 |
 |
 |
| |
EACS 2019: ViiV Healthcare to present 17 abstracts from its portfolio addressing the diverse needs of people living with HIV - PRESS RELEASE
|
| |
| |
Download the PDF here
This is shortened version of press release. Please see attached pdf for full press release including safety etc.
Data presentations expand understanding of ViiV Healthcare's pipeline for heavily treatment-experienced populations, and look deeper into its 2-drug regimen studies
London, 6 November 2019 - ViiV Healthcare, the global specialist HIV company majority-owned by GSK, with Pfizer Inc. and Shionogi Limited as shareholders, announced today that 17 abstracts from its portfolio of late-stage pipeline and approved HIV treatments will be presented at the 17th European AIDS Conference (EACS 2019) in Basel, Switzerland, 6-9 November.
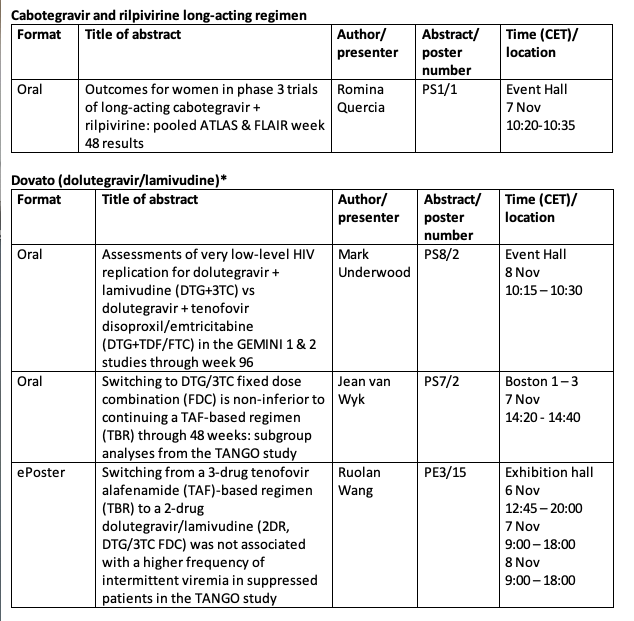
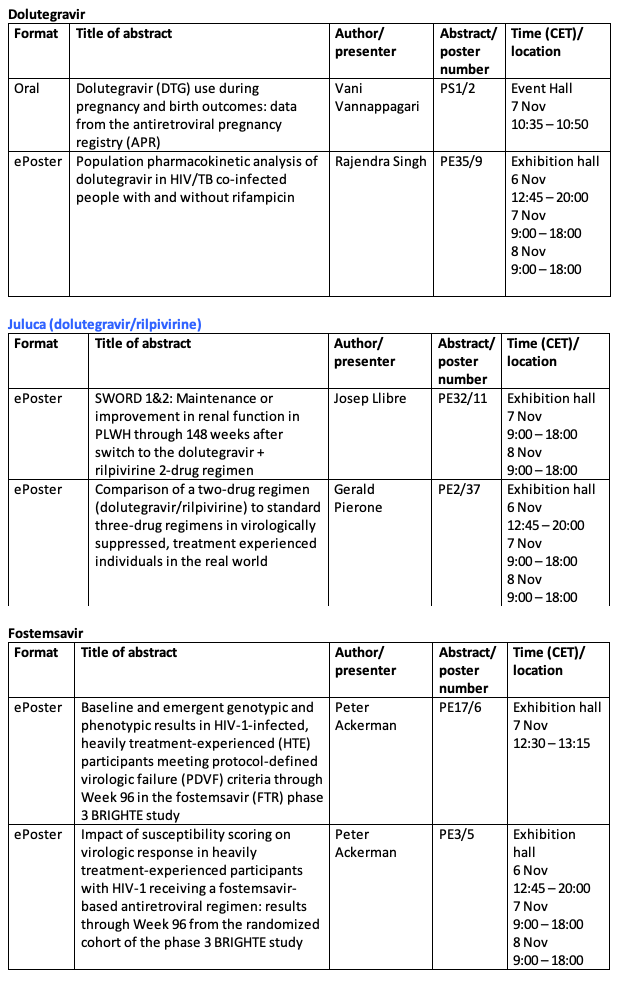
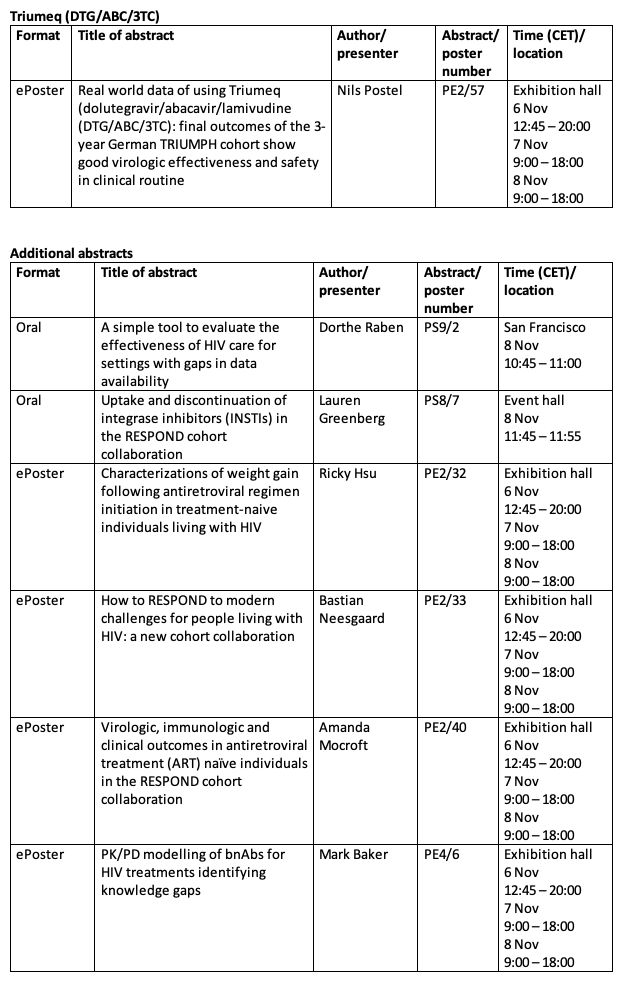
Advances in research and development combined with the availability of effective medicines are changing the HIV landscape beyond 3-drug regimens which have been the mainstay of HIV treatment. ViiV Healthcare has been an integral part of this shift with the development of the 2-drug regimens (2DR) Dovato[1] (dolutegravir/lamivudine) and in partnership with Janssen, Juluca (dolutegravir/rilpivirine). Together with Janssen, ViiV healthcare is also working on cabotegravir and rilpivirine long-acting, an investigational long-acting injectable regimen. We are continuing to expand our innovative treatment portfolio by developing fostemsavir, a first-in-class treatment for people living with HIV (PLHIV) who have very few or no treatment options left available to them.
Kimberly Smith, MD, Head of Research & Development at ViiV Healthcare, said: "Since ViiV Healthcare's inception 10 years ago, we have used our experience in drug development and the time we have spent getting to know people living with HIV to create research into innovative treatments that meet their diverse and changing needs. At EACS 2019, we are presenting data that take a deeper look at how different populations, including women and those who are heavily treatment-experienced, can benefit from our treatment innovations, to give us a fuller understanding of outcomes in these populations."
About cabotegravir
Cabotegravir is an investigational integrase inhibitor (INI) and is not approved by regulatory authorities anywhere in the world. Cabotegravir is being developed by ViiV Healthcare for the treatment and prevention of HIV. It is being evaluated as a long-acting formulation for intramuscular injection and also as a once-daily oral tablet for use as a lead-in, to establish the tolerability of cabotegravir prior to long-acting injection.
About rilpivirine long-acting
Rilpivirine long-acting is an investigational, prolonged-release suspension for intramuscular injection being developed by Janssen Sciences Ireland UC, one of the Janssen Pharmaceutical Companies of Johnson & Johnson, and is not approved by regulatory authorities anywhere in the world.
About rilpivirine
Rilpivirine is a second-generation non-nucleoside reverse transcriptase inhibitor (NNRTI). It is authorised in the EU in combination with other antiretroviral medicinal products, for the treatment HIV-1.
About fostemsavir
Fostemsavir is an investigational prodrug of temsavir, an HIV-1 attachment inhibitor, and is not authorised by regulatory authorities anywhere in the world. Fostemsavir is being developed by ViiV Healthcare for the treatment of HIV-1-infected heavily treatment-experienced patients in combination with other antiretroviral agents.
About Dovato (dolutegravir/lamivudine)
Dovato (dolutegravir 50 mg/ lamivudine 300 mg tablets) is authorised in the EU for the treatment of HIV-1 infection in adults and adolescents above 12 years of age weighing at least 40 kg, with no known or suspected resistance to the integrase inhibitor class, or lamivudine.[viii] Dolutegravir/lamivudine is a once-daily, single-pill, two-drug regimen that combines the integrase strand transfer inhibitor (INSTI) dolutegravir (Tivicay, 50 mg) with the nucleoside analogue reverse transcriptase inhibitor (NRTI) lamivudine (Epivir, 300 mg).
In the US the Food and Drug Administration (FDA) authorised Dovato, a complete, once-daily, single-tablet regimen of dolutegravir (DTG) 50 mg and lamivudine (3TC) 300 mg for the treatment of HIV-1 infection in adults with no antiretroviral (ARV) treatment history and with no known resistance to either DTG or 3TC.[ix]
Like a dolutegravir-based three-drug regimen, dolutegravir/lamivudine uses two drugs to inhibit the viral cycle at two different sites. INSTIs, like dolutegravir, inhibit HIV replication by preventing the viral DNA from integrating into the genetic material of human immune cells (T-cells). This step is essential in the HIV replication cycle and is also responsible for establishing chronic infection. Lamivudine is an NRTI that works by interfering with the conversion of viral ribonucleic acid (RNA) into deoxyribonucleic acid (DNA) which in turn stops the virus from multiplying.
About GSK
GSK is a science-led global healthcare company with a special purpose: to help people do more, feel better, live longer. For further information please visit www.gsk.com.
|
| |
|
 |
 |
|
|