 |
 |
 |
| |
Olfactory Mucosa HIV RNA Correlates With CSF RNA in People On/Off ART
|
| |
| |
25th Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2018, Boston
Mark Mascolini
Olfactory mucosa HIV RNA correlated strongly with cerebrospinal fluid (CSF) RNA and plasma viral load in a study of 47 people on antiretroviral therapy (ART) or naive to ART [1]. Noninvasive nasal brushing to collect HIV RNA in olfactory mucosa cells could prove safer and more acceptable than lumbar puncture to sample CSF.
University of Torino researchers and collaborators noted that CSF represents an imperfect proxy of brain tissue and must be collected by lumbar puncture, an unpopular procedure not without risk. Because no risk factors or clinical signs reliably predict CSF escape (detectable HIV in CSF but not in plasma) or persistent central nervous system (CNS) activation or inflammation, these investigators explored olfactory mucosa (nasal passage lining) samples as an accessible CNS-derived tissue, a marker of activation and inflammation, and a possible replacement for CSF in neurologic studies.
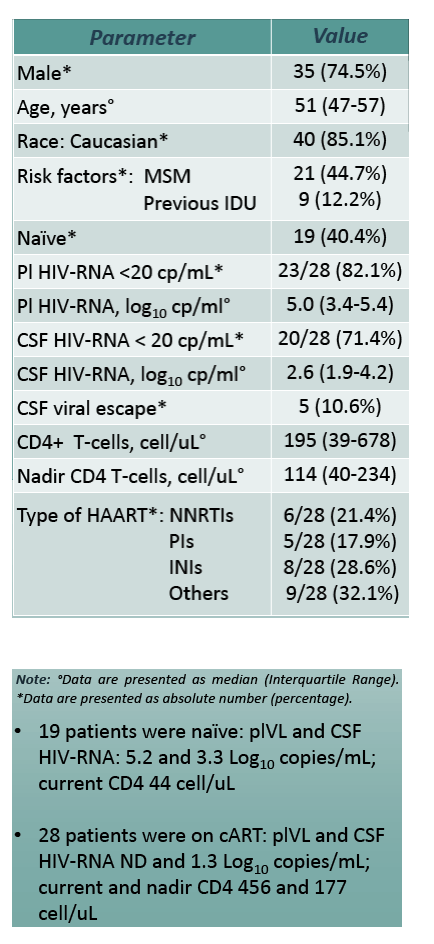
Olfactory mucosa samples can be collected by noninvasive nasal brushing, used in diagnosing certain diseases and the gold standard for Creutzfeldt-Jakob disease diagnosis. This analysis involved 47 HIV-positive people who had lumbar puncture for clinical reasons, followed within 72 hours by nasal brushing. Thirty-five participants (74%) were men and 40 (85%) were white. Median age stood at 51 and median CD4 count at 195. Among 19 antiretroviral-naive participants, median plasma and CSF loads were 5.2 and 3.3 log10 copies and median current CD4 count was 44. Among 28 antiretroviral-treated people, median CSF load measured 1.3 log10 copies, plasma viral load was undetectable, and median current CD4 count stood at 456.
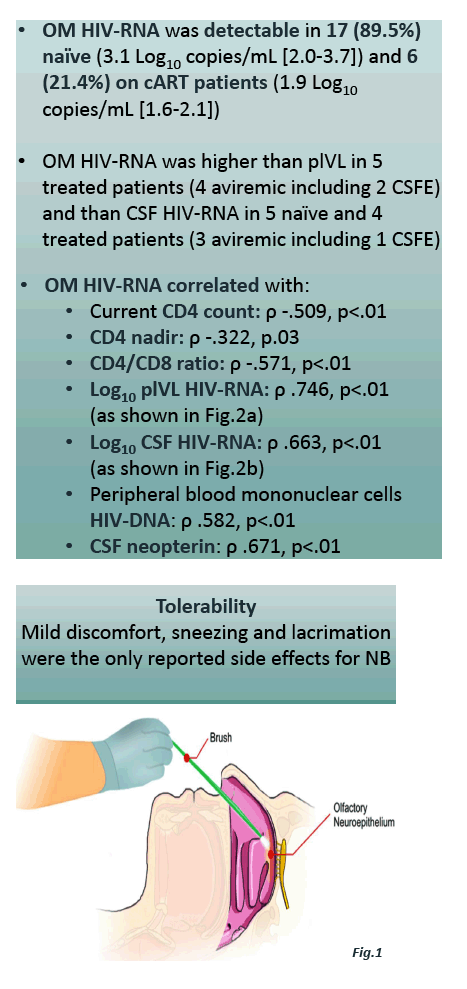
Participants tolerated nasal brushing well, reporting mild discomfort and some sneezing and tearing.
The researchers detected HIV RNA in 17 of 19 olfactory samples (89.5%) from antiretroviral-naive people versus 6 of 28 samples (21.4%) from people on treatment. Olfactory HIV RNA proved higher than plasma load in 5 treated people; and olfactory HIV RNA was higher than CSF RNA in 5 naive and 4 treated participants.
Olfactory mucosa HIV RNA correlated positively with CSF HIV RNA, CSF neopterin (an immune activation marker), plasma HIV RNA, and peripheral blood mononuclear cell (PBMC) HIV DNA:
-- Log CSF HIV RNA: rho = 0.663, P < 0.01
-- CSF neopterin: rho = 0.671, P < 0.01
-- Log plasma HIV RNA: rho = 0.746, P < 0.01
-- PBMC HIV DNA: rho = 0.582, P < 0.01
Olfactory mucosa HIV RNA correlated negatively with 3 markers:
-- Current CD4 count: rho = -0.509, P < 0.01
-- Nadir CD4 count: rho = -0.322, P = 0.03
-- CD4/CD8 ratio: rho = -0.571, P < 0.01
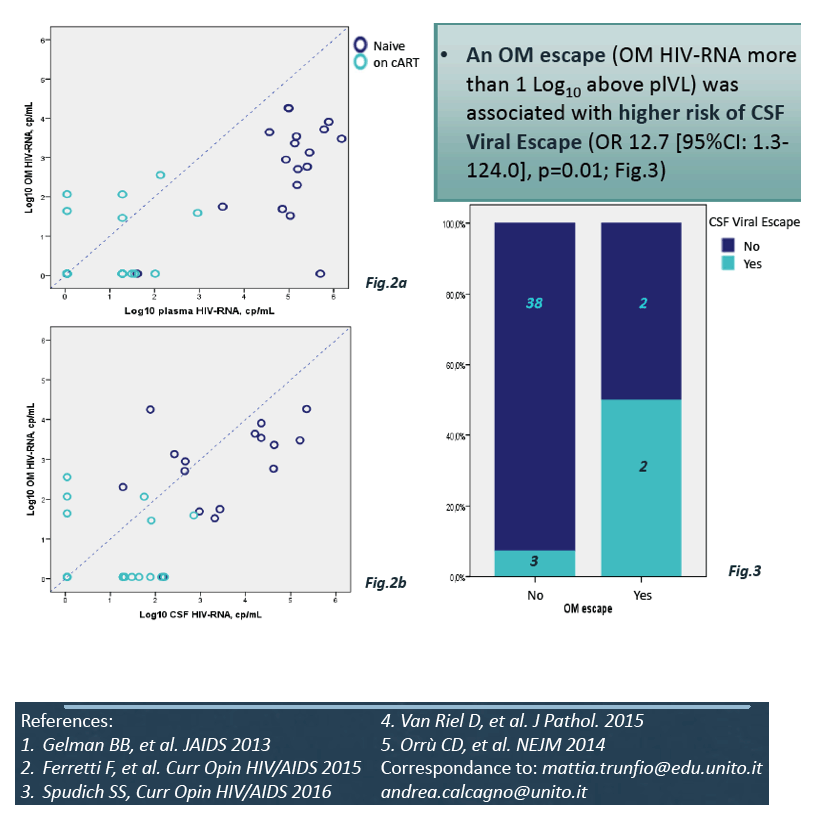
Olfactory mucosa viral escape, defined as olfactory HIV RNA more than 10-fold above plasma viral load, was associated with almost 13-fold higher odds of CSF viral escape (odds ratio 12.7, 95% confidence interval 1.3 to 124.0, P = 0.01).
The researchers proposed that nasal brushing allows safe, noninvasive collection of olfactory mucosa cells, including olfactory neurons. They see a need for further study to determine whether olfactory mucosa HIV RNA can stand as a reliable surrogate for CSF/brain tissue HIV RNA and whether it can identify CSF escape. The association between olfactory mucosa load with HIV DNA and neopterin, they added, "is promising as a marker of residual viral burden and immune activation."
Reference
1. Trunfio M, Allice T, Bertero L, et al. HIV-RNA in the olfactory mucosa of HIV-positive patients. 25th Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2018. Boston. Abstract 443.


Trunfio M1, AlliceT2, BerteroL3, Imperiale D4, CateraS5, PirriatoreV1, Di PerriG1, Amasio E5, Cassoni P3, GhisettiV2, Bonora S1, Calcagno A1
1 Unit of Infectious Diseases, Amedeodi Savoiahospital, Department of Medical Sciences, University of Turin, Turin, Italy-2 Laboratory of Microbiology and Virology, Amedeodi Savoiahospital, Turin, Italy
3 Pathology Division, AOU CittadellaSalute e dellaScienzadi Torino, Department of Medical Sciences, University of Turin, Turin, Italy -4 Unit of Neurology, Maria Vittoria hospital, ASL Citta di Torino, Turin, Italy
5 Unit of Otorhinolaringoiatry, Maria Vittoria hospital, ASL Citta di Torino, Turin, Italy

Program Abstract:
Central nervous system (CNS) HIV infection is assessed on cerebrospinal fluid (CSF) through lumbar puncture (LP), although CSF represents a proxy of brain tissue and LP is a non risk-free procedure. Since olfactory mucosa (OM) is the only CNS tissue that is easily accessible, nasal brushing (NB) may represent an alternative safe diagnostic technique to evaluate CNS viral compartmentalization
Naive and on cART HIV-positive patients undergoing LP for clinical reasons were included. After informed consent, patients underwent (<72 hours from the LP) a NB; OM swabs were inserted in Copan UTM viral transport medium. CSF and plasma HIV-RNA (plVL) were quantified with CAP/CTM v.2.0 HIV-1 (Roche Molecular, USA, detection limit 20 copies/mL). OM HIV-RNA was measured with a modified CAP/CTM procedure. Immunovirological data and CSF biomarkers [ELISA: tau, ptau, neopterin, S100β; immunoturbidimetric method: Reiber diagrams] were recorded. Data are reported as medians (interquartile ranges) and analyzed through non-parametric tests
47 patients were included (74.5% male, 85.1% Caucasian, median age 51 years [47-57]). 19 patients were naïve (plVL and CSF HIV-RNA 5.2 and 3.3 Log10 copies/mL; current CD4 44 cell/uL), while 28 on cART (plVL and CSF HIV-RNA undetectable and 1.3 Log10 copies/mL; current and nadir CD4 456 and 177 cell/uL). CSF escape (CSFE) was observed in 5 patients (10.6%). Mild discomfort, sneezing and lacrimation were the only reported side effects for NB. OM HIV-RNA was detectable in 23 patients (17 naïve: 3.1 Log10 copies/mL [2.0-3.7]; 6 on cART: 1.9 Log10 copies/mL [1.6-2.1]) and correlated with current CD4 count (ρ -.509, p<.01), Log10 plVL and CSF HIV-RNA (ρ .746, p<.01 and ρ .663, p<.01), PBMC HIV-DNA (ρ .582, p<.01) and CSF neopterin (ρ .671, p<.01). OM HIV-RNA was higher than plVL in 5 treated patients (4 aviremic including 2 CSFE) and than CSF HIV-RNA in 5 naïve and 4 treated patients (3 aviremic including 1 CSFE). An OM escape (OM HIV-RNA more than 1 Log10 above plVL) was associated with higher risk of CSFE (OR 12.7, p=.01)
Non-invasive monitoring of HIV tissue replication may have major implications; OM HIV-RNA has been safely measured in around half NB samples. The reported association with HIV-DNA and neopterin is promising as a marker of residual viral burden and immune activation. Further studies are required to establish whether OM HIV-RNA may be a reliable surrogate of CSF/brain tissue HIV-RNA

|
| |
|
 |
 |
|
|