 |
 |
 |
| |
Uptake and virologic outcomes of one-pill versus multi-pill antiretroviral regimen among treatment-naïve non-perinatally HIV-infected youth (2006-2014) - One-Pill ART Promotes HIV Control in US Youth--But Uptake Lower in Girls/Women
|
| |
| |
IDWeek 2016, October 26-30, 2016, New Orleans
Mark Mascolini
HIV-positive 12- to 24-year-olds at 18 US sites had nearly doubled odds of viral suppression with single- versus multipill regimens 15 months after starting antiretroviral therapy (ART) [1]. Boys/men had more than 5 times higher odds of starting a one-pill combination, probably because efavirenz-containing Atripla accounted for almost three-quarters of the single-pill regimens in this analysis.
Teens and young adults with HIV often face stern challenges in maintaining good adherence to ART. Research indicates that one-pill once-daily regimens foster better adherence, but their impact in HIV-positive youth remains unclear. HIV Research Network investigators conducted this study to identify factors linked to starting single-pill ART and to attaining viral suppression in US youth beginning their first regimen.
The analysis involved 12- to 24-year-olds at 18 US sites in the HIV Research Network who started their first antiretroviral regimen in 2006-2014. No participants acquired HIV perinatally. The researchers used multivariable models to pinpoint factors associated with starting a single-pill combination and with attaining virologic suppression, defined as a viral load below 400 copies.
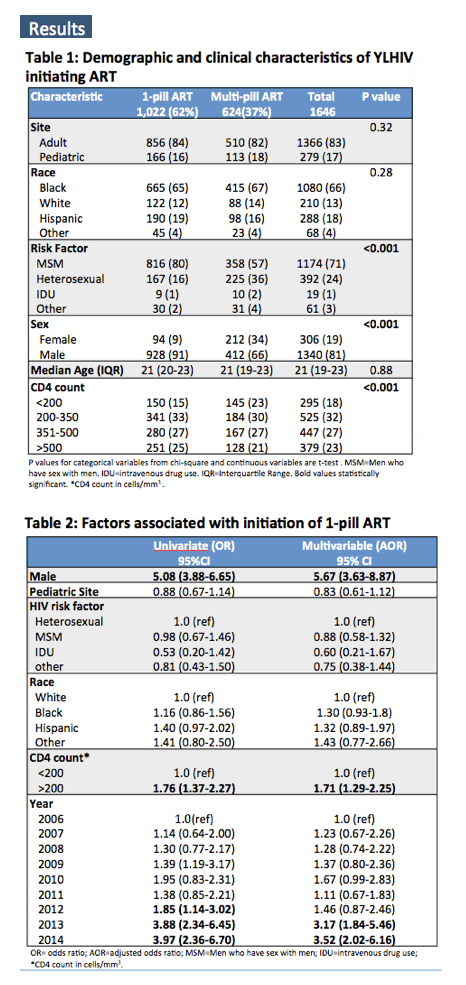
The study group included 1646 participants, 1022 (62%) of whom started one-pill ART and 624 of whom started a multipill regimen. A significantly higher proportion of males (928/1340, 69%) than females (94/306, 31%) started single-pill therapy (P < 0.001). Efavirenz/TDF/FTC proved the most frequently prescribed one-pill regimen (72%), followed distantly by rilpivirine/TDF/FTC (15%), elvitegravir/cobicistat/TDF/FTC (13%), and dolutegravir/ABC/3TC (1%). The proportion of participants starting a single pill climbed from 47% in 2006 to 78% in 2014.
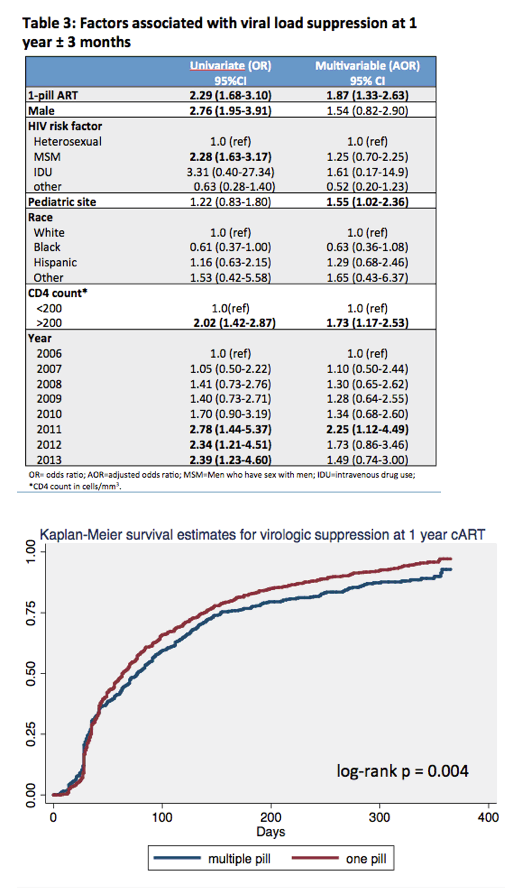
Multivariate analysis identified four independent predictors of starting a one-pill combo at the following adjusted odds ratios (aOR) and 95% confidence intervals (CI):
-- Male vs female: aOR 5.67, 95% CI 3.63 to 8.87
-- Pre-ART CD4 count above 200 vs below: aOR 1.71, 95% CI 1.29 to 2.25
-- Starting ART in 2013 vs 2006: aOR 3.17, 95% CI 1.84 to 5.46
-- Starting ART in 2014 vs 2006: aOR 3.52, 95% CI 2.02 to 6.16
Neither race nor HIV transmission mode predicted starting single-pill therapy in this analysis.
A separate multivariate analysis singled out four independent predictors of viral suppression 15 months after beginning ART, starting with almost doubled odds of suppression when taking a one-pill regimen:
-- One-pill ART: aOR 1.87, 95% CI 1.33 to 2.63
-- Pediatric site: aOR 1.55, 95% CI 1.02 to 2.36
-- Pre-ART CD4 count above 200 vs below: aOR 1.73, 95% CI 1.17 to 2.53
-- Starting ART in 2011 vs 2006: aOR 2.25, 95% CI 1.12 to 4.49
Starting ART in 2012 or 2013 did not independently predict viral suppression; neither did gender, HIV transmission mode, or race.
The researchers observed that the higher likelihood of starting single-pill therapy by males "is likely driven by the potential teratogenicity of efavirenz," and efavirenz/TDF/FTC accounted for almost three quarters of one-pill regimens prescribed. One-pill ART may improve virologic outcome in teens and young adults, the investigators concluded, but "interventions to sustain engagement in care are critical to ultimately improving overall outcomes in this age group."
Reference
1. Griffith DC, Farmer C, Rutstein R, et al. Uptake and virologic outcomes of one-pill versus multi-pill antiretroviral therapy among treatment-naive non-perinatally HIV-infected youth (2006-2014). IDWeek 2016, October 26-30, 2016, New Orleans. Abstract 1544.


Uptake and virologic outcomes of one-pill versus multi-pill antiretroviral regimen among treatment-naïve non-perinatally HIV-infected youth (2006-2014)
David C. Griffith1, Charles Farmer1, Richard Rutstein2, W. Christopher Mathews3, Robert Beil4, P. Todd Korthuis5, Stephen Berry1, Ank E Nijhawan6, Aditya H. Gaur7, Kelly Gebo1, Allison Agwu, MD1 for the HIV the Research Network
(1)Johns Hopkins University School of Medicine, Baltimore, MD, (2)Children's Hospital of Philadelphia, Philadelphia, PA, (3)University of California at San Diego, San Diego, CA, (4)Montefiore Medical Group, New York, NY, (5)Oregon Health & Sciences University, Portland, OR, (6)University of Texas Southwestern Medical Center, Dallas, TX, (7)St. Jude Children's Research Hospital, Memphis, TN

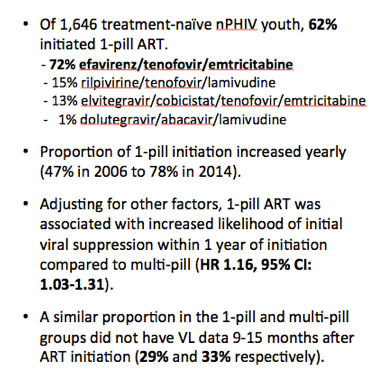
|
| |
|
 |
 |
|
|