 |
 |
 |
| |
Safety and efficacy of the combination Sofosbuvir-Ledipasvir in HCV mono-infected patients from the French observational Cohort ANRS CO22 HEPATHER
The ANRS-AFEF HEPATHER study group
|
| |
| |
Reported by Jules Levin
AASLD: The Liver Meeting 2017, October 20-24, 2017, Washington, DC

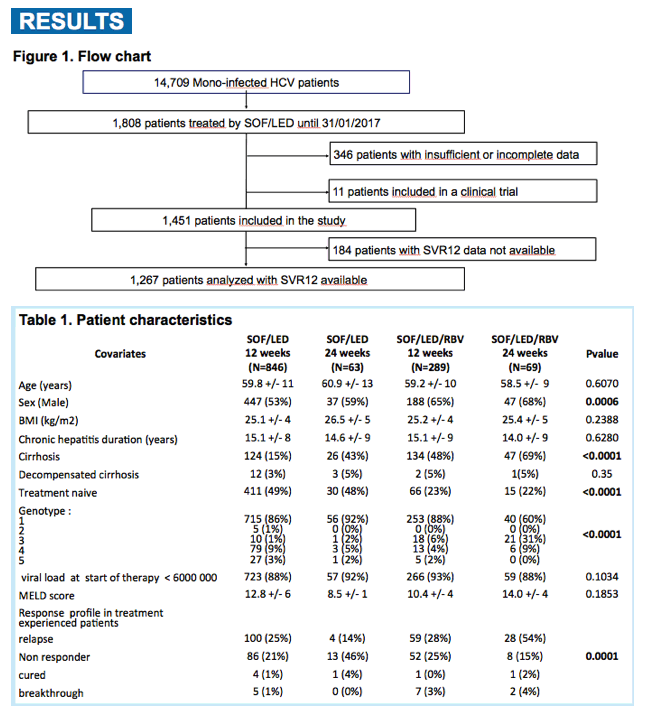
Among 1,267 patients analyzed, 1064 patients had genotype 1, 55 had genotype 2/3, 101 had genotype 4 and 33 had genotype 5/6. Moreover, 358 patients received Ribavirin.
Therapeutic option differed with sex, previous treatment experience, cirrhosis status, genotypes and response profile (for experienced patients). Details on prescription associations with factors are provided in table1.
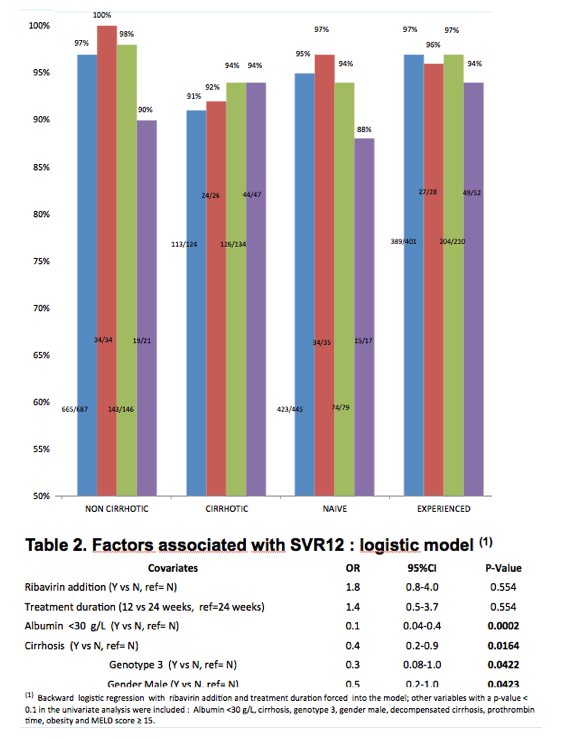
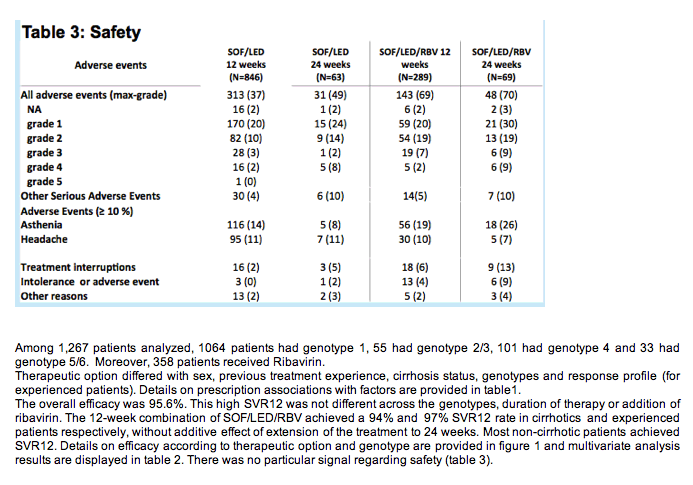
The overall efficacy was 95.6%. This high SVR12 was not different across the genotypes, duration of therapy or addition of ribavirin. The 12-week combination of SOF/LED/RBV achieved a 94% and 97% SVR12 rate in cirrhotics and experienced patients respectively, without additive effect of extension of the treatment to 24 weeks. Most non-cirrhotic patients achieved SVR12. Details on efficacy according to therapeutic option and genotype are provided in figure 1 and multivariate analysis results are displayed in table 2. There was no particular signal regarding safety (table 3).
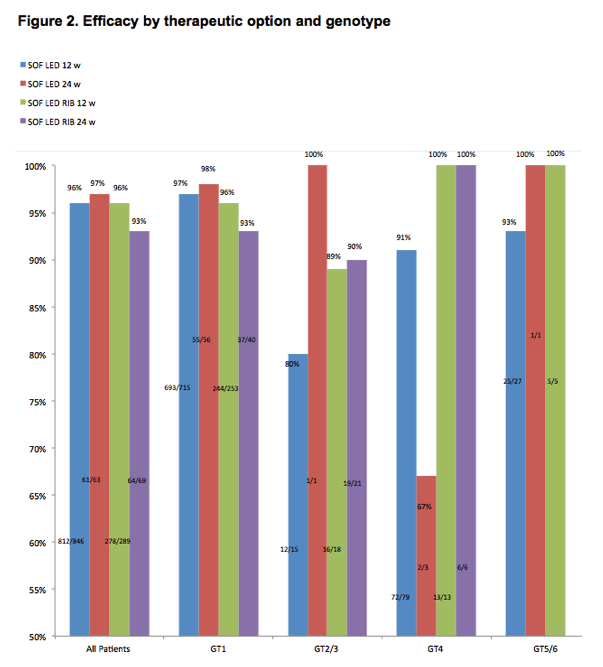
|
| |
|
 |
 |
|
|