 |
 |
 |
| |
Successful switch to once-daily single-tablet regimen (STR) containing elvitegravir (EVG) or dolutegravir (DTG) in virologically suppressed HIV-1 infected patients despite archived resistant quasispecies
|
| |
| |
Reported by Jules Levin
9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris
C. Pronier1,2, A. Maillard1, P. Comacle1, C. Cameli3, J. Rohan3, T. Jovelin3, G. Lagathu1, M. Dupont4, F. Fily4, M. Baldeyrou5, J.-M. Chapplain5, P. Fillatre5,
M. Revest5, F. Souala5, P. Tattevin5, C. Michelet5, C. Arvieux3,5, V. Thibault1,2
1CHU Pontchaillou, Virology, Rennes, France, 2Rennes 1 University, IRSET, Rennes, France, 3COREVIH Bretagne, Rennes, France, 4GHT Rance Emeraude, Infectious Disease,
Saint-Malo, France, 5CHU Pontchaillou, Infectious Disease, Rennes, France

abstract
Background: The main objective of this study was to assess switches with integrase strand-transfer inhibitor (INSTI)-based regimen STR therapy in ARV-treated patients with undetectable HIV-1 viral load (VL).
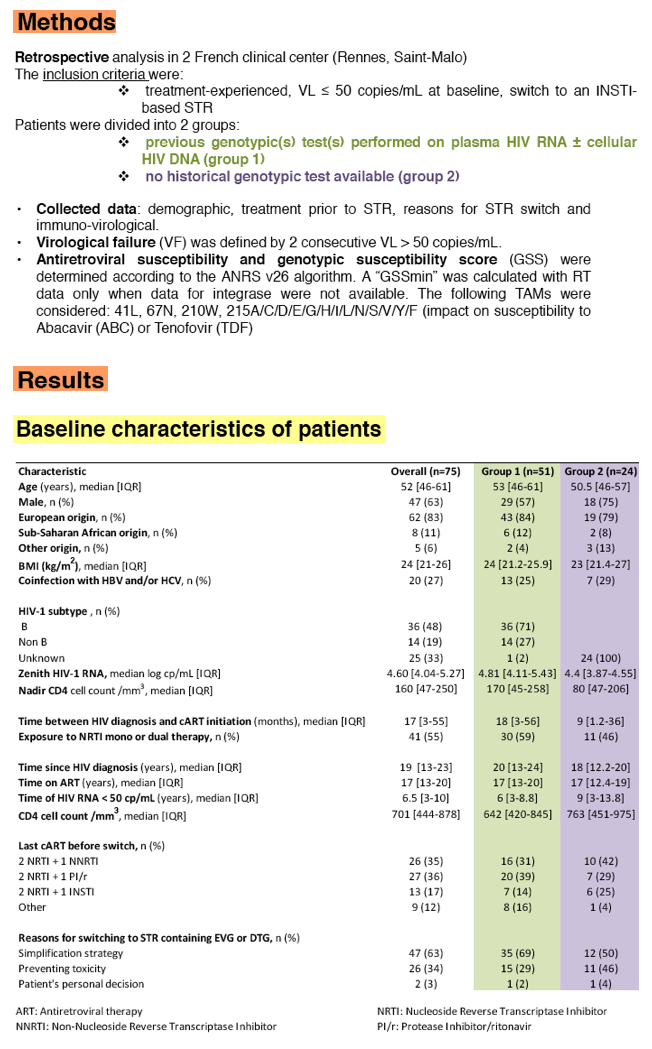
Methods: A retrospective analysis was conducted on 75 HIV-1 infected,
treatment-experienced adults from 2 French centers (Rennes, Saint-Malo). The inclusion criteria were:
i) treatment-experienced,
ii) VL≤ 50 copies/mL at baseline and
iii) switch to an INSTI-based STR.
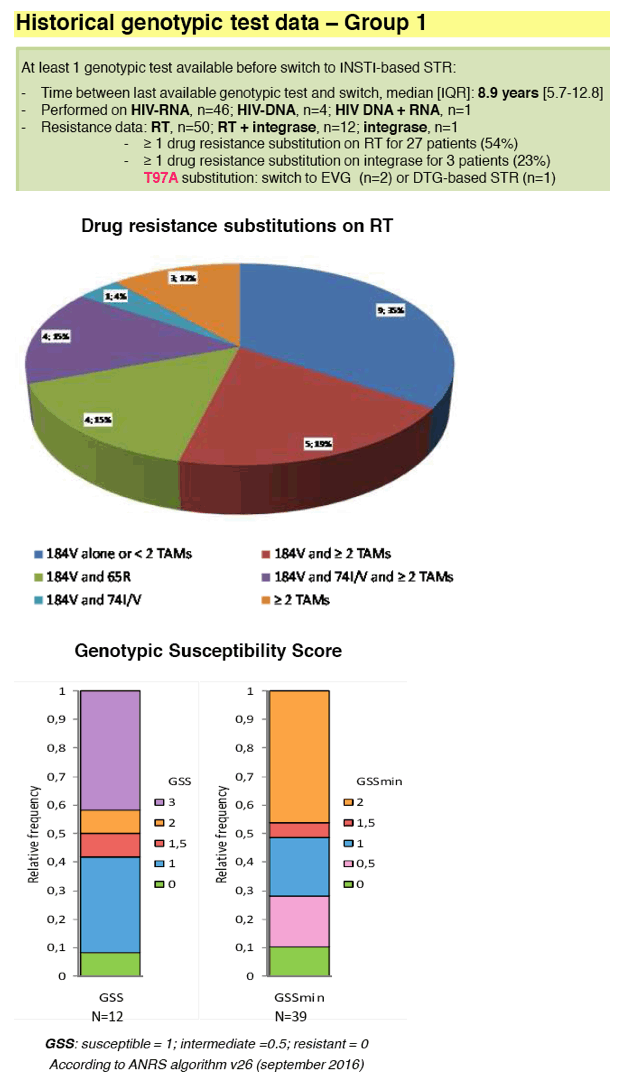
Patients were divided into two groups: previous genotypic(s) test(s) performed on plasma HIV RNA and/or cellular HIV DNA (group 1; n=51, 68%) and no historical genotypic test available (group 2; n=24, 32%). Following data were collected: demographic, treatment prior to STR, reasons for STR switch and immuno-virological data. Virological failure (VF) was defined by 2 consecutive VL >50 copies/mL. Antiretroviral susceptibility and genotypic susceptibility scores (GSS) were determined according to the ANRS v26 algorithm.
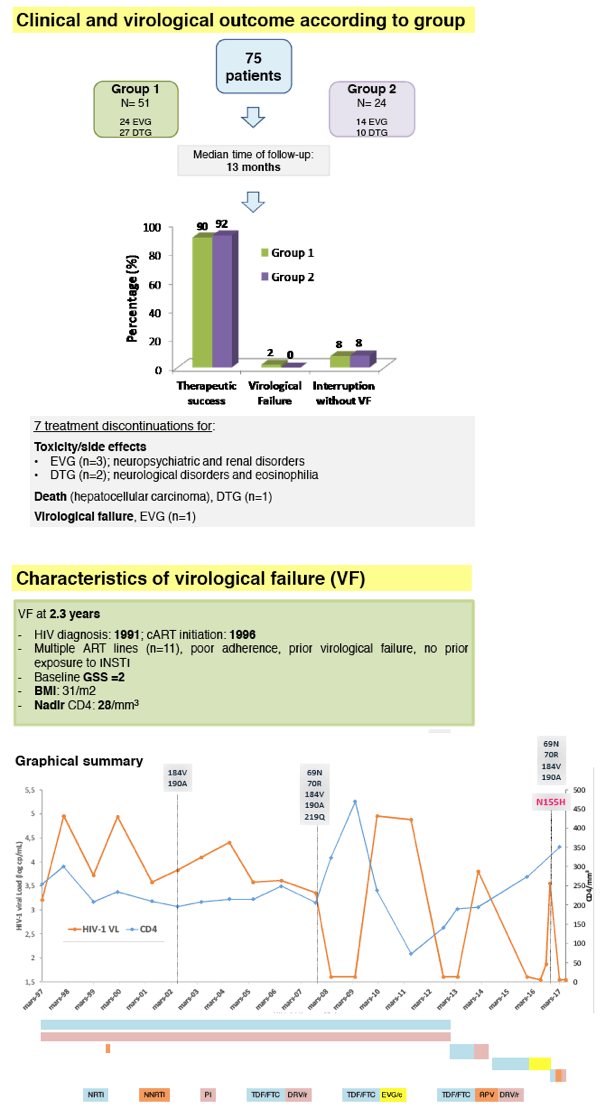
Results: Seventy-five patients were included, mostly men (n=47, 63%), median age of 52 years. EVG or DTG-based STR were prescribed in 38 and 37 patients, respectively. Patients were heavily pre-treated for a median duration of 18 years and virological success since a median of 7 years. Main reasons for switch were simplification (n= 47, 63%) and tolerability (n=26, 35%). Patients were followed-up for a median period of 13 months [range 1-84] after switching. Median CD4 count remained stable (701 vs 681 cells/mm3) at month 12. Seven patients discontinued the new regimen due to side effects (EVG; n=3, DTG; n=2), death (hepatocellular carcinoma; DTG) and VF (EVG), one patient each.
Twenty-seven out of 50 patients (54%) in group 1 displayed virus with ≥1 drug resistance substitution on reverse-transcriptase gene (M184V; n=22, K65R; n=4, L74I/V; n=5, ≥2 TAMs; n=13) and 3 out of 14 on integrase gene (T97A).
All patients, except one, remained virologically suppressed. VF was observed at month 28 in one patient (group 1), with a baseline GSS =2 and N155H substitution selected at VF.
Conclusions: This study reflecting routine clinical practice confirms the efficacy of INSTI-based STR in virologically suppressed, ARV-experienced patients, with a low prevalence of viral failure despite archived resistant quasispecies
|
| |
|
 |
 |
|
|