 |
 |
 |
| |
FDA approves new HIV treatment (Long-Acting Ibalizumab) for patients who have limited treatment options
|
| |
| |
LONG-ACTING IBALIZUMAB IN PATIENTS WITH MULTI-DRUG RESISTANT HIV-1: A 24-WEEK STUDY
http://www.natap.org/2017/CROI/croi_20.htm
FDA News Release
FDA approves new HIV treatment for patients who have limited treatment options
Today, the U.S. Food and Drug Administration approved Trogarzo (ibalizumab-uiyk), a new type of antiretroviral medication for adult patients living with HIV who have tried multiple HIV medications in the past (heavily treatment-experienced) and whose HIV infections cannot be successfully treated with other currently available therapies (multidrug resistant HIV, or MDR HIV).Trogarzo is administered intravenously once every 14 days by a trained medical professional and used in combination with other antiretroviral medications.
"While most patients living with HIV can be successfully treated using a combination of two or more antiretroviral drugs, a small percentage of patients who have taken many HIV drugs in the past have multidrug resistant HIV, limiting their treatment options and putting them at a high risk of HIV-related complications and progression to death," said Jeff Murray, M.D., deputy director of the Division of Antiviral Products in the FDA's Center for Drug Evaluation and Research. "Trogarzo is the first drug in a new class of antiretroviral medications that can provide significant benefit to patients who have run out of HIV treatment options. New treatment options may be able to improve their outcomes."
The safety and efficacy of Trogarzo were evaluated in a clinical trial of 40 heavily treatment-experienced patients with MDR HIV-1 who continued to have high levels of virus (HIV-RNA) in their blood despite being on antiretroviral drugs. Many of the participants had previously been treated with 10 or more antiretroviral drugs. The majority of participants experienced a significant decrease in their HIV-RNA levels one week after Trogarzo was added to their failing antiretroviral regimens. After 24 weeks of Trogarzo plus other antiretroviral drugs, 43 percent of the trial's participants achieved HIV RNA suppression.
The clinical trial focused on the small patient population with limited treatment options and demonstrated the benefit of Trogarzo in achieving reduction of HIV RNA. The seriousness of the disease, the need to individualize other drugs in the treatment regimen, and safety data from other trials were considered in evaluating the Trogarzo development program.
A total of 292 patients with HIV-1 infection have been exposed to Trogarzo IV infusion. The most common adverse reactions to Trogarzo were diarrhea, dizziness, nausea and rash. Severe side effects included rash and changes in the immune system (immune reconstitution syndrome).
The FDA granted this application Fast Track, Priority Review and Breakthrough Therapy designations. Trogarzo also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
The FDA granted approval of Trogarzo to TaiMed Biologics USA Corp.
The FDA, an agency within the U.S. Department of Health and Human Services, protects the public health by assuring the safety, effectiveness, security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices. The Agency is also responsible for the safety and security of our nation's food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products.
------------------------
TMB-355 i.v. Clinical Results
Phase 3 Trial
http://www.taimedbiologics.com/pipeline/33
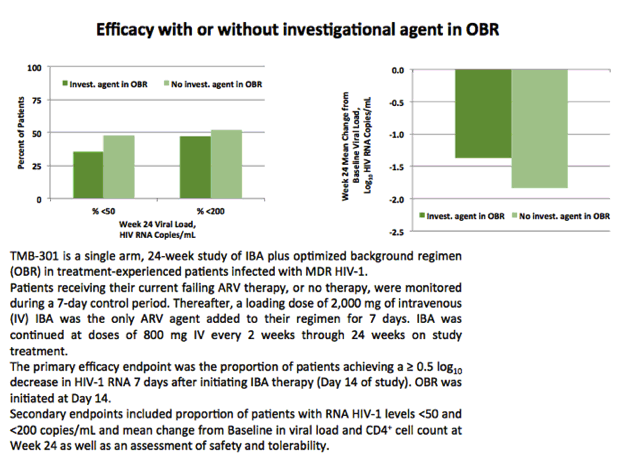
The Phase 3 trial was a single-arm, 24-week study of ibalizumab plus optimized background regimen (OBR) in 40 treatment-experienced patients infected with multi-drug resistant HIV-1. The primary objective of the study was to demonstrate the antiviral activity of ibalizumab seven days after the first dose of ibalizumab. Patients receiving their current failing antiretroviral therapy (ART), or no therapy, were monitored during a 7-day control period. Thereafter a loading dose of 2,000mg of i.v. ibalizumab was the only ART added to their regimen. The primary efficacy endpoint was the proportion of patients achieving a ≥ 0.5 log10 decrease in HIV-1 RNA seven days after initiating ibalizumab therapy, day 14 of the study. Ibalizumab was continued at doses of 800 mg i.v. every two weeks through 24 weeks on study treatment.
The primary endpointshowed the 82.5% of patients achieved more than ~3-fold reduction (0.5 log reduction) in vial load 7 days after loading dose. A significant decrease in viral load after receiving a loading dose of ibalizumab 2,000 mg intravenously in addition to their failing ART (or no therapy). At the completion of the 24-week study, the viral load decreases were maintained for a period of 23 weeks after starting the OBR. After 24 weeks of treatment, the mean reduction in viral load was 1.6 log10 (to 3% of baseline viral load) and a total of 48% of patients had a reduction in viral load of more than 2.0 log10 (to 1% of baseline viral load). At the end of the treatment period the proportion of study participants with undetectable viral load (HIV-1 <50 copies/mL) was 43% (mean viral load reduction of 3.1 log10, to 0.08% of baseline viral load) and 50% of patients had a viral load lower than 200 copies/mL.
A mean increase in CD4+ T cell of 48 cells/μL was observed. When subdivided by CD4+ cell count at baseline, patients with a lower CD4+ T cell count at baseline those with the lowest count (<50 CD4+ T cells/μL, 17 patients) had an increase of 9, those with 50-200 CD4+ T cells/μL (10 patients) had an increase of 75 cells/μL and those with >200 CD4+ T cells/μL (13 patients) had an increase of 78 cells/ μL. The safety results in this Phase 3 trial are consistent with previously observed in the Phase 2b study. Other than for one case of immune reconstitution inflammatory syndrome, an inflammatory response in HIV-infected patients that may be triggered after changing to more active antiretroviral therapy (ART), no serious adverse events (SAEs) were considered to be related to ibalizumab. Most treatment-emergent adverse events reported were mild to moderate in severity. No notable trends in laboratory abnormalities were observed. Additionally, no anti-ibalizumab antibodies were detected in any blood samples of patients.
Patients enrolled in the Phase III trial had high pre-existing levels of drug resistance and advanced clinical disease. Patients had a mean HIV-1 viral load of 100,287 copies/mL, with 18% having viral loads above 100,000 copies/mL. The median CD4 count was 73 cells/mm3 and nearly 30% had less than 10 CD4 cells/mm3. More than 85% of patients had more than one identified mutation conferring resistance to the Nucleoside Reverse Transcriptase Inhibitors (NRTI), Non Nucleoside Reverse Transcriptase Inhibitors (NNRTI) or Protease Inhibitors (PI) and more than 60% of patients had resistance to at least one Integrase Inhibitor (INI). Study patients were infected with HIV-1 resistant to more than 75% of all drugs in the NRTI, NNRTI and PI classes and to 1-2 drugs from the INI class, on average. Finally, just over 50% of patients had HIV-1 with resistance to all available drugs from at least three classes of ART.
The Expanded Access Program (EAP) for ibalizumab in Resistant HIV-1 is currently recruiting in the U.S. For more details, please visit the ibalizumab EAP website.
|
| |
|
 |
 |
|
|