| |
SVR Reduced HCC by 71%
|
| |
| |
from Jules: there never was any doubt that SVR would reduce or eliminate risk for HCC. In this study cirrhosis prior to treatment had a higher HCC risk then for those without cirrhosis, but that is to be expected and merely reinforces how crucial it is to treat HCV as early as possible, and of course to screen ALL for HCV, as I have said for years we should have routine annual HCV screening for ALL. This study highlights also what I and others have said, the economic cost savings - effectiveness - of screening ALL and treating HCV is obvious, the long term costs of NOT screening & treating results in HCC and or=ther serious disease complications that is much more costly in the long term. Now prices have been reduced by 70% for DAA HCV regimens to approximately as low as $15,000 for some HCV drug formularies to $25,000 for a course of treatment or a cure, cure/SVR rates are 95% +. It is obscene & outrageous that federal and state officials are not trying to put together HCV elimination programs, which means universal screening, linkage to care, treatment and special support services for those hard to treat patient populations, this using ir with a history of IDU. There are highly marginalized patient populations, of note those currently using in drugs and injecting drugs, and for this population special support services are necessary, but there are so many ways to do this. Its not like HIV which is lifetime therapy, HCV treatment is 8 or 12 weeks and then its over, so designing support services programs are very time limited. BUT this will take a commitment from state & federal officials to spend resources and money & time to do this. And that is whats lacking - the willingness to make this commitment, and of course the political will, because HCV affected communities do not have the political juice that HIV had and still has.
Download the PDF here
----------------------------------
HCV eradication induced by direct-acting antiviral agents reduces the risk of hepatocellular carcinoma
published Jan 2018 Jnl of Hepatology, as presented at AASLD....
Eradication of HCV induced by DAAs is associated with a 71% reduction in HCC risk - (10/24/17)
--------------------------
Highlights
⋅DAA-induced SVR is associated with a 71% reduction in HCC risk.
⋅SVR is associated with a similar reduction in HCC risk no matter what regimen is used to achieve it.
⋅Treatment with DAAs is not associated with increased HCC risk compared with interferon.
Background & Aims
It is unclear whether direct-acting antiviral (DAA) treatment-induced sustained virologic response (SVR) reduces the risk of hepatocellular carcinoma (HCC) in patients with HCV infection. Therefore, in the current study, our aim was to determine the impact of DAA-induced SVR on HCC risk.
Methods
We identified 62,354 patients who initiated antiviral treatment in the Veterans Affairs (VA) national healthcare system from 1 January 1999 to 31 December 2015, including 35,871 (58%) interferon (IFN)-only regimens, 4,535 (7.2%) DAA + IFN regimens, and 21,948 (35%) DAA-only regimens. We retrospectively followed patients until 15 June 2017 to identify incident cases of HCC. We used Cox proportional hazards regression to determine the association between SVR and HCC risk or between type of antiviral regimen (DAA-only vs. DAA + IFN vs. IFN-only) and HCC risk.
Results
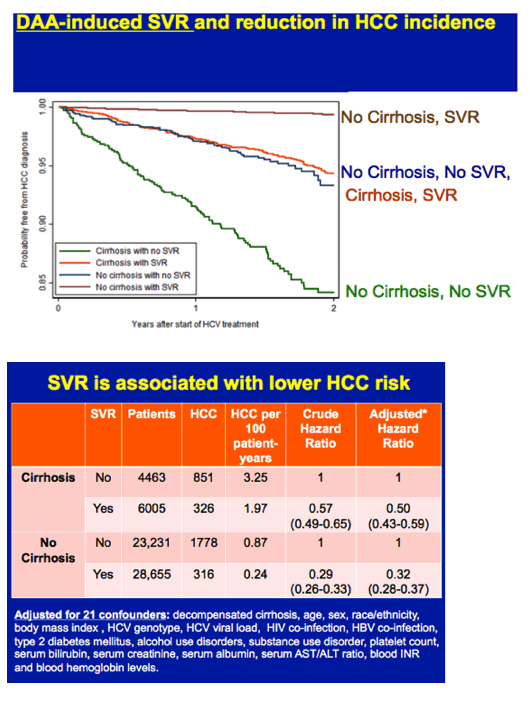
We identified 3,271 incident cases of HCC diagnosed at least 180 days after initiation of antiviral treatment during a mean follow-up of 6.1 years. The incidence of HCC was highest in patients with cirrhosis and treatment failure (3.25 per 100 patient-years), followed by cirrhosis and SVR (1.97), no cirrhosis and treatment failure (0.87), and no cirrhosis and SVR (0.24). SVR was associated with a significantly decreased risk of HCC in multivariable models irrespective of whether the antiviral treatment was DAA-only (adjusted hazard ratio [AHR] 0.29; 95% CI 0.23-0.37), DAA + IFN (AHR 0.48; 95% CI 0.32-0.73) or IFN-only (AHR 0.32; 95% CI 0.28-0.37). Receipt of a DAA-only or DAA + IFN regimen was not associated with increased HCC risk compared with receipt of an IFN-only regimen.
Conclusions
DAA-induced SVR is associated with a 71% reduction in HCC risk. Treatment with DAAs is not associated with increased HCC risk compared with treatment with IFN.
Lay summary
It was unclear whether direct-acting antiviral treatment-induced sustained virologic response reduces the risk of liver cancer in patients with HCV infection. We demonstrated that eradication of HCV infection with direct-acting antiviral agents reduces the risk of liver cancer by 71%.
Introduction
Eradication of HCV might be expected to reduce the risk of hepatocellular carcinoma (HCC) by preventing the future development of cirrhosis or by reversing early cirrhosis, a major risk factor for HCC. In addition, HCV might itself promote carcinogenesis,1 such that its eradication directly decreases HCC risk. Indeed, a meta-analysis of 18 studies of interferon (IFN)-based antiviral treatments for HCV suggested that sustained virologic response (SVR) was associated with reduced risk of HCC (relative risk 0.24; 95% CI 0.18-0.31).2 IFN-based treatments have now been replaced by direct-acting antiviral agents (DAAs). It is unclear whether the impact of SVR on HCC risk is different depending on whether SVR is achieved with either IFN-based regimens or DAAs. Surprisingly, recent studies suggested little or no impact of DAA-based antiviral treatment on HCC risk and even reported that DAAs might increase the risk of HCC recurrence.[3], [4], [5], [6], [7], [8] However, these studies were grossly underpowered, had limited follow-up time, mostly studied HCC recurrence rather than incidence, and did not compare those who achieved SVR because of DAAs to those who did not with respect to HCC risk.
In the current study, our aim was to determine the extent to which eradication of HCV with DAA-based treatments was associated with reduction in the risk of HCC, and whether this association was different for SVRs achieved by DAA vs. IFN-based regimens. We also aimed to determine whether receipt of DAA-based treatment compared with IFN-based treatment was associated with HCC risk.
Results
Characteristics of study population
Among the 62,354 patients who initiated their first antiviral regimen from 1 January 1999 to 31 December 2015, 34,660 (55.6%) achieved SVR. The antiviral treatments included 35,871 (58%) IFN-only regimens, 4,535 (7.3%) DAA + IFN regimens, and 21,948 (35%) DAA-only regimens. The distribution of different DAA regimens is shown (Table 1).
SVR rates were highest in the DAA-only regimens (90.7%), followed by the DAA + IFN regimens (60.9%), and lowest in the IFN-only regimens (33.4%). Patients were mostly male (96.6%) and the majority were White (55.6%), although there was a significant representation of other racial/ethnic groups. Mean age was 55.8 years and 16.8% had cirrhosis, 4.7% decompensated cirrhosis, and 1.1% had undergone liver transplantation. Genotype 1 HCV infection predominated (77.4%) followed by genotypes 2 (13.5%), 3 (8.3%), and 4 (0.8%).
Among those on an 'IFN-only' regimen, patients who achieved SVR were more likely to have genotype 2 or 3 HCV and less likely to have diabetes, cirrhosis, or markers of advanced fibrosis and/or cirrhosis (e.g. low platelet count or low albumin level) compared with patients who did not achieve SVR (Table 2). Among those on a DAA-only regimen, patients who achieved SVR were more likely to have genotype 1 infection and also less likely to have diabetes, cirrhosis, or markers of advanced fibrosis and/or cirrhosis compared with patients who did not achieve SVR.
Compared with patients who received IFN-only regimens, those patients who received DAA-only or DAA + IFN regimens were older, more likely to be Black, less likely to have genotype 2 or 3 HCV, and more likely to have advanced fibrosis or cirrhosis (Table 2). Also, compared with patients who received IFN-only regimens, those patients who received DAA-only or DAA + IFN regimens had lower mean serum platelet count and albumin level.
Association between SVR and HCC risk
Out of the 62,354 patients in our study, we identified 3,271 incident cases of HCC diagnosed more than 180 days after initiation of the first antiviral regimen during a mean follow-up of 6.1 years. HCC incidence was lower in the patients who achieved SVR (0.43 per 100 patient-years) than in treatment failures (1.14 per 100 patient-years) (Table 3).
Patients who achieved SVR had a lower incidence of HCC compared with those who did not achieve SVR among both patients with cirrhosis (1.97 vs. 3.25 per 100 patient-years) and those without (0.24 vs. 0.87 per 100 patient-years), also illustrated by the Kaplan-Meier curves (Fig. 1). The incidence of HCC was highest in patients with cirrhosis and treatment failure (3.25), followed by cirrhosis and SVR (1.97), and no cirrhosis and treatment failure (0.87), and was lowest in patients with no cirrhosis and SVR (0.24) (Table 3 and Fig. 1A). The same pattern was observed among all patients (Fig. 1A), as well as among the subgroups treated with IFN-only (Fig. 1B), DAA + IFN (Fig. 1C) and DAA-only (Fig. 1D).
Among all patients, SVR was associated with a 61% reduction in the risk of HCC (adjusted hazard ratio [AHR] 0.39; 95% CI 0.35-0.43) in multivariable analyses adjusting for baseline confounders (Table 3). SVR was associated with a decrease in HCC risk among both patients with cirrhosis (AHR 0.50; 95% CI 0.43-0.59) and patients without cirrhosis (AHR 0.32; 95% CI 0.28-0.37).
SVR was associated with a similar and significantly decreased risk of HCC in multivariable models irrespective of whether the antiviral treatment was IFN-only (AHR 0.32; 95% CI 0.28-0.37), DAA + IFN (AHR 0.48; 95% CI 0.32-0.73) or DAA-only (AHR 0.29; 95% CI 0.23-0.37) (Table 3).
In secondary analyses limited to only two years of follow-up, performed because mean follow-up was inevitably shorter in the DAA-only group (1.53 years) and DAA + IFN group (3.6 years) than in the IFN-only group (9.1 years), SVR was still associated with a significantly reduced risk of HCC irrespective of whether the antiviral treatment was IFN-only (AHR 0.56; 95% CI 0.38-0.81), DAA + IFN (AHR 0.49; 95% CI 0.27-0.86), or DAA-only (AHR 0.28; 95% CI 0.22-0.35) (Table S3).
Also, in secondary analyses in which we looked at the regimen that achieved SVR or a clustered analysis of all antiviral regimens, instead of the first antiviral regimen, SVR was similarly associated with a reduction in HCC risk (Table S4).
Association between type of antiviral treatment regimen and HCC risk
Although HCC incidence was higher after DAA-only treatment (1.32 per 100 patient-years) than after DAA + IFN (1.06 per 100 patient-years) or IFN-only (0.81 per 100 patient-years) treatment, there was no significant association between treatment regimen and HCC risk after adjusting for important confounders (Table 4). This was because of the higher prevalence in the DAA-only group of risk factors for HCC, such as cirrhosis, advanced age, diabetes, low platelet count, and low serum albumin level. When analysing separately patients with or without cirrhosis during the period 2009-2015, there was little difference in HCC incidence by antiviral treatment group and no association between antiviral treatment group and HCC risk. Also, when limiting follow-up to two years, there was no association between antiviral treatment group and HCC risk (Table 4).
Discussion
Most patients with HCV in the USA will undergo DAA-based antiviral treatment in the next few years and the majority of them will achieve SVR. Our results suggest that DAA-induced SVR is associated with a 71% reduction in HCC risk (AHR 0.29; 95% CI 0.23-0.37) compared with treatment failure. The reduction in HCC risk associated with SVR was similar irrespective of whether SVR was achieved by DAA-only, DAA + IFN, or IFN-only regimens. This suggests that eradication of HCV reduces HCC risk independently of how it is achieved. In contrast to prior reports that suggested an increased HCC risk in patients treated with DAAs,[3], [7] we found that receipt of DAA-only antiviral treatment was not associated with increased risk of HCC compared with receipt of IFN-only antiviral treatment.
It is still unclear whether DAA-induced SVR is associated with a reduction in HCC risk. It might seem reasonable to expect that HCV eradication would reduce HCC risk by preventing or reversing cirrhosis, or by eliminating any direct carcinogenic effect of HCV. However, recent studies have demonstrated little or no impact of DAA-based antiviral treatment on HCC risk and even suggest that DAAs increase the risk of HCC recurrence.[3], [4], [5], [6], [7], [8] A recent retrospective Spanish study reported an unexpectedly high rate of HCC recurrence (27.6% after a median follow-up of 5.7 months) following HCV antiviral treatment with DAAs in 58 patients with HCC who had achieved complete response before antiviral treatment,3raising concerns that DAAs somehow promote HCC recurrence. However, a French prospective cohort study found a similar risk of HCC recurrence when comparing 189 patients who received DAAs (24/189 or 12.7% with recurrence, incidence 0.73 per 100 patient-months) to 78 who did not receive DAAs (16/78 or 20.5% with recurrence, incidence 0.66/100 patient-months).4 Finally, an Italian study reported that, after DAA therapy, 9 out of 285 patients with cirrhosis without prior HCC developed new HCC during a 24-week follow-up, whereas 17 out of 59 patients with prior HCC had recurrence; the authors concluded that 'DAA-induced resolution of HCV infection does not seem to reduce the occurrence of HCC'.5 However, these studies were underpowered, had limited follow-up time, studied mostly HCC recurrence rather than incidence, and did not compare those who achieved SVR resulting from DAAs with those who did not with respect to HCC risk.
In contrast to these studies, we found that DAA-induced SVR was associated with a 71% reduction in the risk of HCC and that an approximately similar reduction in HCC risk was associated with SVR induced by DAA-only (AHR 0.29; 95% CI 0.23-0.37), DAA + IFN (AHR 0.48; 95% CI 0.32-0.73) or IFN-only (AHR 0.32; 95% CI 0.28-0.37) regimens. This suggests that eradication of HCV reduces the risk of HCC irrespective of the antiviral regimen used to achieve it. However, our study was designed to address the impact of SVR on HCC incidence, not HCC recurrence. Hence, patients with a history of HCC before antiviral treatment were excluded and our study offers no insight into the impact of antiviral treatment on HCC recurrence.
We observed a lower relative reduction in HCC risk associated with SVR in patients with cirrhosis (AHR 0.50; 95% CI 0.43-0.59) than in patients without cirrhosis (AHR 0.32; 95% CI 0.28-0.37). However, the absolute reduction in HCC risk was greater in patients with cirrhosis (from 3.25 to 1.97 per 100 patient-years) than in patients without (from 0.73 to 0.18 per 100 patient-years), as would be expected given the higher baseline HCC risk of patients with cirrhosis. It is generally believed that HCC risk in patients with HCV increases dramatically once they develop cirrhosis, but a lower HCC risk is still present in pre-cirrhotic liver disease, especially in advanced fibrosis. The risk of HCC in patients with HCV but without cirrhosis might be explained by the presence of occult cirrhosis at least in parts of the liver, progression from advanced fibrosis to cirrhosis during follow-up, or the development of HCC in pre-cirrhotic advanced fibrosis. It is likely that in both patients with and without cirrhosis, SVR reduces HCC risk at least in part by reversing hepatic fibrosis.
The best way to determine whether treatment with DAAs affects HCC risk is to randomise patients to treatment with DAAs vs. no treatment and then follow them up for a long period of time. Such a study design would be unethical. Another possibility would be to compare patients who received DAAs as part of their routine clinical care to those who were untreated with respect to HCC risk. However, this approach is subject to considerable ascertainment bias (the untreated patients are less likely to be found to have HCC in a given time frame than the treated patients because of less screening and less contact with hepatologists and other medical providers) and selection bias (the reasons why certain patients are not treated with DAAs despite their excellent SVR rate might be difficult to determine and accurately adjust for), and confounding by indication (the indication for offering antiviral treatment might be associated with both treatment and outcome). Instead, we compared patients treated with DAAs in the 'current era' to patients treated with IFN in the IFN era to avoid as much as possible these sources of bias. However, this comparison could still be biased by the fact that patients with more advanced liver disease are now candidates for DAAs who were not candidates for IFN and these patients have significantly higher HCC risk. We believe that our adjustment for baseline characteristics adequately accounted for this potential confounding. In addition, the incidence of HCC has been increasing over time25 and was lower during the IFN era than during the DAA era. We minimised this source of bias by limiting analyses to regimens initiated from 2009 to 2015, as well as by adjusting for baseline characteristics that are at least partly responsible for the increasing HCC incidence over time. Any residual bias would be in favour of showing increased HCC risk in the patients treated with DAA only; therefore, it is reassuring that no such association was found.
Our study was limited by the relatively short follow-up in the DAA-only group, although we extended follow-up for HCC incidence to a time as close as possible to the time of manuscript preparation (June 2017) to maximise follow-up time. This yielded a substantial mean follow-up time of 1.53 years even in the DAA-only group (with follow-up time starting 180 days after antiviral treatment initiation). It was reassuring that similar associations between SVR and HCC risk were observed among the DAA + IFN and the IFN-only groups, which had much longer follow-up than the DAA-only group. The association between SVR and HCC risk might not be causative: it is theoretically possible that unknown factors that lead to treatment failure might also lead to the future development of HCC, other than the 21 baseline characteristics that we carefully adjusted for that included known markers of advanced fibrosis and/or cirrhosis and risk factors for HCC. It is also possible that HCC was present but undiagnosed at the time of antiviral treatment, also leading to reduced SVR. However, we excluded all HCCs that presented within 180 days of antiviral treatment initiation; in addition, the HCC cumulative incidence curves in the SVR and treatment failure groups continued to diverge for many years after treatment (Fig. 1). A final limitation of the study is that the ICD-10 code for HCC (C22.0) that replaced the ICD-9 code for HCC (155.0) in October 2015 is not yet validated using VA data. However, given that a single ICD-10 code directly replaced a single ICD-9 code, it is reasonable to expect a similarly high positive predictive value.
The substantial strengths of the study include the large sample size, large number of incident HCCs, and long follow-up time. Baseline characteristics necessary for multivariable analyses to adjust for potential confounding were available. All patients were derived from a single, national healthcare system with fairly uniform antiviral treatment practices and guidelines across its facilities.
In conclusion, DAA-induced SVR is associated with a 71% reduction in HCC risk compared with treatment failure. Eradication of HCV is associated with a similar reduction in HCC risk irrespective of the regimen that is used to achieve the eradication. We found no evidence that treatment with DAAs was associated with increased risk of HCC compared with treatment with IFN.
| |
| |
| |
|
|
|