| |
ASM: HIV-1 Seroconversion Across 17 International Demonstration Projects Using Pre-exposure Prophylaxis (PrEP) With Oral Emtricitabine/Tenofovir Disoproxil Fumarate (FTC/TDF)
|
| |
| |
Reported by Jules Levin
ASM June 16-20 Boston, MA
Scott McCallister, David Magnuson, Rebecca Guzman, Valentina Shvachko, Keith Rawlings, Robertino Mera Gilead Sciences, Inc., Foster City, CA, USA

Program ABSTRACT
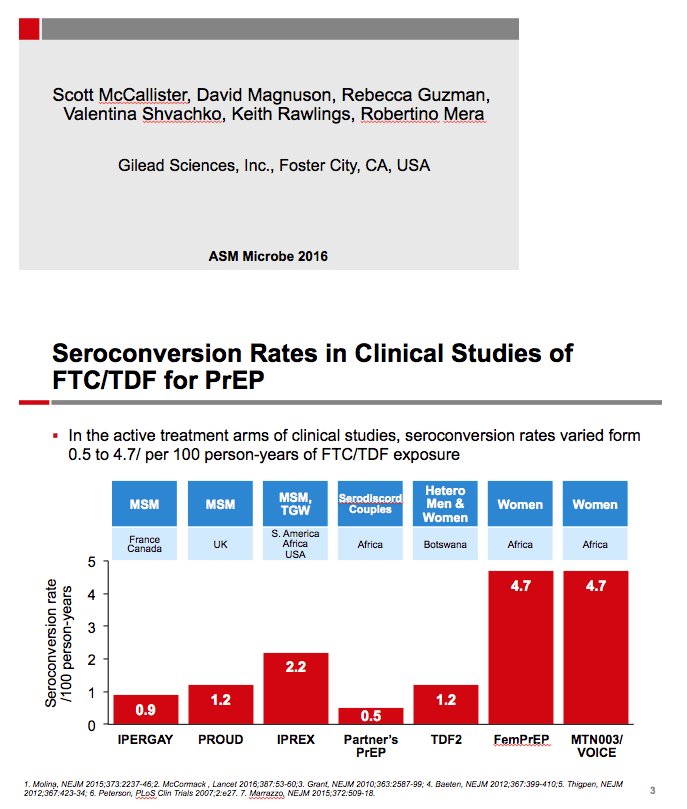
Background: HIV-1 seroconversion rates in carefully managed PrEP clinical studies varied by the treatment population evaluated, from 1.2/100 person years (PY) in United Kingdom urban clinics (PROUD) to 1.9/100 PY in the US, South America and Africa (iPrEX). We collected HIV-1 seroconversion data with use of FTC/TDF for PrEP from diverse countries and populations in a real-world setting to compare to clinical study data.
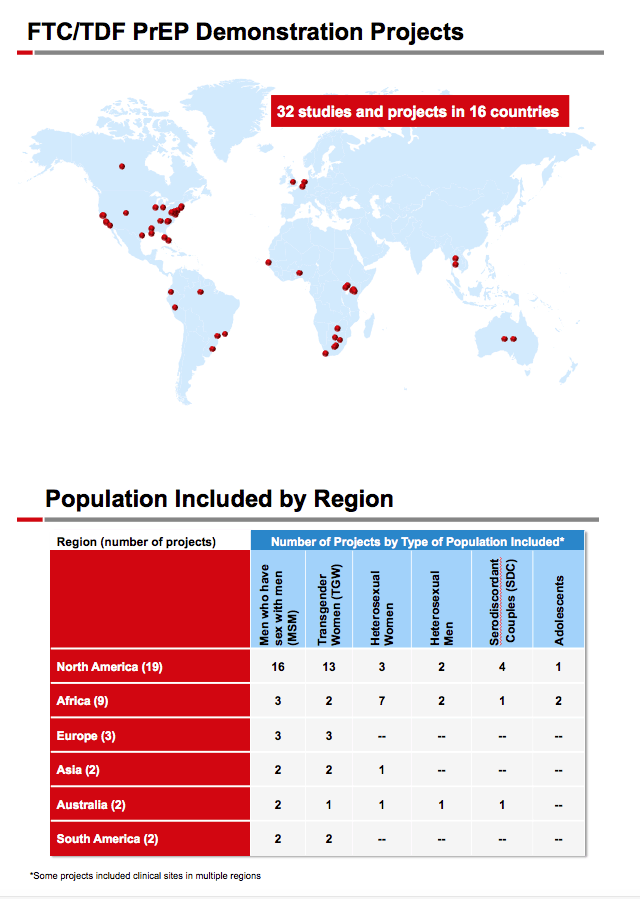
Methods: Patient time exposure from 2011 through mid-2015 was collected from 4,981 HIV-negative adults participating in 17 ongoing international PrEP demonstration projects from North and South America, Asia, and Africa; all participants received open-label FTC/TDF for daily use. All studies conducted HIV-1 and sexually transmitted infection testing. Time exposed to FTC/TDF while at risk of seroconversion was analyzed; participants in deferred FTC/TDF and other non-PrEP arms were excluded and exact Poisson statistics were used to compute rates and confidence intervals.
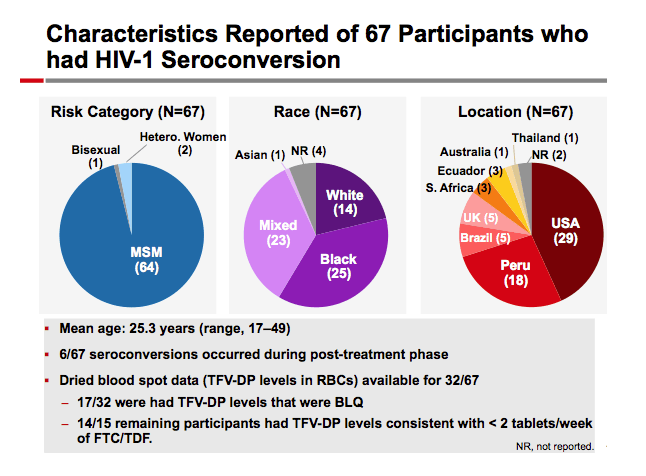
Results: The 4,981 participants from the 17 studies had 4,171 PY of exposure to FTC/TDF. A total of 54 HIV-1 seroconversions occurred, a rate of 1.29/100 PY (95% CI: 0.97-1.69). Median time to seroconversion was 268 days, range 4-601 (IQR: 171-425). 9/54 (14.8%) reported signs or symptoms of acute retroviral syndrome at the time of seroconversion. Mean age of those who seroconverted: 25.1 + 7 years; 61% were <25 years old. 96% of those seroconverting were male, contributing 3826.2 PY of FTC/TDF exposure, for a rate of 1.36/100 PY (95% CI: 1.02-1.78). Women had 278.6 PYs of FTC/TDF exposure for a rate of 0.72/100 PY (95% CI: 0.09-2.60). 25/54 seroconverters were from the USA. Race for seroconverters: black 43%, mixed 39%, white 11%, Asian 2%. Genotypic resistance data for 18 of 54 seroconverters was available; 15/18 (83%) had wild-type virus, 3/18 had M184V/I; there were no reports of K65R or K70R.
Conclusions: This large meta-analysis of HIV-1 seroconversion data with the use of FTC/TDF for PrEP demonstrates a low overall seroconversion rate in a real world setting with an upper confidence interval below 2 infections per 100 PY. While these data are reassuring and inform prevention programs in diverse settings, more data are needed in women and transgendered individuals.





|
|
| |
| |
|
|
|