forestall serious non-AIDS diseases, prolong life, and prevent transmission during sex, conception, or breast feeding lay behind the WHO's decision to recommend treatment for everyone with HIV, whatever their CD4 count.62 The WHO's antiretroviral options A, B, and B+ to block mother-to-child transmission went out the window with this decision to urge immediate and lifelong therapy not just for mothers with HIV, but for all.
And as evidence of ART's benefits accrues, estimates of wider use and its epidemic-choking impact climb. The WHO reckons that in 2014 three quarters of 1.5 million pregnant women with HIV across the world got ART to stymie HIV transmission.23 The global health group figured that 36.9 million people had HIV by the end of 2014, and by mid-2015 about 15.8 million had started ART. The resulting proportion of treated people-43%-falls far short of the 90% target.68 But it represents a huge improvement over the tiny percentage of people treated only 10 or 15 years ago.
A nationwide 2000-2011 study in France determined that no woman who started ART before becoming pregnant, continued treatment through delivery, and maintained a viral load below 50 copies/mL transmitted HIV to her newborn.69 Across the globe, the WHO estimates that new HIV infections in children plunged 48% from 2009 to 2014, thanks largely to ART.70 Total new HIV diagnoses fell by about half as ART uptake ballooned in British Columbia (1996-2012)71 and San Francisco (2004-2008).72. Yet despite widening ART use and falling viral loads across the United States,73 HIV incidence in the country remains stuck at 40,000 new cases yearly.74 Around the world the WHO figures that new HIV infections dropped 35% from 2000 to 2015, but 2 million people-including 170,000 children-still got infected in 2014.23,75 These dismaying statistics show that Test-and-Treat needs sterner application to stop the epidemic.
One glaring shortcoming in global HIV prevention probably explains why burgeoning ART uptake has not contained the epidemic faster: The "test" half of the Test-and-Treat equation remains largely unfulfilled. Millions of HIV-positive people never had an HIV test, don't know they're infected, continue having sex with HIV-negative partners, and keep having babies. The WHO figures that only 53% of infected people around the world know they carry HIV.23 Even in the United States, where the CDC has pushed hard for universal adult HIV testing for a decade, an estimated 13% of HIV-positive people (156,300) remain undiagnosed.76 The crippling toll of undiagnosed HIV on attempts to contain the epidemic came clear in an innovative ATHENA Cohort study in the Netherlands.77 Researchers used HIV sequence data and individual clinical findings from 617 recently infected MSM to determine how these men picked up HIV. A hefty 71% got infected by still-undiagnosed men.
Giving ART to everyone diagnosed with HIV will never rein in the epidemic as long as such mammoth proportions of infected people remain ignorant of their serostatus. The ambitious 90-90-90 goals from UNAIDS aim to test 90% of all HIV-positive people, to treat 90% of those diagnosed, and to make viral loads undetectable in 90% of those treated.68 But much work remains before the world merely approaches those targets. A CDC team observes that wide point-of-care testing in regions hard hit by HIV "has not been fully exploited" because of challenges in rolling out the technology and bolstering quality assurance.78 WHO analyses of HIV testing data project a 35% to 45% jump in uptake of rapid testing in low- and middle-income countries, but the WHO team figures that increase will probably leave these countries short of the 90% testing target.79

With so much room to improve HIV testing uptake, ideas on how to do so abound. Among the most promising are community-based testing and rapid testing. Meta-analysis of 21 studies involving home-based HIV testing in sub-Saharan Africa found that 58.1% to 99.8% of 474,377 participants accepted testing, and a pooled proportion of 76.7% of individuals received test results.80 The researchers concluded that home-based testing "could be a valuable tool for treatment and prevention efforts." Meta-analysis of 117 studies involving 864,651 people tied community-based HIV testing to more than a 10-fold jump in testing uptake.81 The approaches assessed included door-to-door testing, mobile testing, self-testing, and church- and school-based testing. A Cochrane systematic review and meta-analysis of 13 studies-in sub-Saharan Africa, the United States, and Australia-linked rapid HIV testing to a 3-fold increase in HIV testing uptake and a 2-fold leap in receiving test results.82 The studies involved people in diverse settings, including homes, primary care clinics, antenatal clinics, TB programs, emergency departments, bathhouses, and prisons. The analysis linked 3 years of community-based testing to an 11% drop in HIV incidence.
But even if testing reaches millions more at-risk people, challenges to Test-and-Treat will persist. Researchers who conducted the HPTN 052 trial observe that diagnosing people in the early stages of HIV infection-when transmission risk is highest-will remain difficult.83 They note that the trial did not establish the value of immediate ART in MSM and drug injectors, though one suspects the same prevention principles apply in those populations because of declining HIV incidence in San Francisco72 and British Columbia,71 which have large MSM and drug-injecting populations and have put the Test-and-Treat formula into practice. Finally, the HPTN 052 team cautions that "the full benefits of this strategy will probably need universal access to very early ART and excellent adherence to treatment."83 Developing an injectable antiretroviral combination that could be given every 1 or 2 months84 would vastly simplify ART delivery, improve adherence, and enhance prospects for treatment as prevention.
Modeling studies differ greatly in projecting how treatment-as-prevention will affect the HIV epidemic. Three of nine models positing 95% ART access in South Africa and 95% retention in care predicted that HIV incidence would dwindle below 0.1% yearly by 205085-an epidemic-squelching outcome by any standard. But overall analysis of models including immediate ART for the newly diagnosed showed that projected impacts "varied widely across models, maintaining substantial uncertainty about the theoretical prospect for elimination of HIV from the population using ART alone over the next four decades."85 As wags observe, modeling means not having to say you're certain, and that dictum certainly holds true when modeling the end of the HIV epidemic.
Combining Prevention Tools to Stop HIV
Strategies that thwart HIV transmission have proved remarkably effective in big clinical trials and teeming cohort studies. Three antiretroviral tools-PrEP, treatment to prevent mother-to-child transmission (PMTCT), and rapid diagnosis and treatment of chronic infection (Test-and-Treat)-can prevent 90% or more new HIV infections. But the two most widely deployed ART strategies-PMTCT and Test-and-Treat-remain woefully underused because so many HIV-positive people remain untested.
UNAIDS recognized the primacy of expanded HIV testing in stopping the epidemic by setting the daunting target of diagnosing 90% of HIV-positive people as the first step in its 90-90-90 campaign.68 But even if millions more infected people learn they carry HIV, no single strategy will stifle the epidemic on its own because some people will always resist that strategy. Recognizing these principles leads to a simple two-step approach to limiting the epidemic with the tools in hand:
1. Expand HIV testing to reach a large majority of infected people.
2. Combine two or more prevention strategies (including at least one antiretroviral strategy).
Several published studies have sized up prevention combinations, each including some antiretroviral intervention. A modeling study assessed the impact of condoms and TDF/FTC PrEP, separately or together, on HIV incidence in African-American MSM,86 the population with the highest HIV incidence in the United States. In a hypothetical cohort of 10,000 black MSM who never use PrEP, 323 men who always use condoms will pick up HIV. Among 10,000 MSM who never use condoms, 295 with 90% PrEP adherence will get HIV infection. Among 10,000 MSM who consistently use PrEP and condoms, 87 will acquire HIV, a rate indicating 92% prevention effectiveness for PrEP plus condoms.
Meta-analysis of 11 studies in China assessed the impact of consistent condom use and ART for positive partners in HIV-discordant heterosexual couples.87 Pooled HIV incidence stood at 0.92 per 100 person-years in HIV-negative partners with a spouse taking ART compared with 2.45 per 100 person-years in negative partners with an untreated spouse. An HIV-positive partner taking ART more than halved HIV incidence (incidence rate ratio [IRR] 0.47, 95% confidence interval [CI] 0.43 to 0.52). ART plus consistent condom use cut HIV incidence by two thirds (IRR 0.33, 95% CI 0.17 to 0.64).
Three models of HIV prevalence in sub-Saharan Africa predicted that increasing ART use alone would lower prevalence from less than 1% up to 2% over the next 20 years.88 Combining ART with TDF/FTC PrEP over the next two decades would cut HIV prevalence 1.2% in one model, 2.8% in another, and 4.4% in the third model. The ATHENA cohort analysis of 617 recently infected MSM used HIV sequence analysis and modeling to calculate that 61% of these new infections could have been prevented if (1) all at-risk MSM got tested for HIV annually, (2) all positive men started ART, and (3) half of all negative men started PrEP.77
A demonstration project involving 1000 HIV-discordant couples at the Kenyan and Ugandan sites of the Partners PrEP trial yielded data indicating that combining PrEP and ART slices HIV risk 96% (Table 4).89 In the demonstration project, HIV-negative partners started PrEP and HIV-positive partners started ART. Through 1.5 years of follow-up, 2 HIV-negative people (both with faulty PrEP adherence) became infected for an incidence of 0.2 per 100 person-years. In a comparable group of untreated people from the placebo arm of Partners PrEP trial, modeled HIV incidence stood at 5.2 per 100 person-years. Comparing the PrEP/ART group to the untreated group, the researchers calculated an IRR of 0.04 (95% CI 0.01 t 0.19), a rate meaning PrEP plus ART cut HIV incidence 96% in these African couples.
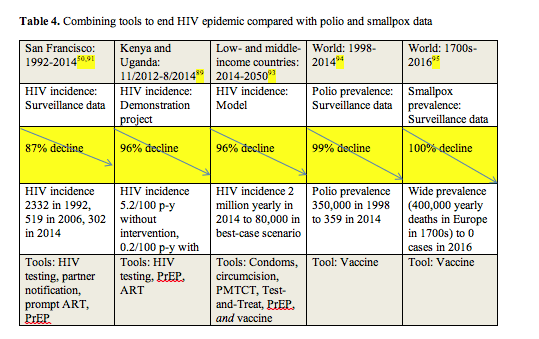
San Francisco embarked on a full-bore program to prevent new HIV infections in 2010, recommending ART for all with HIV 2 years before US health authorities took that step. The city aims to begin ART on the day a person gets diagnosed. San Francisco's public health team backs that strategy by promoting HIV testing, partner notification, linkage to or reengagement in care, and free PrEP.50,90,91 As a result, HIV incidence dropped from a higher of 2332 new infections in 1992 to 302 cases in 2014, an 87% plunge.
What impact would a San Francisco-like plan have if adopted across the United States-or across the world? The CDC estimates that 44,073 people in the United States got infected with HIV in 2014.74 If that number fell 87%, national HIV incidence would be 5729 yearly. The WHO calculates that 2 million people acquired HIV in 2014.23 If that number fell 87%, global HIV incidence would shrivel to 260,000 yearly.
Even a wealthy country like the United States faces a stiff challenge in applying a San Franciscan plan nationally. San Francisco has an estimated 82% of HIV-positive residents in care and 72% with an undetectable viral load.91 Across the United States in 2012, only 39% of HIV-positive people were in care and only 30% had an undetectable viral load.90 Globally, only a little more than half of HIV-positive people even know they carry the virus.23
Keys to improving the continuum of care are no secret. Under the auspices of IAPAC, an expert international committee sifted data from 1047 articles to formulate 36 evidence-based recommendations to hike HIV testing, linkage to and retention in care, ART coverage, ART adherence, and viral suppression.92 These steps are working in San Francisco. And they're working in much larger cities with marginalized populations, like New York. From 2006 to 2013, new HIV diagnoses ebbed 32% in New York City, thanks partly to a health department drive supporting individual entry to care and reentry of those who lost touch with their providers.90
What will it take to get global HIV incidence below the 260,000 yearly diagnoses attainable with an incidence drop similar to that seen in San Francisco50 (calculated just above)? A blue-ribbon panel suggests a way to get below a quarter million new infections yearly-but it won't be easy (Table 4).93 If global prevention efforts built on an ambitions baseline program including PMTCT, condom use, and male circumcision to include a Test-and-Treat initiative, highly effective PrEP, and an effective HIV vaccine, by 2050 we could count only 80,000 new HIV cases yearly in low- and middle-income countries under the most optimistic scenario and 270,000 new cases on the high end of that projection. Dropping from 2 million new infections yearly today to 80,000 would represent a 96% plummet. The baseline program plus Test-and-Treat alone would bring HIV incidence only to about 500,000 cases yearly by 2050. Reaching a global incidence of 80,000 new HIV infections yearly may seem miraculous, but it would still be an epidemic by most standards. For example, the WHO tallied 28,635 Ebola cases through January 3, 2016 in the recent West African outbreak.94
Data from cities like San Francisco, Vancouver, and even New York show that diligent application of HIV prevention tools in hand today can make a big dent in the epidemic. But even when combined, those tools cannot do what a polio vaccine did for that viral infection-lower prevalence by 99% since 1988 to 359 reported cases in 2014.95 And they cannot do what a smallpox vaccine did for that viral infection-make it go away.96
References
1. Fauci AS, Marston HD. Ending the HIV-AIDS pandemic-follow the science. N Engl J Med. 2015;373:2197-2199. Web site: http://www.ncbi.nlm.nih.gov/pubmed/26624554.
2. Carey RF, Lytle CD, Cyr WH. Implications of laboratory tests of condom integrity. Sex Transm Dis. 1999;26:216-220.
3. Murphy EM, Greene ME, Mihailovic A, Olupot-Olupot P. Was the "ABC" approach (abstinence, being faithful, using condoms) responsible for Uganda's decline in HIV? PLoS Med. 2006;3:e379.
4. AIDS.MD. ABC of HIV prevention. Web site: http://aids.md/aids/index.php?cmd=item&id=264.
5. Pinkerton SD, Abramson PR. Effectiveness of condoms in preventing HIV transmission. Soc Sci Med. 1997;44:1303-1312.
6. Weller S, Davis K. Condom effectiveness in reducing heterosexual HIV transmission. Cochrane Database Syst Rev. 2002;1(1):CD003255.
7. Giannou FK, Tsiara CG, Nikolopoulos GK, et al. Condom effectiveness in reducing heterosexual HIV transmission: a systematic review and meta-analysis of studies on HIV serodiscordant couples. Expert Rev Pharmacoecon Outcomes Res. 2015;21:1-11.
8. Ahmed S, Lutalo T, Wawer M, et al. HIV incidence and sexually transmitted disease prevalence associated with condom use: a population study in Rakai, Uganda. AIDS. 2001;15:2171-2179.
9. Detels R, English P, Visscher BR, et al. Seroconversion, sexual activity, and condom use among 2915 HIV seronegative men followed for up to 2 years. J Acquir Immune Defic Syndr. 1989;2:77-83.
10. Smith DK, Herbst JH, Zhang X, Rose CE. Condom effectiveness for HIV prevention by consistency of use among men who have sex with men in the United States. J Acquir Immune Defic Syndr. 2015;68:337-344.
11. Remis RS, Alary M, Liu J, Kaul R, Palmer RW. HIV transmission among men who have sex with men due to condom failure. PLoS One. 2014;9:e107540.
12. UNFPA, WHO, UNAIDS. Position statement on condoms and the prevention of HIV, other sexually transmitted infections and unintended pregnancy. July 7, 2015. Web site: http://www.unaids.org/en/resources/presscentre/featurestories/2015/july/20150702_condoms_prevention.
13. Walusaga HA, Kyohangirwe R, Wagner GJ. Gender differences in determinants of condom use among HIV clients in Uganda. AIDS Patient Care STDS. 2012;26:694-9.
14. UNAIDS. Global report. UNAIDS report on the global AIDS epidemic 2013. Web site: http://www.unaids.org/en/resources/campaigns/globalreport2013..
15. Khawcharoenporn T, Kendrick S, Smith K. HIV risk perception and preexposure prophylaxis interest among a heterosexual population visiting a sexually transmitted infection clinic. AIDS Patient Care STDS. 2012;26:222-233.
16. Herbenick D, Schick V, Reece M, et al. Characteristics of condom and lubricant use among a nationally representative probability sample of adults ages 18-59 in the United States. J Sex Med. 2013;10:474-483.
17. Liz Hamel L, Firth J, Hoff T, Kates J, Levine S, Dawson L. HIV/AIDS in the lives of gay and bisexual men in the United States. Section 4. Condom use and HIV testing. Henry J Kaiser Family Foundation. September 25, 2014.
18. Herbst JH, Sherba RT, Crepaz N, et al. A meta-analytic review of HIV behavioral interventions for reducing sexual risk behavior of men who have sex with men. J Acquir Immune Defic Syndr. 2005;39:228-241.
19. Johnson WD, Diaz RM, Flanders WD, et al. Behavioral interventions to reduce risk for sexual transmission of HIV among men who have sex with men. Cochrane Database Syst Rev. 2008;16:CD001230.
20. Lu H, Liu Y, Dahiya K, et al. Effectiveness of HIV risk reduction interventions among men who have sex with men in China: a systematic review and meta-analysis. PLoS One. 2013;8:e72747.
21. Johnson LF, Hallett TB, Rehle TM, Dorrington RE. The effect of changes in condom usage and antiretroviral treatment coverage on human immunodeficiency virus incidence in South Africa: a model-based analysis. J R Soc Interface. 2012;9:1544-1554.
22. UNAIDS. Condomize! Campaign hits AIDS 2012. July 24, 2012. Web site: http://www.unaids.org/en/resources/presscentre/featurestories/2012/july/20120725condomize.
23. WHO. HIV/AIDS. Fact sheet No. 360. November 2015. Web site: http://www.who.int/mediacentre/factsheets/fs360/en/.
24. Auvert B, Taljaard D, Lagarde E, Sobngwi-Tambekou J, Sitta R, Puren A. Randomized, controlled intervention trial of male circumcision for reduction of HIV infection risk: the ANRS 1265 Trial. PLoS Med. 2005;2:e298.
25. Bailey RC, Moses S, Parker CB, et al. Male circumcision for HIV prevention in young men in Kisumu, Kenya: a randomised controlled trial. Lancet. 2007;369:643-656.
26. Gray RH, Kigozi G, Serwadda D, et al. Male circumcision for HIV prevention in men in Rakai, Uganda: a randomised trial. Lancet. 2007;369:657-666.
27. Wawer MJ, Makumbi F, Kigozi G, et al. Circumcision in HIV-infected men and its effect on HIV transmission to female partners in Rakai, Uganda: a randomised controlled trial. Lancet. 2009;374:229-237.
28. Sánchez J, Sal Y Rosas VG, Hughes JP, et al. Male circumcision and risk of HIV acquisition among MSM. AIDS. 2011;25:519-523.
29. UNAIDS. Male circumcision: global trends and determinants of prevalence, safety and acceptability. 2007.
30. UNAIDS. Male circumcision a critical new HIV prevention tool. 2011.
31. World Health Organization. 10 million men stepped up for HIV prevention through voluntary medical male circumcision services. December 2015. Web site: WHO_HIV_2015.50_eng.pdf.
32. World Health Organization. Voluntary medical male circumcision for HIV prevention in 14 priority countries in East and Southern Africa. July 2015. Web site: http://www.who.int/hiv/pub/malecircumcision/brief2015/en/.
33. Lau FK, Jayakumar S, Sgaier SK. Understanding the socio-economic and sexual behavioural correlates of male circumcision across eleven voluntary medical male circumcision priority countries in southeastern Africa. BMC Public Health. 2015;15:813.
34. National Bureau of Statistics of China monitoring report of migrant workers in China in 2011. Beijing: National Bureau of Statistics of China. 2012. [Cited in: Ning C, Jiang J, Ye L, et al. Comparison of three intervention models for promoting circumcision among migrant workers in western China to reduce local sexual transmission of HIV. PLoS One. 2013;8:e76107.]
35. Zeng Y, Zhang L, Li T, et al. Risk factors for HIV/syphilis infection and male circumcision practices and preferences among men who have sex with men in China. Biomed Res Int. 2014;2014:498987.
36. Tieu HV, Phanuphak N, Ananworanich J, et al. Acceptability of male circumcision for the prevention of HIV among high-risk heterosexual men in Thailand. Sex Transm Dis. 2010;37:352-355.
37. Chariyalertsak S, Kosachunhanan N, Saokhieo P, et al. HIV incidence, risk factors, and motivation for biomedical intervention among gay, bisexual men, and transgender persons in northern Thailand. PLoS One. 2011;6:e24295.
38. Weiss SM, Zulu R, Jones DL, Redding CA, Cook R, Chitalu N. The Spear and Shield intervention to increase the availability and acceptability of voluntary medical male circumcision in Zambia: a cluster randomised controlled trial. Lancet HIV. 2015;2:e181-189.
39. Baeten JM, Donnell D, Ndase P, et al. Antiretroviral prophylaxis for HIV prevention in heterosexual men and women. N Engl J Med. 2012;367:399-410.
40. Thigpen MC, Kebaabetswe PM, Paxton LA, et al. Antiretroviral preexposure prophylaxis for heterosexual HIV transmission in Botswana. N Engl J Med. 2012;367:423-434.
41. Grant RM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363:2587-2599.
42. Molina JM, Capitant C, Spire B, et al. On-demand preexposure prophylaxis in men at high risk for HIV-1 infection. N Engl J Med. 2015;373:2237-2246.
43. Choopanya K, Martin M, Suntharasamai P, et al. Antiretroviral prophylaxis for HIV infection in injecting drug users in Bangkok, Thailand (the Bangkok Tenofovir Study): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2013;381:2083-2090.
44. US Public Health Service. Preexposure prophylaxis for the prevention of HIV infection in the United States-2014. A clinical practice guideline. 2014. Web site: www.cdc.gov/hiv/pdf/PrEPguidelines2014.pdf.
45. Anderson PL, Glidden DV, Liu A, et al. Emtricitabine-tenofovir concentrations and pre-exposure prophylaxis efficacy in men who have sex with men. Sci Transl Med. 2012;4:151ra125.
46. Peabody R. Non-daily PrEP regimens provide extra options, but adherence is often better with daily dosing. aidsmap. July 21, 2015. Web site: http://www.aidsmap.com/page/2986241/.
47. Trezza C, Ford SL, Spreen W, Pan R, Piscitelli S. Formulation and pharmacology of long-acting cabotegravir. Curr Opin HIV AIDS. 2015;10:239-245.
48. Landovitz RJ, Kofron R, McCauley M. The promise and pitfalls of long-acting injectable agents for HIV prevention. Curr Opin HIV AIDS. 2016;11:122-128.
49. Abraham BK, Gulick R. Next-generation oral preexposure prophylaxis: beyond tenofovir. Curr Opin HIV AIDS. 2012;7:600-606.
50. McNeil DG Jr. San Francisco is changing the face of AIDS treatment. New York Times. October 5, 2015.
51. Smith DK, Van Handel M, Wolitski RJ, et al. Vital signs: estimated percentages and numbers of adults with indications for preexposure prophylaxis to prevent HIV acquisition-United States, 2015. MMWR Morb Mortal Wkly Rep. 2015;64:1291-1295.
52. PrEPWatch. Where do you fit on the map? Web site: http://www.prepwatch.org/.
53. Hood JE, Buskin SE, Dombrowski JC, et al. Dramatic increase in preexposure prophylaxis use among MSM in King County, Washington. AIDS. 2015 Nov 10. Epub ahead of print.
54. King HL, Keller SB, Giancola MA, et al. Pre-exposure prophylaxis accessibility research and evaluation (PrEPARE Study). AIDS Behav. 2014;18:1722-1725.
55. Bil JP, Davidovich U, van der Veldt WM, et al. What do Dutch MSM think of preexposure prophylaxis to prevent HIV-infection? A cross-sectional study. AIDS. 2015;29:955-964.
56. Karris MY, Beekmann SE, Mehta SR, Anderson CM, Polgreen PM. Are we prepped for preexposure prophylaxis (PrEP)? Provider opinions on the real-world use of PrEP in the United States and Canada. Clin Infect Dis. 2014;58:704-712.
57. Supervie V, Barrett M, Kahn JS, et al. Modeling dynamic interactions between pre-exposure prophylaxis interventions & treatment programs: predicting HIV transmission & resistance. Sci Rep. 2011;1:185.
58. Juusola JL, Brandeau ML, Owens DK, Bendavid E. The cost-effectiveness of preexposure prophylaxis for HIV prevention in the United States in men who have sex with men. Ann Intern Med. 2012;156:541-550.
59. Schneider K, Gray RT, Wilson DP. A cost-effectiveness analysis of HIV preexposure prophylaxis for men who have sex with men in Australia. Clin Infect Dis. 2014;58:1027-1034.
60. Mayer KH. Antiretroviral chemoprophylaxis: state of evidence and the research agenda. Clin Infect Dis. 2014;59(S1):S47–S51.
61. Celum CL, Delany-Moretlwe S, McConnell M, et al. Rethinking HIV prevention to prepare for oral PrEP implementation for young African women. J Int AIDS Soc. 2015;18(4Suppl 3):20227.
62. World Health Organization. Guideline on when to start antiretroviral therapy and on pre-exposure prophylaxis for HIV. September 2015. Web site: http://www.who.int/hiv/pub/guidelines/earlyrelease-arv/en/.
63. Connor EM, Sperling RS, Gelber R, et al. Reduction of maternal-infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. Pediatric AIDS Clinical Trials Group Protocol 076 Study Group. N Engl J Med. 1994;331:1173-1180.
64. Chasela C, Hudgens M, Jamieson D, et al. Maternal or infant antiretroviral drugs to reduce HIV-1 transmission. N Engl J Med. 2010;362:2271-2281.
65. Shapiro RL, Hughes MD, Ogwu A, et al. Antiretroviral regimens in pregnancy and breast-feeding in Botswana. N Engl J Med. 2010;362:2282-2294.
66. Kesho Bora Study Group, de Vincenzi I. Triple antiretroviral compared with zidovudine and single-dose nevirapine prophylaxis during pregnancy and breastfeeding for prevention of mother-to-child transmission of HIV-1 (Kesho Bora study): a randomised controlled trial. Lancet Infect Dis. 2011;11:171-180.
67. Cohen MS, Chen YQ, McCauley M, et al. Prevention of HIV-1 infection with early antiretroviral therapy. N Engl J Med. 2011;365:493-505.
68. UNAIDS. 90–90–90: An ambitious treatment target to help end the AIDS epidemic. 2014.
69. Mandelbrot L, Tubiana R, Le Chenadec J, et al. No perinatal HIV-1 transmission from women with effective antiretroviral therapy starting before conception. Clin Infect Dis. 2015:61:1715-1725.
70. World Health Organization. Mother-to-child transmission. Web site: http://www.wpro.who.int/hiv/topics/pmtct/about/en/.
71. Montaner JS, Lima VD, Harrigan PR, et al. Expansion of HAART coverage is associated with sustained decreases in HIV/AIDS morbidity, mortality and HIV transmission: the "HIV Treatment as Prevention" experience in a Canadian setting. PLoS One. 2014;9:e87872.
72. Das M, Chu PL, Santos GM, et al. Decreases in community viral load are accompanied by reductions in new HIV infections in San Francisco. PLoS One. 2010;5:e11068.
73. Althoff KN, Buchacz K, Hall HI, et al. U.S. trends in antiretroviral therapy use, HIV RNA plasma viral loads, and CD4 T-lymphocyte cell counts among HIV-infected persons, 2000 to 2008. Ann Intern Med. 2012;157:325-335.
74. Centers for Disease Control and Prevention. HIV Surveillance Report, 2014. Vol. 26. November 2015. Web site: http://www.cdc.gov/hiv/library/reports/surveillance/
75. UNAIDS. 2015 progress report on the global plan towards the elimination of new HIV infections among children and keeping their mothers alive. Web site: http://www.unaids.org/en/resources/documents/2015/JC2774_2015ProgressReport_GlobalPlan.
76. Centers for Disease Control and Prevention. HIV/AIDS. HIV testing. Background. 2015. Web site: http://www.cdc.gov/hiv/testing/.
77. Ratmann O, van Sighem A, Bezemer D, et al. Sources of HIV infection among men having sex with men and implications for prevention. Sci Transl Med. 2016:8:320ra2.
78. Fonjungo PN, Boeras DI, Zeh C, Alexander H, Parekh BS, Nkengasong JN. Access and quality of HIV-related point-of-care diagnostic testing in global health programs. Clin Infect Dis. 2016;62:369-374.
79. Perriens J, Habiyambere V. The demand for CD4 testing, viral load and early infant diagnostic testing in low and middle income countries. will grow strongly between now and 2018-but not enough to reach the 90-90-90 targets. World Health Organization. July 2015. Web site: http://www.who.int/hiv/pub/posters/ias2015-poster2/en/.
80. Sabapathy K, Van den Bergh R, Fidler S, Hayes R, Ford N. Uptake of home-based voluntary HIV testing in sub-Saharan Africa: a systematic review and meta-analysis. PLoS Med. 2012;9:e1001351.
81. Suthar AB, Ford N, Bachanas PJ, et al. Towards universal voluntary HIV testing and counselling: a systematic review and meta-analysis of community-based approaches. PLoS Med. 2013;10:e1001496.
82. Pottie K, Medu O, Welch V, et al. Effect of rapid HIV testing on HIV incidence and services in populations at high risk for HIV exposure: an equity-focused systematic review. BMJ Open. 2014;4:e006859.
83. Cohen MS, Smith MK, Muessig KE, Hallett TB, Powers KA, Kashuba AD. Antiretroviral treatment of HIV-1 prevents transmission of HIV-1: where do we go from here? Lancet. 2013;382:1515-1524.
84. ClinicalTrials.gov. ClinicalTrials.gov Identifier NCT02120352. A phase IIb study to evaluate a long-acting intramuscular regimen for maintenance of virologic suppression (following induction with an oral regimen of GSK1265744 and abacavir/lamivudine) in human immunodeficiency virus type 1 (HIV-1) infected, antiretroviral therapy-naive adult subjects. Web site: https://clinicaltrials.gov/ct2/show/NCT02120352.
85. Eaton JW, Johnson LF, Salomon JA, et al. HIV treatment as prevention: systematic comparison of mathematical models of the potential impact of antiretroviral therapy on HIV incidence in South Africa. PLoS Med. 2012;9:e1001245.
86. Smith DK, Herbst JH, Rose CE. Estimating HIV protective effects of method adherence with combinations of preexposure prophylaxis and condom use among African American men who have sex with men. Sex Transm Dis. 2015;42:88-92.
87. Liu H, Su Y, Zhu L, Xing J, Wu J, Wang N. Effectiveness of ART and condom use for prevention of sexual HIV transmission in serodiscordant couples: a systematic review and meta-analysis. PLoS One. 2014;9:e111175.
88. van de Vijver DA, Nichols BE, Abbas UL, et al. Preexposure prophylaxis will have a limited impact on HIV-1 drug resistance in sub-Saharan Africa: a comparison of mathematical models. AIDS. 2013;27:2943-2951.
89. Baeten J, Heffron R, Kidoguchi L, et al. Near elimination of HIV transmission in a demonstration project of PrEP and ART. Conference on Retroviruses and Opportunistic Infections. February 23-26, 2015. Seattle, Washington. Abstract 24.
90. Frieden TR, Foti KE, Mermin J. Applying public health principles to the HIV epidemic--how are we doing? N Engl J Med. 2015;373:2281-7.
91. San Francisco Department of Public Health. HIV epidemiology annual report. 2014.
92. International Advisory Panel on HIV Care Continuum Optimization. IAPAC Guidelines for Optimizing the HIV Care Continuum for Adults and Adolescents. J Int Assoc Provid AIDS Care. 2015;14(Suppl 1):S3-S34.
93. Stover J, Hallett TB, Wu Z, et al. How can we get close to zero? The potential contribution of biomedical prevention and the investment framework towards an effective response to HIV. PLoS One. 2014;9:e111956.
94. World Health Organization. Ebola situation report. Web site: http://apps.who.int/ebola/ebola-situation-reports.
95. World Health Organization. Poliomyelitis. Fact sheet No. 114. October 2015.
96. Centers for Disease Control and Prevention. Emergency preparedness and response. Smallpox. Web site: http://www.bt.cdc.gov/agent/smallpox/.
|
|
| |
| |
|
|
|