 |
 |
 |
| |
MK-8591 Protects 8 of 8 Macaques From Rectal SHIV 6 Days After Dosing
|
| |
| |
9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris
Mark Mascolini
Weekly injections of MK-8591, a nucleoside reverse transcriptase translocation inhibitor (NRTTI), repeatedly protected 8 of 8 macaques from intrarectal challenge with a simian HIV (SHIV109CP3) 6 days after dosing [1].
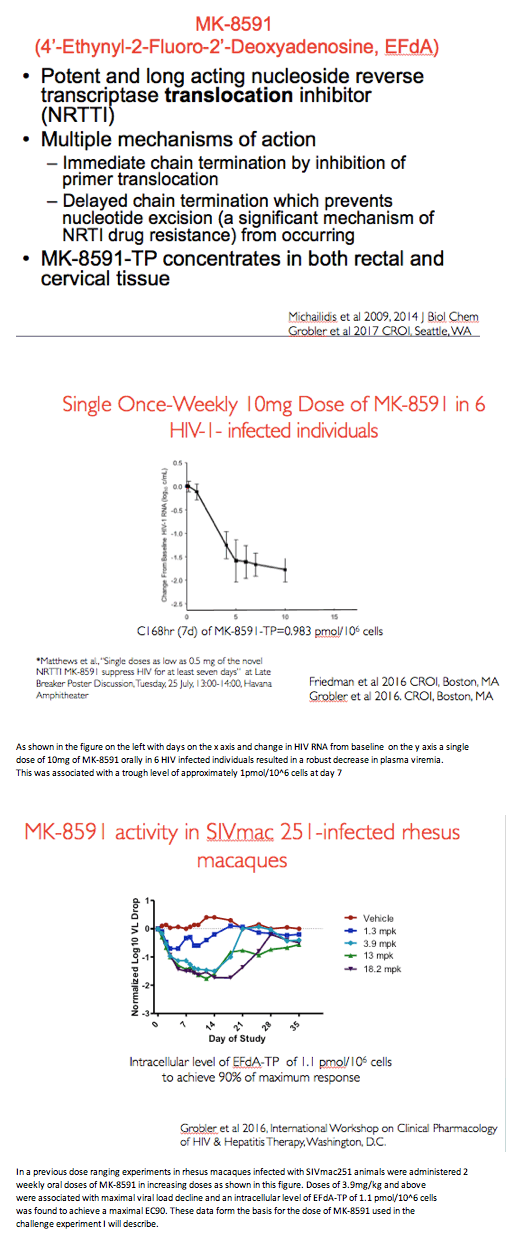
MK-8591, also known by the chemical shorthand EFdA, thwarts HIV by several mechanisms [2] and concentrates in both rectal and cervical tissue. In 6 people with HIV, a single 10-mg dose of the drug lowered viral load about 1.5 log10 at a day-7 trough level of approximately 1 pmol/million cells.
Researchers at New York's Aaron Diamond AIDS Research Center, Merck, and Tulane National Primate Research Center conducted this study in 16 male macaques to test MK-8591 against weekly intrarectal SHIV challenge. Eight animals received weekly MK-8591 at an oral gavage dose of 3 mg/kg for up to 14 weeks, while 8 control animals received only an inert solution. Six days after dosing, researchers exposed all animals to intrarectal SHIV. Challenges continued weekly until animals became infected or received 12 challenges.
All 8 macaques not dosed with MK-8591 became infected, 6 of them after the first SHIV challenge, 1 after the second challenge, and 1 after the fourth challenge. All 8 MK-8591-treated macaques remained free of SHIV infection throughout all challenges and to the end of the 168-day study. The researchers calculated that animals treated with MK-8591 had a 41.5-fold lower risk of infection than untreated animals (P < 0.0001).
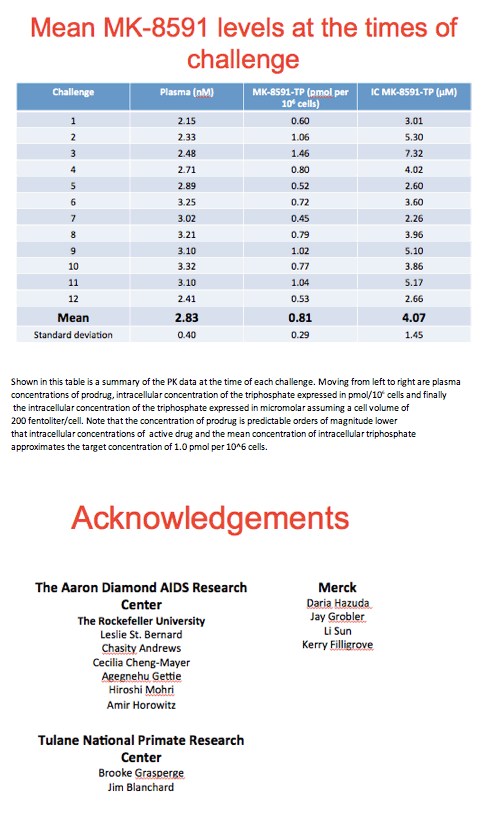
Proviral DNA could not be detected in peripheral blood mononuclear cells of any treated macaques but could be detected in all untreated animals. Virus-specific antibody responses remained undetectable in all treated macaques but were readily detected in untreated monkeys. Drug level monitoring at the time of SHIV challenge indicated that the average intracellular MK-8591 triphosphate concentration approximated a target concentration of 1.0 pmol/million cells.
The investigators noted that protective intracellular active MK-8591 concentrations can be attained in humans at low doses of the drug. They proposed that their findings "support the potential use of MK-8591 for HIV-1 prophylaxis in individuals at high risk of acquiring infection."
References
1. Markowitz M, Gettie A, St. Bernard L, et al. Weekly oral MK-8591 protects male rhesus macaques against repeated low dose intrarectal challenge with SHIV109CP3. 9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris. Abstract MOAX0203LB.
2. Michailidis E, Huber AD, Ryan EM, et al. 4'-Ethynyl-2-fluoro-2'-deoxyadenosine (EFdA) inhibits HIV-1 reverse transcriptase with multiple mechanisms. J Biol Chem. 2014;289:24533-24548. http://www.jbc.org/content/289/35/24533.long
Antiviral Activity of EFdA [MK-8591] Against NRTI-Sensitive and -Resistant Strains of HIV-2 - (02/24/17)
MK-8591 Concentrations at Sites of HIV Transmission and Replication - (02/23/17)
EFdA / MK-8591 - HIV pre-exposure prophylaxis for women and infants prevents vaginal and oral HIV transmission in a preclinical model of HIV infection - (08/05/16)
Pre-exposure Prophylaxis with EFdA Offers Strong Protection against High Dose Mucosal HIV Challenges - [Long-Acting New NRTI for Treatment & PrEP] - (10/24/16)
Efficacy of once-weekly MK-8591 in SIV infected rhesus macaques - (06/14/15)
CROI:Long-Acting Oral and Parenteral Dosing of MK-8591 for HIV Treatment or Prophylaxis - (02/24/16)
CROI:A Single Monotherapy Dose of MK-8591,a Novel NRTI, Suppresses HIV for Ten Days - (02/24/16)



|
| |
|
 |
 |
|
|