 |
 |
 |
| |
Week-96 Potency of Dolutegravir/3TC in GEMINI Trials of First-Line ART - "Durable efficacy of dolutegravir (DTG) plus lamivudine (3TC) in antiretroviral treatment-naive adults with HIV-1 infection--96-week results from the GEMINI studies"
|
| |
| |
10th IAS Conference on HIV Science (IAS 2019), July 21-24, 2019, Mexico City
Mark Mascolini
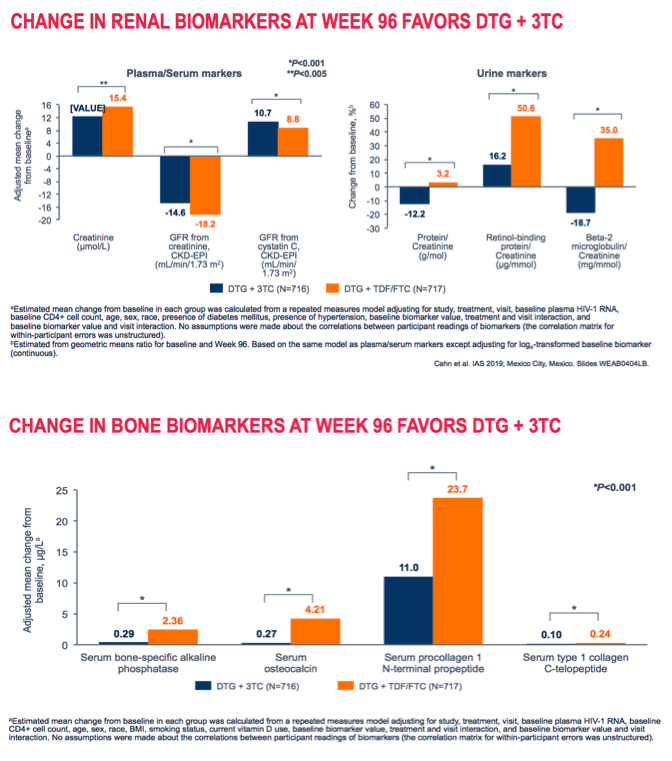
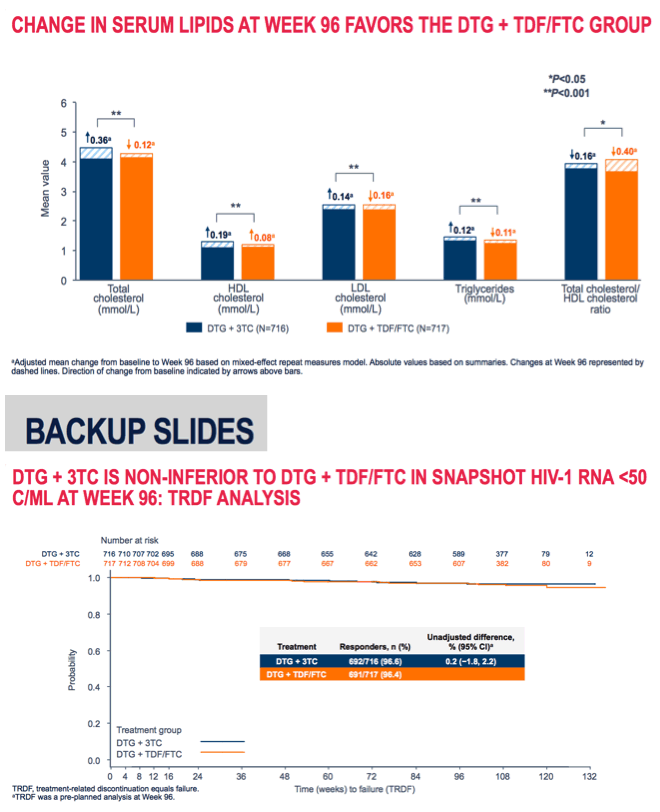
Week-96 results confirmed that dolutegravir/lamivudine (DTG/3TC) remained virologically noninferior to DTG + tenofovir/emtricitabine (TDF/FTC) as a first-line regimen for previously untreated people [1]. Some kidney and bone markers favored DTG/3TC over the 3-drug regimen. Lipid changes tended to favor DTG + TDF/FTC.
The week-48 primary analysis of GEMINI-1 and -2 found DTG/3TC virologically noninferior to
DTG + TDF/FTC in antiretroviral-naive people, with sub-50-copy response rates of 90% or higher in both study arms (intention-to-treat-exposed snapshot analysis) [2]. The FDA and the European Medicines Agency subsequently licensed DTG/3TC as a once-daily single-tablet combination for untreated people with no mutations conferring resistance to DTG or 3TC. Data presented at IAS 2019 updated the 48-week findings through 96 weeks.
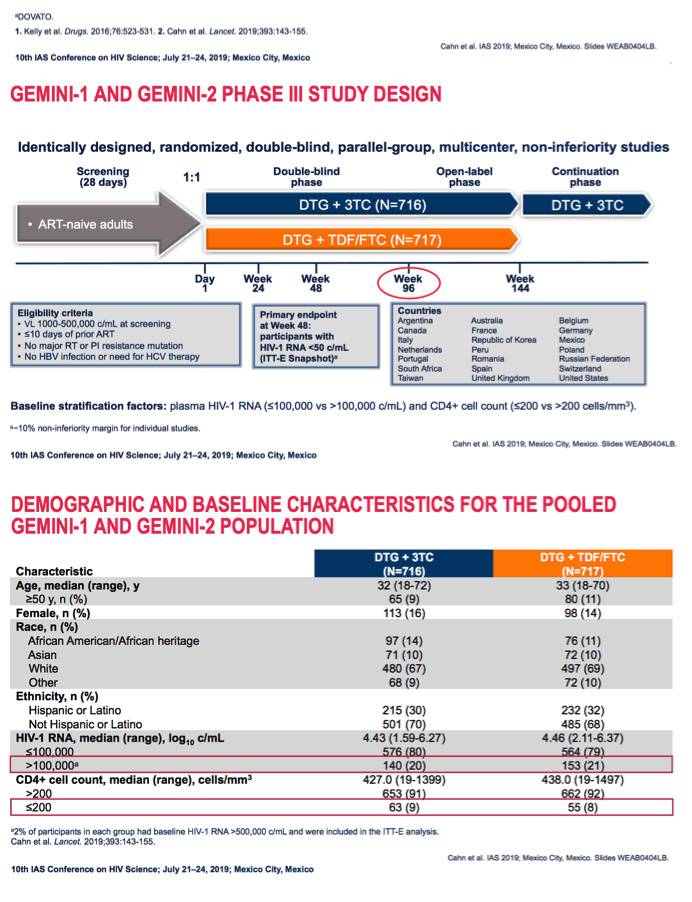
Participants needed a viral load between 1000 and 500,000 copies, 10 or fewer days of prior antiretroviral therapy, no major reverse transcriptase or protease resistance mutations, and no HBV or need for HCV therapy. GEMINI investigators randomized 716 people to DTG/3TC and 717 to DTG + TDF/FTC in a double-blind design.
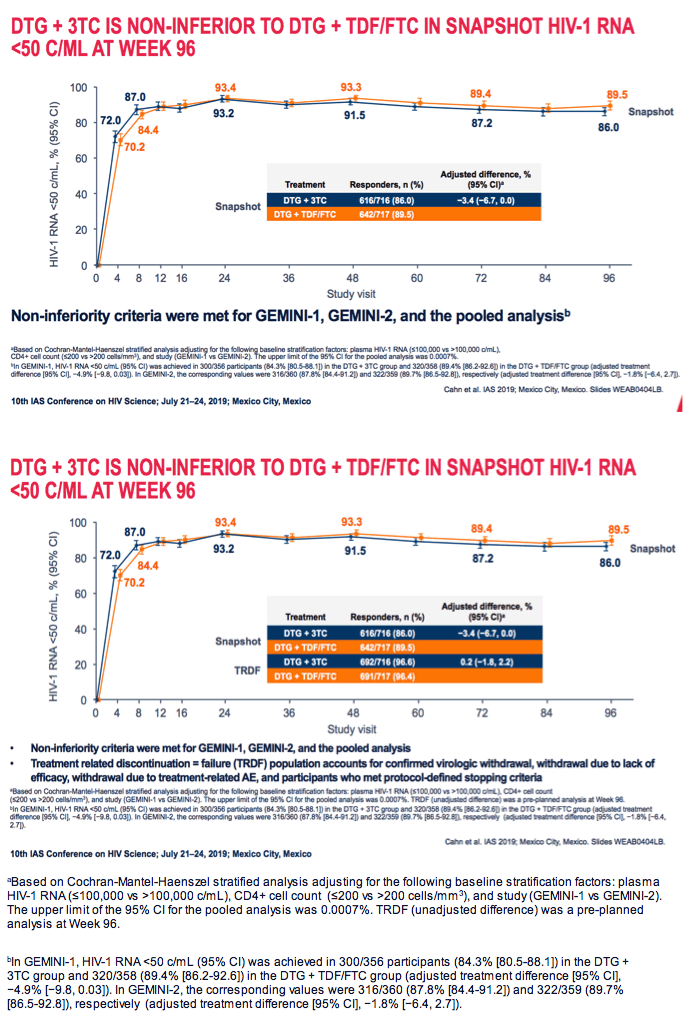
Median ages of the 2- and 3-drug groups were 32 and 33, and only about 10% were 50 or older. About 15% overall were women, and two thirds were white. One fifth had a pretreatment viral load above 100,000 copies, and 9% had a CD4 count below 200. In the pooled snapshot analysis, 91.5% on DTG/3TC and 93.3% on DTG + TDF/FTC had a viral load below 50 copies at week 48. At week 96 those proportions stood at 86.0% and 89.5%.
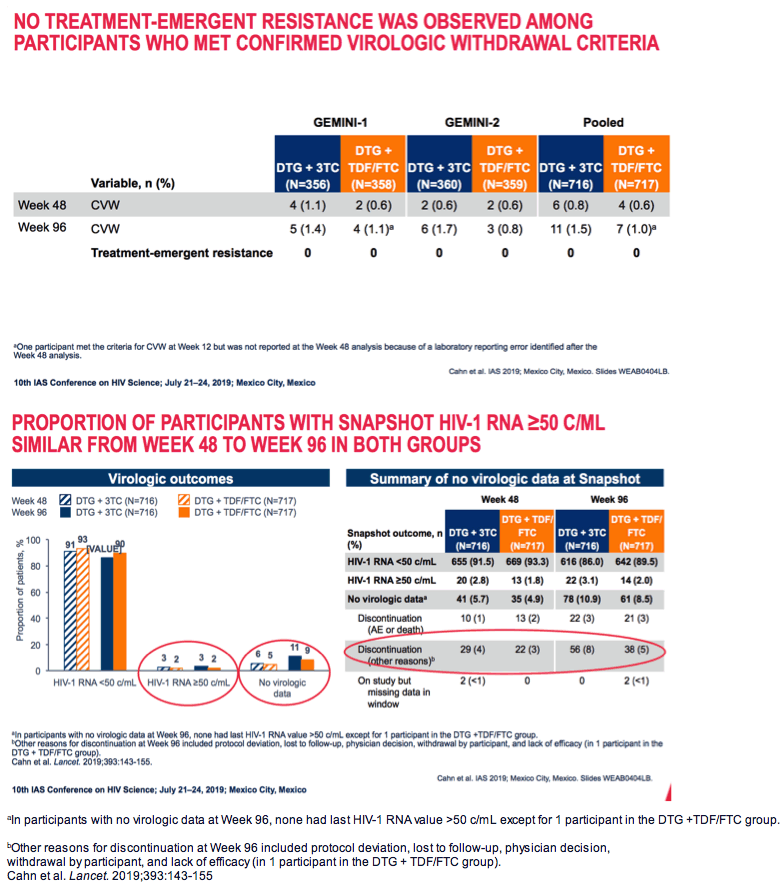
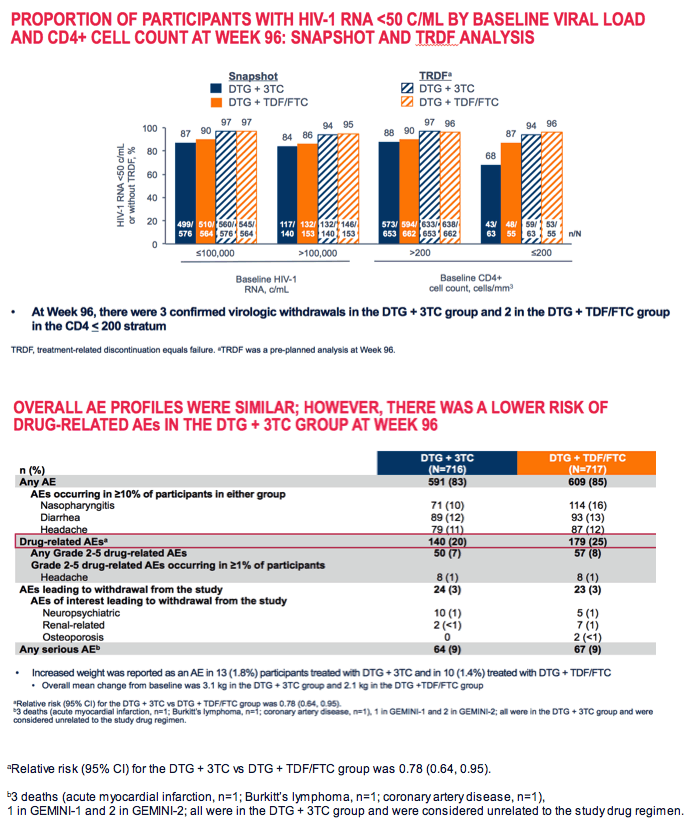
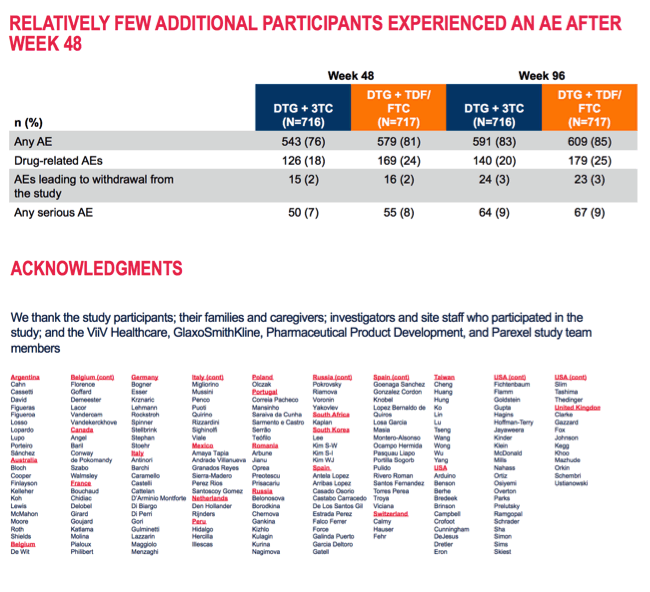
Week-96 discontinuations for adverse events or death stood at 3% in both study arms. Week-96 discontinuations for other reasons came to 8% in DTG/3TC group and 5% in the 3-drug arm. Snapshot virologic response rates at 96 weeks did not differ substantially between the two groups among people with a pretreatment viral load above or below 100,000 copies or in those with an initial CD4 count above 200. But among people with a starting CD4 count below 200, week-96 sub-50-copy response rates were substantially lower with 2 drugs than with 3 (68% versus 87%).
At 96 weeks 11 people in the DTG/3TC arm and 7 in the DTG + TDF/FTC arm met confirmed virologic withdrawal criteria. None of those 18 people had treatment-emergent resistance mutations.
The drug-related adverse event rate was slightly lower with 2 drugs than with 3: 20% versus 25%. But only 3% in each trial arm dropped out because of an adverse event. Serious adverse events arose in 9% in each study arm.
Through 96 weeks changes in 6 kidney function markers significantly favored DTG/3TC, as would be expected in comparison with a TDF-containing regimen. The same was true for 4 bone markers. Week-96 lipid changes tended to favor the 3-drug regimen: total cholesterol, "bad" LDL cholesterol, and triglycerides all rose with DTG/3TC and fell with the 3-drug regimen (P < 0.001 for all). "Good" HDL cholesterol rose significantly more with DTG/3TC than with the other regimen (P < 0.001). Total-to-HDL cholesterol ratio fell through 96 weeks with both regimens, but more with DTG + TDF/FTC (-0.40 versus -0.16, P < 0.05). (A lower ratio predicts a lower risk of heart disease.)
The GEMINI investigators believe their findings further support DTG/3TC as "a compelling option" for HIV therapy.
References
1. Cahn P, Sierra Madero J, Arribas J, et al. Durable efficacy of dolutegravir (DTG) plus lamivudine (3TC) in antiretroviral treatment-naive adults with HIV-1 infection--96-week results from the GEMINI studies. 10th IAS Conference on HIV Science (IAS 2019), July 21-24, 2019, Mexico City. Abstract WEAB0404LB.
2. Cahn P, Madero JS, Arribas JR, et al. Dolutegravir plus lamivudine versus dolutegravir plus tenofovir disoproxil fumarate and emtricitabine in antiretroviral-naive adults with HIV-1 infection (GEMINI-1 and GEMINI-2): week 48 results from two multicentre, double-blind, randomised, non-inferiority, phase 3 trials. Lancet. 2019;393:143-155. http://www.natap.org/2018/HIV/PIIS0140673618324620.pdf


|
| |
|
 |
 |
|
|