 |
 |
 |
| |
Protection against repeated vaginal SHIV challenges by bNAb 3BNC117 and 10-1074
|
| |
| |
One bNAb Protects Macaques From Vaginal SHIV--Two bNAbs Protect Longer
25th Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2018, Boston
Mark Mascolini
A single subcutaneous dose of a broadly neutralizing antibody (bNAb) protected macaques from weekly intravaginal challenge with simian HIV (SHIV) for a median of 5 weeks [1]. Single doses of two bNAbs extended that protection to a median of 11.5 weeks.
Besides experimental antiretrovirals, passive immunization with bNAbs offers another avenue to long-acting preexposure prophylaxis (PrEP) against HIV infection. Two newer-generation bNAbs, 3BNC117 and 10-1074, proved generally safe and well tolerated in phase 1 trials enrolling people with or without HIV. Single 20-mg/kg intravenous injections of 3BNC117 or 10-1074 durably protected macaques against rectal SHIV exposure [2]. CDC researchers and collaborators from other centers conducted a new study to assess those bNAbs at a lower dose, singly and together. 3BNC117 targets the CD4 binding site and 10-1074 homes to the V3 glycan supersite on HIV Env.
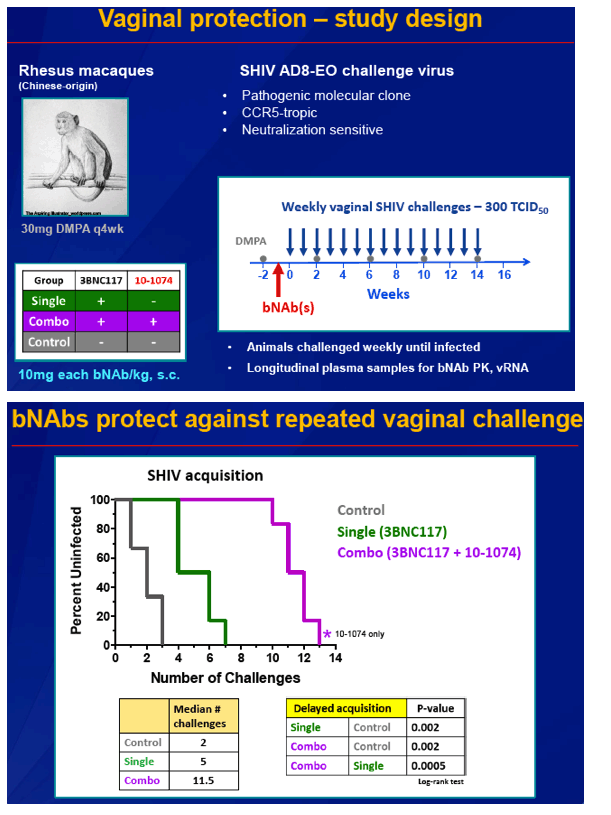
To normalize SHIV susceptibility, animals received 30 mg of DMPA intramuscularly 2 weeks before the first intravaginal SHIV challenge and every 4 weeks after that. One week before SHIV challenge, the monkeys got a single subcutaneous 10-mg/kg dose of 3BNC117 alone or that bNAb plus the same dose of 10-1074. Then they had weekly intravaginal SHIV challenges at a dose of 300 TCID50. Challenge continued until a plasma viral load assay confirmed systemic SHIV infection. For each of the two experiments, 6 animals got one or both bNAbs and 3 control animals got challenged with no bNAb. The researchers also used regular plasma samples to measure bNAb concentrations.
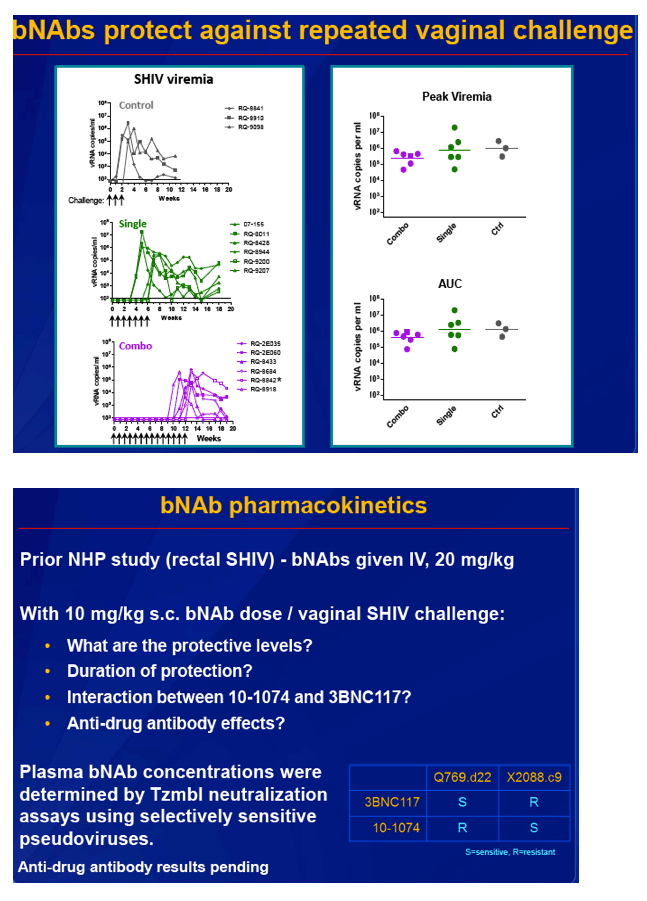
Untreated control animals became infected after a median of 2 intravaginal challenges, animals treated with 3BNC117 alone became infected after a median of 5 challenges (P = 0.002 versus controls), and animals treated with both 3BNC117 and 10-1074 became infected after a median of 11.5 challenges (P = 0.0005 versus 3BNC117 alone, P = 0.002 versus controls).
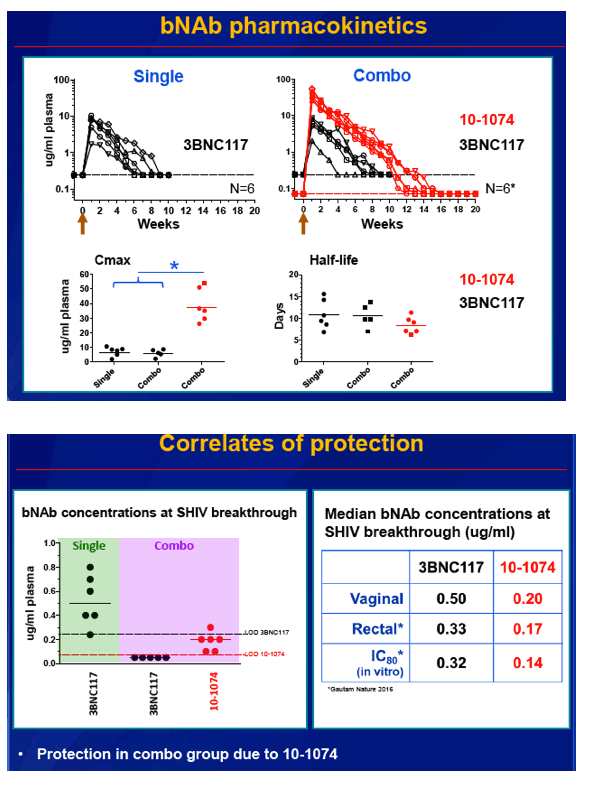
Median bNAb concentrations at SHIV infection were 0.50 ug/mL for 3BNC117 and 0.20 ug/mL for 10-1074. Those values compared with 0.33 ug/mL for 3BNC117 and 0.17 ug/mL for 10-1074 in the rectal challenge study [2]. Estimated plasma half-life was similar for the two bNAbs. But average maximum concentration (Cmax) was 6 times higher for 10-1074 than for 3BNC117 (38.8 versus 6.5 ug/mL, P = 0.001). As a result, more 10-1074 stayed in circulation than 3BNC117 and accounted for the greater protection against SHIV seen with two bNAbs than one.
The researchers believe their findings support continued clinical development of 10-1074 and 3BNC117 as PrEP for women.
References
1. Garber DA, Adams D, Mitchell J, et al. Protection against repeated vaginal SHIV challenges by bNAb 3BNC117 and 10-1074. 25th Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2018. Boston. Abstract 82.
2. Gautam R, Nishimura Y, Pegu A, et al. A single injection of anti-HIV-1 antibodies protects against repeated SHIV challenges. Nature. 2016;533:105-109.
WEBCAST: http://www.croiwebcasts.org/console/player/37184?mediaType=slideVideo&&crd_fl=1&ssmsrq=1520966409899&ctms=5000&csmsrq=942
CROI: Cabotegravir long-acting protects macaques against repeated penile SHIV exposures (03/12/18)
CROI: Oral TAF/FTC PrEP Prevents Vaginal SHIV Infection in Monkeys - Mark Mascolini (03/09/18)
Three-in-One Antibody Protects Monkeys from SHIV - HIV-Like Virus - (09/20/17)
ACTG STUDY - Protocol A5377: First-in-humans Tri-specific Antibody. A Phase 1, First-in-human, Single Ascending Dose Study of SAR441236, a Tri-specific Broadly Neutralizing Antibody



|
| |
|
 |
 |
|
|