 |
 |
 |
| |
Phase III Randomized, Controlled Clinical Trial of Bictegravir Coformulated with FTC/TAF in a Fixed-dose Combination (B/F/TAF) versus Dolutegravir (DTG) + F/TAF in Treatment-naïve HIV-1 Positive Adults: Week 96
|
| |
| |
Bictegravir/F/TAF Noninferior to DTG Plus F/TAF at 96 Weeks: No Resistance
HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow
Mark Mascolini
A single-tablet combination of bictegravir with emtricitabine and tenofovir alafenamide (B/F/TAF) proved virologically noninferior to dolutegravir (DTG) plus F/TAF at 96 weeks in 645 previously untreated people [1]. No resistant HIV emerged in any study participants.
European and US regulators have licensed single-tablet B/F/TAF, though bictegravir (an integrase inhibitor) is not available as a stand-alone antiretroviral [2]. Six previous trials have found B/F/TAF noninferior to contemporary comparator regimens, including single-tablet dolutegravir/abacavir/lamivudine.
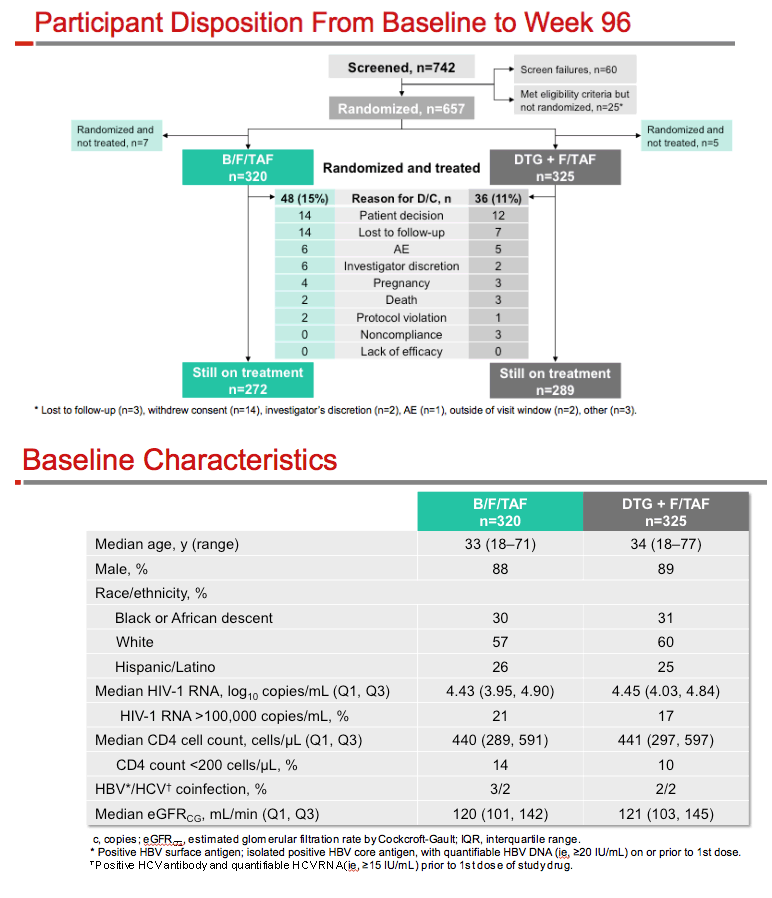
GS-US-380-1490 is a placebo-controlled trial that randomized 645 antiretroviral-naive people to once-daily B/F/TAF or DTG+F/TAF. Median ages in the B/F/TAF and DTG arms were 33 and 34, proportions of men 88% and 89%, and proportions of blacks 30% and 31%. Median pretreatment viral loads were similar in the B/F/TAF and DTG arms, 4.43 and 4.45 log10 copies (about 27,500 copies), and about 20% of participants had a pretreatment load above 100,000 copies. Analysis of the primary endpoint, proportion of participants with a week-48 viral load below 50 copies, established the virologic noninferiority of B/F/TAF to DTG+F/TAF (89.4% versus 92.9%, P = 0.12) [3].
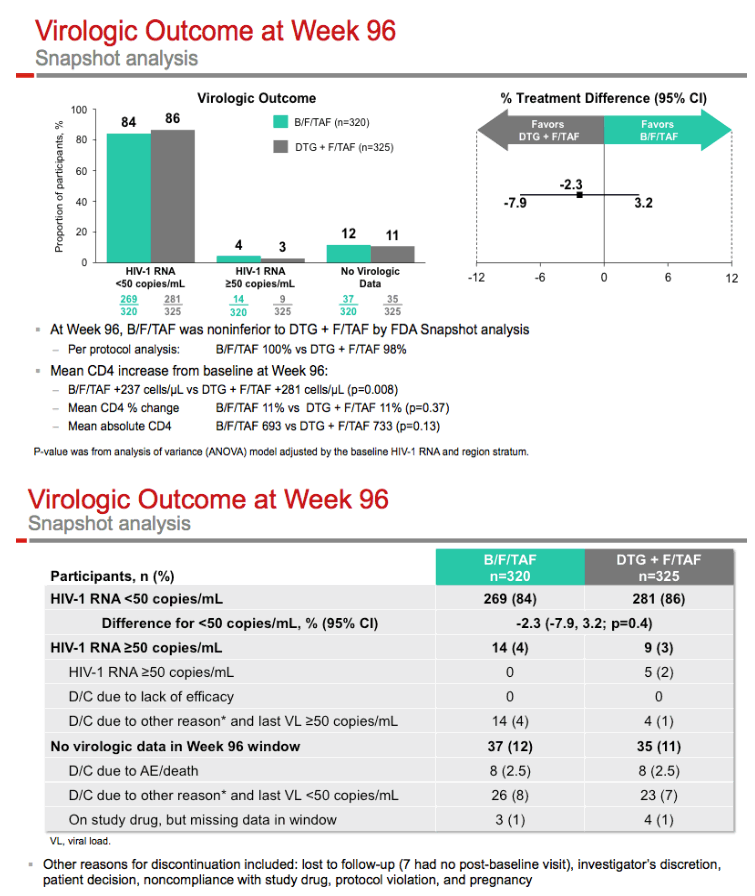
After 96 weeks, 84.1% randomized to B/F/TAF and 86.5% randomized to DTG+F/TAF had a viral load under 50 copies, a result confirming the continued noninferiority of B/F/TAF to the other regimen (difference -2.3%, 95% confidence interval -7.9% to 3.2%, P = 0.41). At week 96, no one randomized to B/F/TAF and 5 randomized to the DTG combination had a viral load above 50 copies, but resistant HIV emerged in no one in either study arm. In a per-protocol analysis, 100% assigned to B/F/TAF and 98% assigned to DTG+F/TAF had a sub-50-copy viral load. Average CD4 counts rose 11% with both regimens.
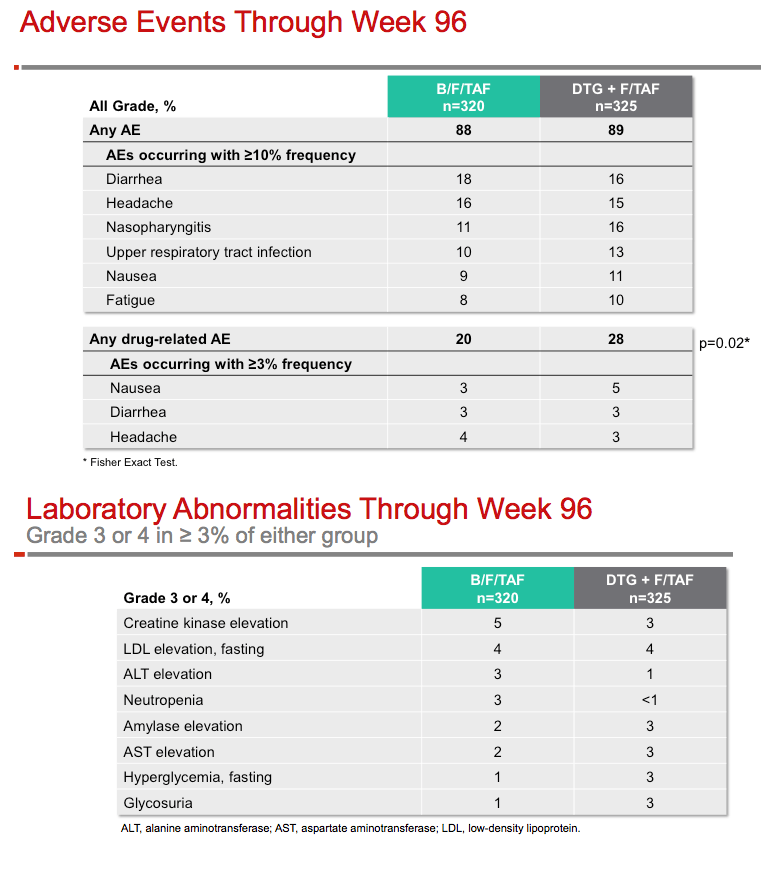
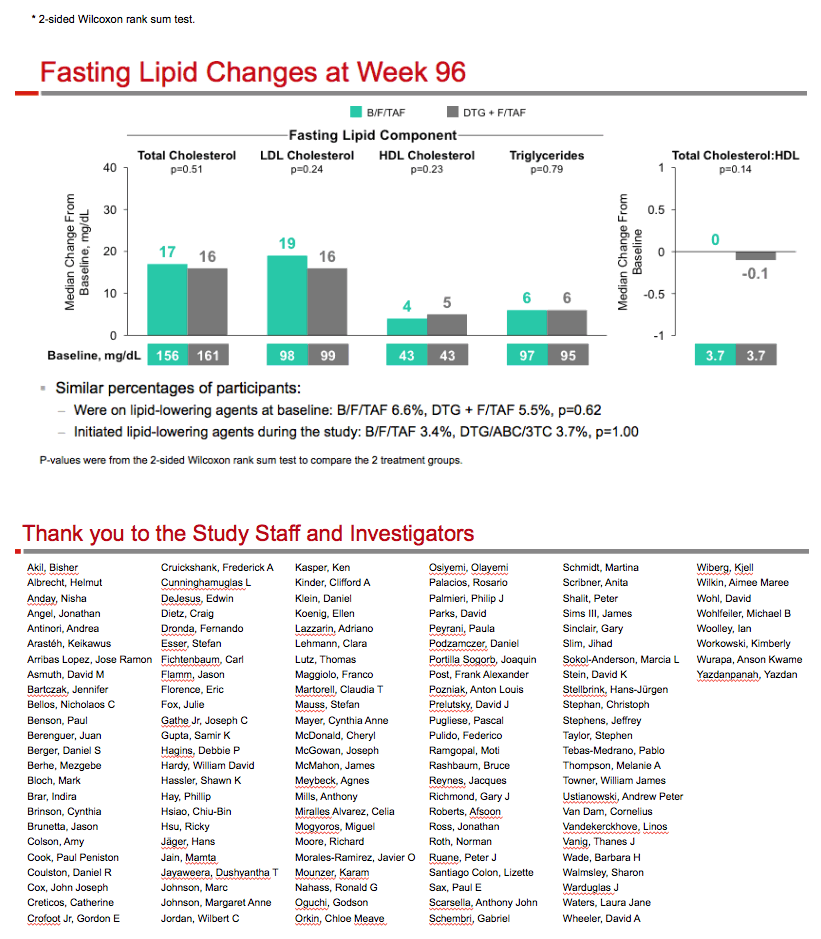
Through 96 weeks, 6 people in the B/F/TAF arm (1.9%) and 5 in the DTG+F/TAF arm (1.6%) stopped study drugs because of an adverse event. Implicated events in the B/F/TAF group were cardiac arrest, atypical chest pain, abdominal distention, sleep disorder, paranoia, and depression. The most frequent drug-related adverse events were nausea, diarrhea, and headache, each affecting 5 or fewer people in each arm. The most frequent grade 3 or 4 lab abnormalities were rising creatine kinase (5 B/F/TAF and 3 DTG+F/TAF), climbing low-density lipoprotein (LDL) cholesterol (4 and 4), and alanine aminotransferase elevation (3 and 1). Lipid elevations were similar in the two treatment groups (LDL +19 mg/dL with B/F/TAF, +16 mg/dL with DTG+F/TAF).
References
1. Stellbrink H, Arribas J, Stephens J, et al. Phase III randomized, controlled clinical trial of bictegravir coformulated with FTC/TAF in a fixed-dose combination (B/F/TAF) versus dolutegravir (DTG) + F/TAF in treatment-naive HIV-1 positive adults: week 96. HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow. Abstract O211.
2. US Department of Health and Human Services. AIDSinfo. Bictegravir. https://aidsinfo.nih.gov/drugs/570/bictegravir/0/professional
3. Sax PE, Pozniak A, Montes ML, et al. Coformulated bictegravir, emtricitabine, and tenofovir alafenamide versus dolutegravir with emtricitabine and tenofovir alafenamide, for initial treatment of HIV-1 infection (GS-US-380-1490): a randomised, double-blind, multicentre, phase 3, non-inferiority trial. Lancet. 2017;390:2073-2082.
Phase III Randomized, Controlled Clinical Trial of Bictegravir Coformulated with FTC/TAF in a Fixed-dose Combination (B/F/TAF) versus Dolutegravir (DTG) + F/TAF in Treatment-naïve HIV-1 Positive Adults: Week 96
Hans Jurgen Stellbrink,1 Jose Arribas,2 Jeffrey L. Stephens,3 Helmut Albrecht,4 Paul E. Sax,5 Franco Maggiolo,6 Catherine Creticos,7 Claudia T. Martorell,8 Xuelian Wei,9 Kirsten White,9 Sean E. Collins,9 Andrew Cheng,9 Hal Martin9
1ICH Study Center, Hamburg, Germany; 2Hospital Universitario La Paz, Madrid, Spain; 3Mercer University School of Medicine, Macon, GA, US; 4Palmetto Health , Richland, SC, US; 5Brigham and Women's Hospital, Boston, MA, US; 6Azienda Ospedaliera Papa Giovanni XXIII, Bergamo, Italy; 7Howard Brown Health Center, Chicago, IL. US; 8Infectious Diseases and The Research Institute, Springfield, MA, US; 9Gilead Sciences, Inc., Foster City, CA



|
| |
|
 |
 |
|
|