 |
 |
 |
| |
Retesting of suspected low-level HIV-1 viral load blips: A new paradigm to prevent extra clinic visits and unnecessary patient anxiety
|
| |
| |
Reported by Jules Levin
IDWeek 2016 Oct 26-30 New Orleans
Joe Eron, William Garner, Lilian Wei, Lijie Zhong, Michael Miller, Javier Szwarcberg, Hal Martin, Andrew Plummer, Kim Lindstrom, James Porter, David Piontkowsky, Angela Light, Heinz Reiske, Erin Quirk, and Kirsten White
Program abstract:
Retesting of suspected low-level HIV-1 viral load blips: A new paradigm to prevent extra clinic visits and unnecessary patient anxiety
Session: Oral Abstract Session: HIV Clinical Management
Friday, October 28, 2016: 10:30 AM
Room: 275-277
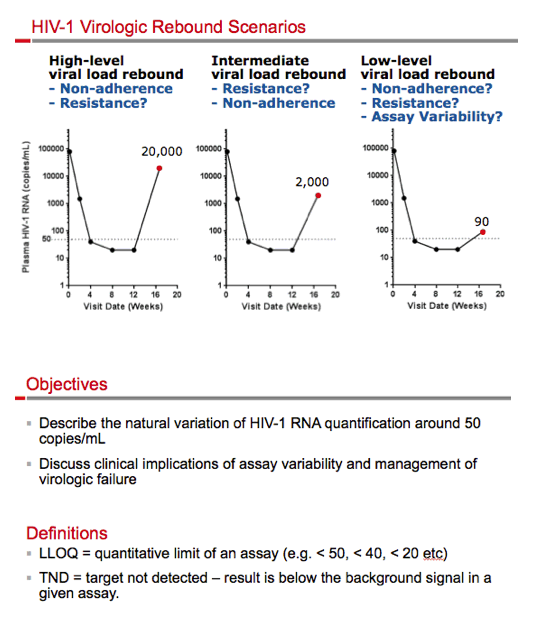
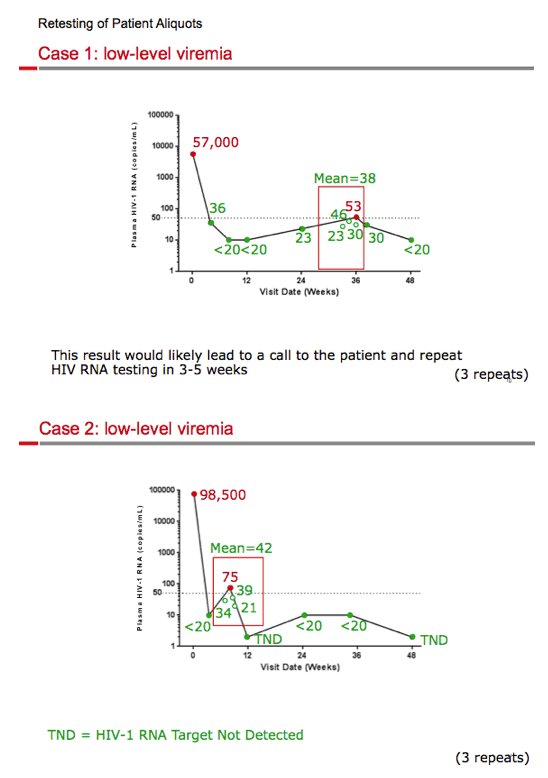
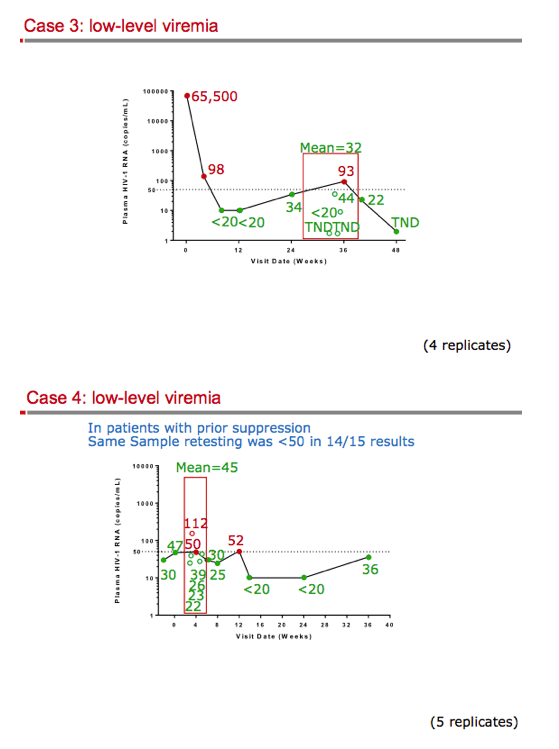
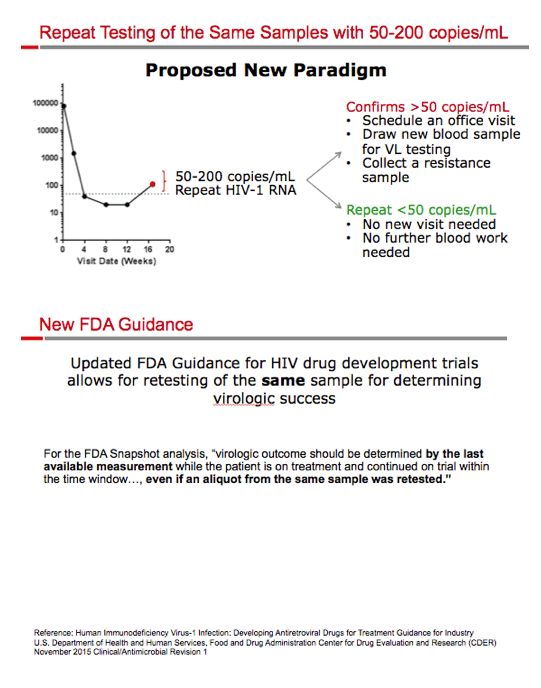
Background: Successful HIV-1 treatment is the achievement and maintenance of plasma HIV-1 RNA at or near undetectable levels. All biological assays show data variability-including HIV-1 RNA assays-especially near the lower limit of quantitation. Recent FDA guidance for clinical trials in HIV proposes retesting of the same sample with an initial ≥50 c/mL result, since this may produce a result of <50 c/mL and eliminate a virologic failure classification. In clinical practice, after achieving <50 c/mL, an HIV-1 RNA value ≥50 c/mL often triggers a follow-up clinic visit, retest, and a potential regimen change. For patients, a report of being 'detectable' can cause substantial anxiety. We tested the hypothesis that many low-level viral load blips are due to assay variation and might be eliminated with repeat testing of a second aliquot from the same sample.
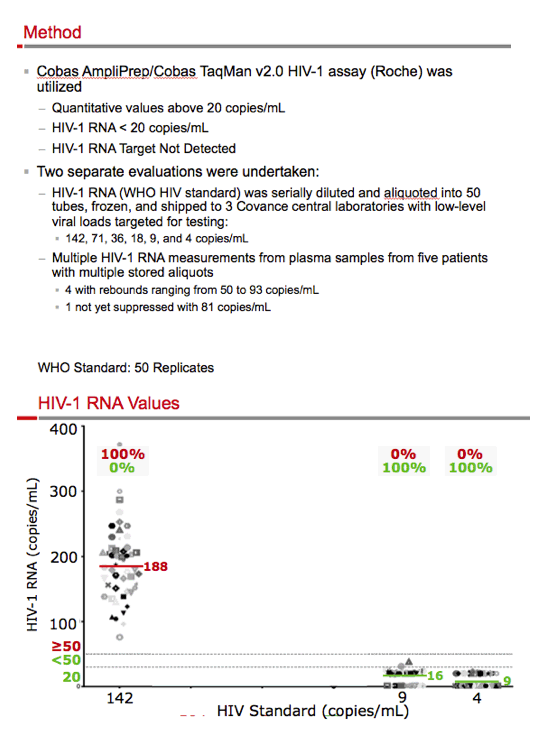
Methods: 50 aliquots of diluted WHO HIV standard at viral loads <200 c/mL and 4-6 aliquots of plasma samples from 4 patients experiencing blips of 50-200 c/mL after having had HIV-1 RNA <50 c/mL were quantified using the Cobas AmpliPrep/Cobas TaqMan v2.0 HIV-1 assay (Roche) at Covance central laboratories.
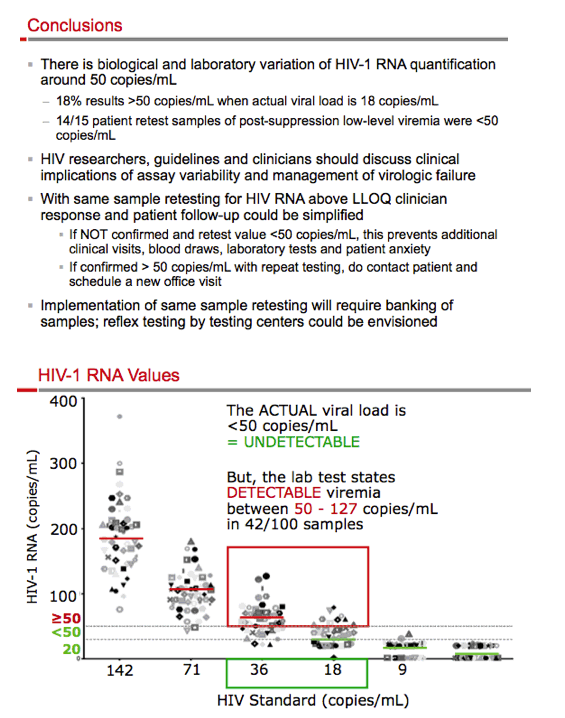
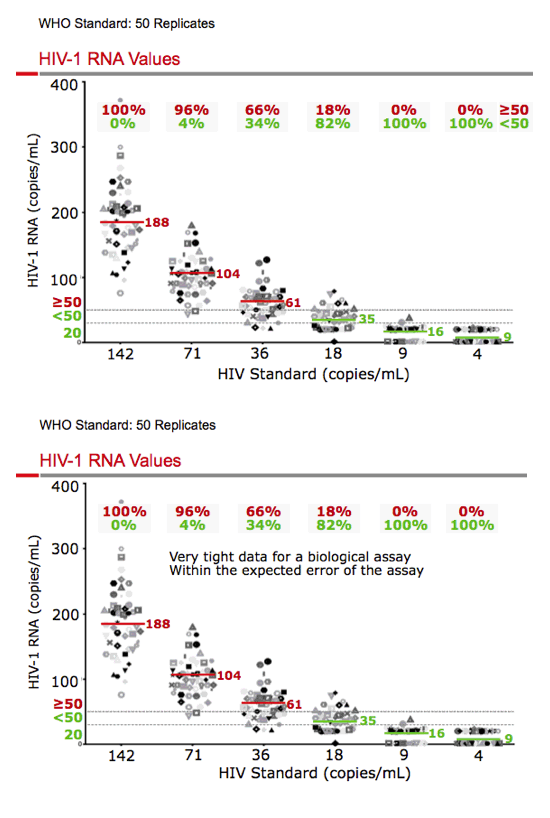
Results: The TaqMan assay of the WHO standards at viral loads <200 c/mL performed within the expected variability according to assay specifications. However, standards with 36 c/mL and 17 c/mL of HIV-1 RNA showed values of ≥50 c/mL in 66% and 18% of tests, respectively. In patients treated with antiretrovirals and having unexpected ≥50 c/mL results, retesting of multiple aliquots of stored plasma found <50 c/mL in most cases upon retesting (15/16; 94%).
Conclusion: HIV-1 RNA assays have expected variability around 50 c/mL that affect clinical trial results and clinical practice. In patients with a history of viral load suppression, unexpected low-level viremia may be due to assay variability rather than poor drug adherence or true virologic failure. Retesting a stored aliquot of the same sample may differentiate between assay variability and virologic failure as the source of perceived viremia. This retesting strategy could save time, money, and anxiety for patients and their providers, and decrease follow-up clinic visits without increasing the risk of virologic failure and resistance development.

|
| |
|
 |
 |
|
|