 |
 |
 |
| |
Impact of microbiota on female genital
tissue and plasma concentrations of dapivirine
|
| |
| |
Reported by Jules Levin
IAS Science 2017 Paris France July 23-26
Sharon Hillier
Authors
S. Hillier1,2, L. Meyn1, K. Bunge1, M. Austin2, B. Moncla1,2, C. Dezzutti1,2, B. Devlin3, M. Marzinke4, C. Hendrix4, L. Rohan2,5
Institutions
1University of Pittsburgh, Obstetrics, Gynecology and Reproductive Sciences, Pittsburgh, United States, 2Magee-Womens Research Institute, Pittsburgh, United States, 3International Partnership for Microbicides, Silver Spring, United States, 4Johns Hopkins University, Baltimore, United States, 5University of Pittsburgh, Pharmaceutical Science, Pittsburgh, United States

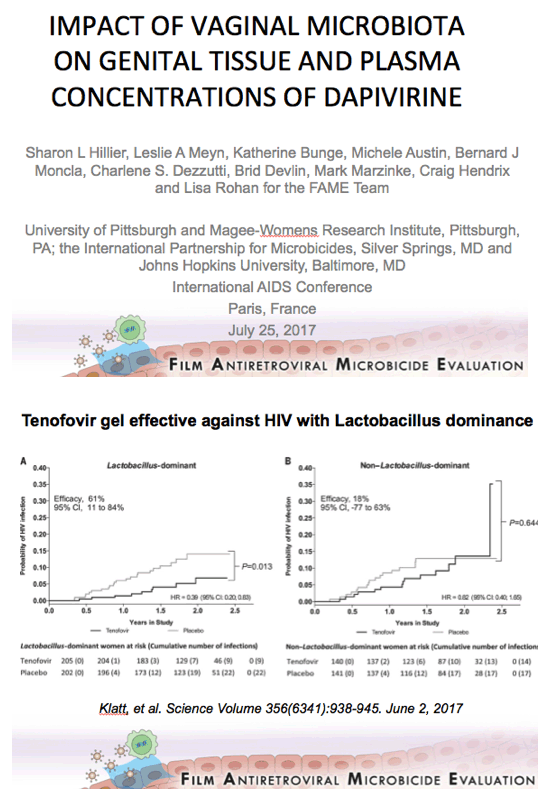
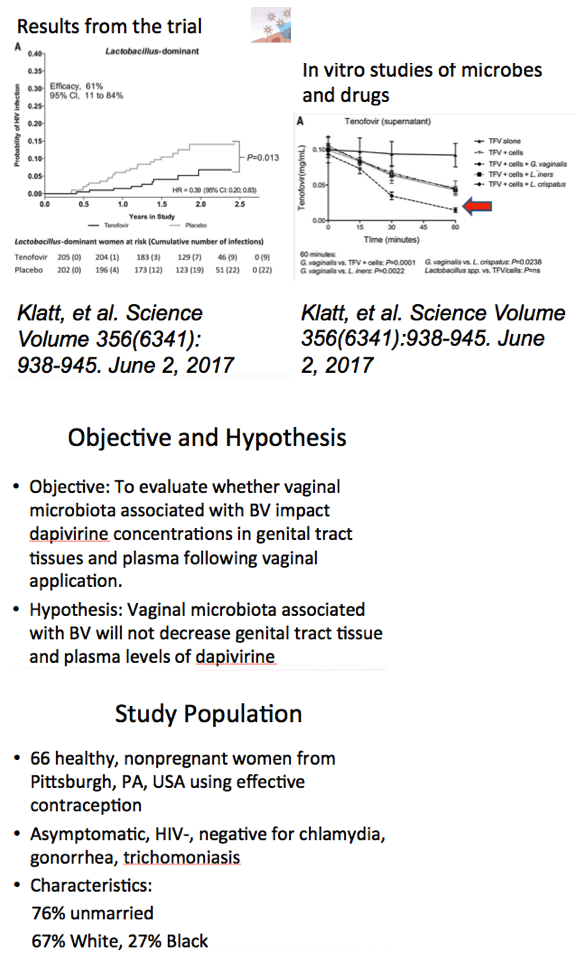
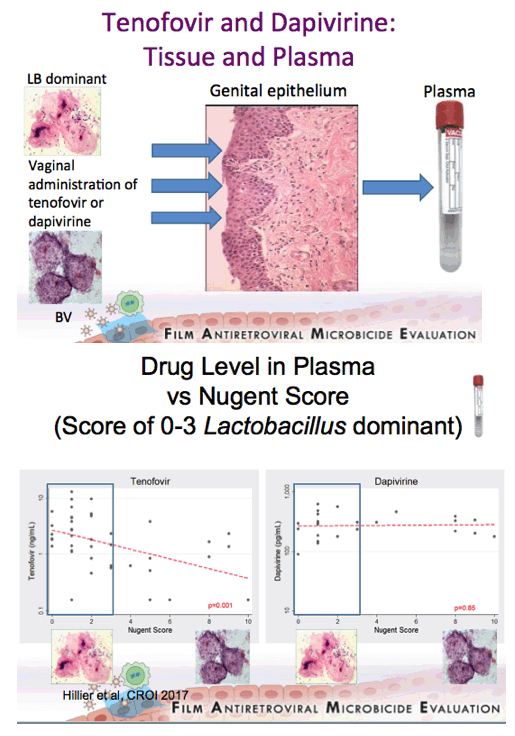
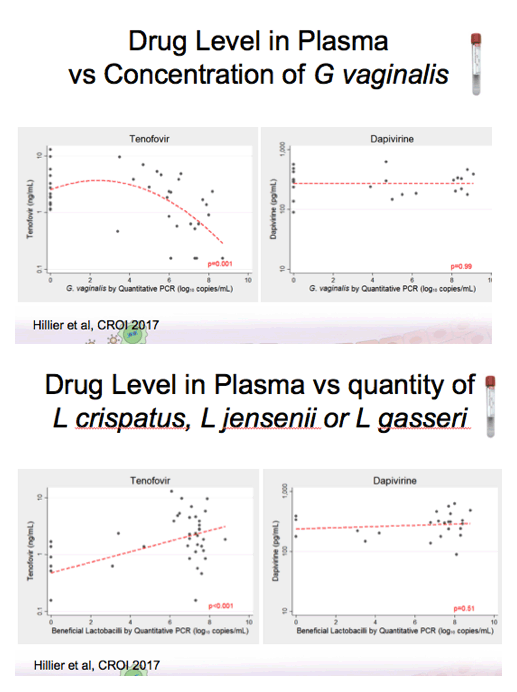
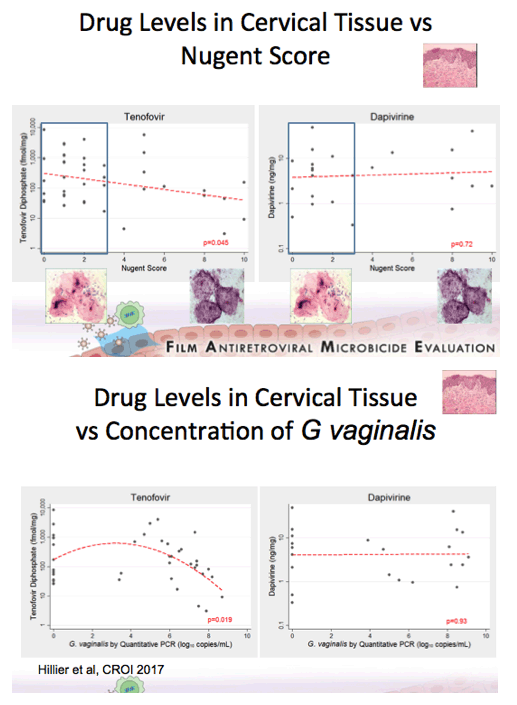
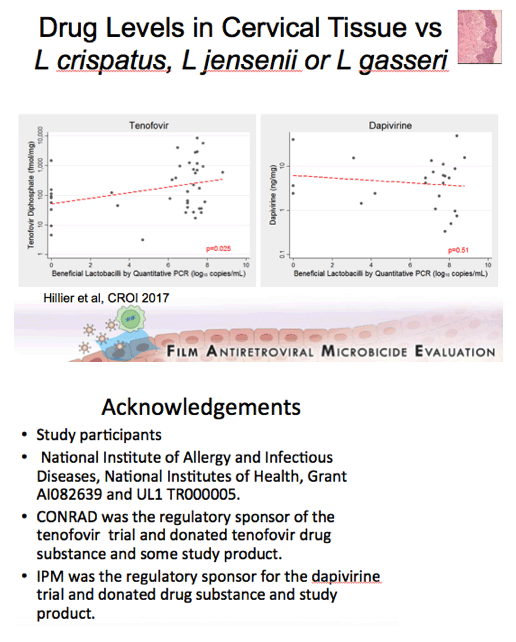
Background: Women having a non-Lactobacillus dominant vaginal microbiota enrolled in CAPRISA 004 had lower detection of tenofovir (TFV) in cervicovaginal lavage (CVL) fluid than women having a Lactobacillus dominant microbiota. In FAME-04, decreased concentrations of TFV diphosphate in genital tissues and TFV in the plasma were highly correlated to higher Nugent score and increased vaginal concentrations of Gardnerella vaginalis and Atopobium vaginae. Vaginal rings containing dapivirine, a nonnucleoside reverse transcriptase inhibitor, have been shown to reduce incident HIV. The objective of this secondary analysis was to evaluate whether vaginal microbiota associated with bacterial vaginosis similarly impacted dapivirine concentrations in genital tract tissues and plasma following vaginal application.
Methods: 24 healthy HIV negative women (mean age 27, 58% white) used either dapivirine 0.05% gel (1.25 mg) or films (1.25 mg) for 6 days at home. On the 7th day, women inserted the final dose in the clinic with confirmation of correct product placement. Two hours later, cervical and vaginal biopsies along with CVL and plasma were obtained for dapivirine quantification using a validated liquid chromatography tandem mass spectrometry assay. Vaginal samples for diagnosis of bacterial vaginosis using the Nugent criteria and quantitative polymerase chain reaction (qPCR) detection of G. vaginalis and A. vaginae were collected prior to product use. The relationship between vaginal microbiota and dapivirine levels was assessed using linear regression models.
Results:There was no association between increasing concentrations of G. vaginalis in the vagina detected by qPCR and dapivirine concentrations in vaginal tissue, cervical tissue, CVL, or plasma (p=0.45, 0.93, 0.51, 0.99 respectively). Similarly, vaginal concentrations of A. vaginae were not associated with dapivirine concentrations in CVL, vaginal and cervical tissues or plasma (p≥0.31). Nugent criteria associated with bacterial vaginosis were not associated with lower CVL, tissue or plasma concentrations of dapivirine (p≥0.19).
Conclusions: In contrast to tenofovir, genital and plasma concentrations of dapivirine, were not impacted by increasing concentrations of vaginal bacteria associated with bacterial vaginosis. While replication of these results is needed, these data suggest that the levels of dapivirine following vaginal application should not be impacted by the microbiota associated with bacterial vaginosis.

IMPACT OF VAGINAL MICROBIOTA ON GENITAL TISSUE AND PLASMA CONCENTRATIONS OF TENOFOVIR - (03/10/17).....http://www.natap.org/2017/CROI/croi_174.htm
|
| |
|
 |
 |
|
|