 |
 |
 |
| |
Darunavir/Ritonavir Plus 3TC Noninferior to Triple Therapy at 24 Weeks - ANDES Study - A Phase 4, Randomized, Open Label, Study of a Ritonavir-Boosted Darunavir in Fixed Dose Combination (FD-DRV/r) plus Lamivudine Versus FD-DRV/r plus Lamivudine/Tenofovir in Naïve HIV-1 Infected Subjects
|
| |
| |
9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris
Mark Mascolini
A generic fixed-dose combination of darunavir/ritonavir (DRV/r) plus lamivudine (3TC) proved noninferior to generic DRV/r plus tenofovir (TDF)/3TC) at 24 weeks in previously untreated people [1]. The ANDES study will continue through 48 weeks [2].
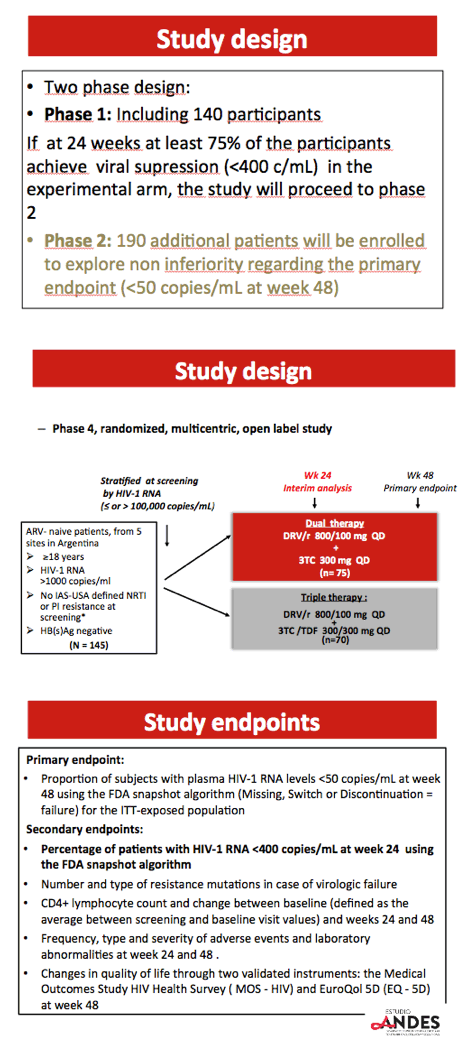
Previous studies of two-drug regimens showed noninferiority to standard triple combinations in people starting their first regimen or achieving viral control with a standard regimen (GARDEL, OLE, ATLAS-M, DUAL). Argentina has licensed a generic fixed-dose combination of DRV/r (800/100 mg once daily) for treatment-naive people. ANDES aimed to compare generic DRV/r plus 3TC with DRV/r plus TDF/3TC in two phases: The first phase compared the two regimens in 140 participants. If at least 75% in the experimental arm reached a viral load below 400 copies by 24 weeks, the second phase would add 190 participants with a primary endpoint comparing viral suppression (below 50 copies) at 48 weeks.
All participants were at least 18 years old, had a detectable viral load, and had no nucleoside or protease inhibitor mutations at screening. Researchers randomized them to dual therapy (DRV/r plus 3TC) or triple therapy (DRV/r plus TDF/3TC) at standard doses. They used the FDA snapshot algorithm to determine a secondary endpoint of the two-phase trial--the proportion with a viral load below 400 copies at week 24.
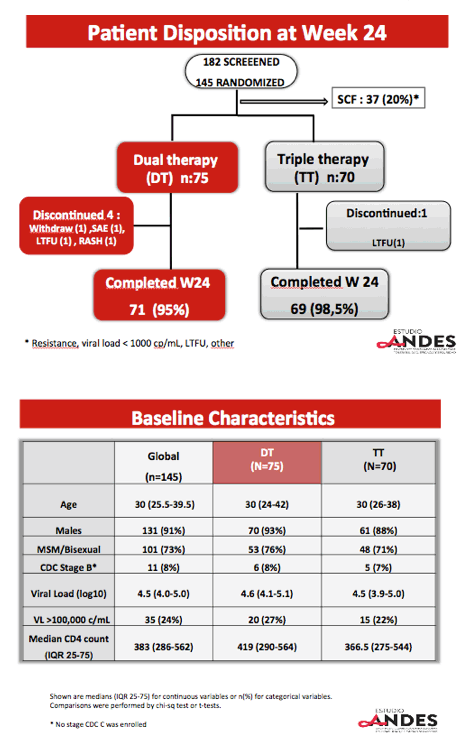
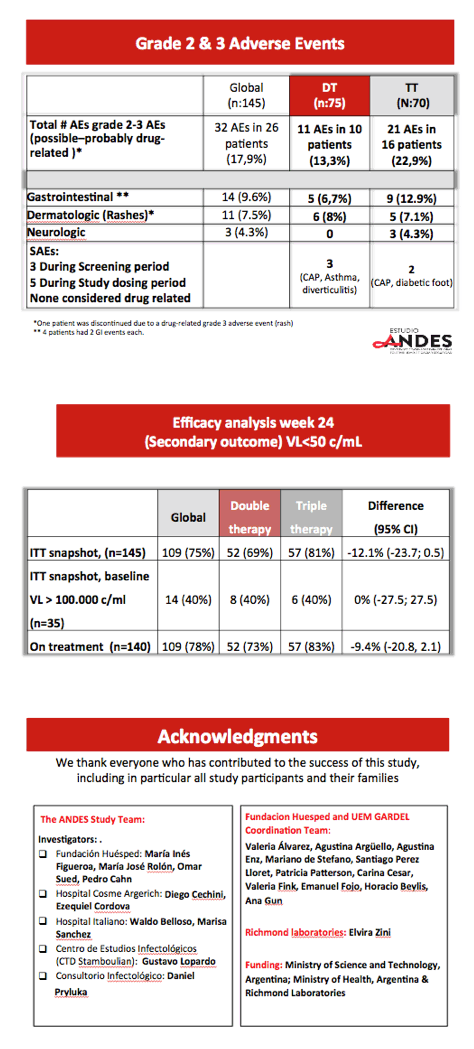
Researchers randomized 75 participants to dual therapy and 70 to triple therapy. Four people in the dual-therapy group stopped treatment, 1 because of a serious adverse event (rash). One person dropped out of the triple-therapy arm. The groups did not differ substantially in median age (30 in both groups), proportion of men (91% overall), baseline viral load (median 4.5 log10 overall, about 32,000 copies), or median CD4 count (383 overall). About one quarter in each study arm had a pretreatment viral load above 100,000 copies.
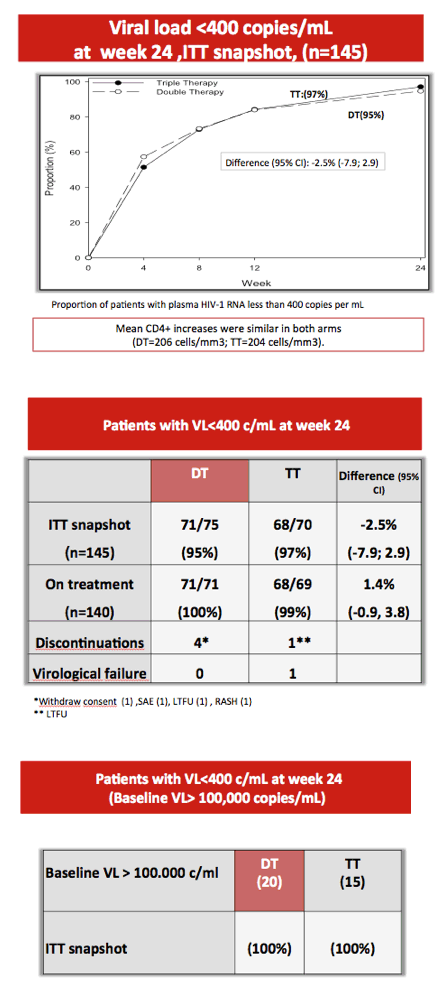
After 24 weeks 95% in the dual-therapy group and 97% in the triple-therapy group had a viral load below 400 copies (difference -2.5%, 95% CI -7.0 to 2.9), a result establishing the noninferiority of dual therapy to triple therapy in previously untreated people. Median CD4 gains measured 206 with dual therapy and 204 with triple therapy. One person in the triple-therapy arm and none in the dual-therapy arm had protocol-defined virologic failure. Everyone with a pretreatment load above 100,000 copies had a load below 400 copies at week 24.
The rate of grade 2 or 3 adverse events possibly or probably related to study drugs was higher with triple therapy than dual therapy (22.9% versus 13.3%). Gastrointestinal and neurologic complaints proved more frequent in the triple-therapy arm. Similar proportions in the double-therapy and triple-therapy arms had rash (8% and 7.1%).
The trial has begun its second phase [2]. If DRV/r plus 3TC continues to exert virologic control through 48 weeks, the researchers suggested it "may provide further evidence about the potential efficacy of dual therapy based on 3TC and a drug with a high genetic barrier" to resistance.
References
1. Sued O, Figueroa MI, Gun A, et al. Dual therapy with darunavir/ritonavir plus lamivudine for HIV-1 treatment initiation: week 24 results of the randomized ANDES study. 9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris. Abstract MOAB0106LB.
2. ClinicalTrials.gov. Darunavir/ritonavir + lamivudine versus darunavir/ritonavir +emtricitabine/tenofovir in naive HIV-1 infected subjects (ANDES). ClinicalTrial.gov identifier NCT02770508. https://clinicaltrials.gov/ct2/show/NCT02770508


|
| |
|
 |
 |
|
|